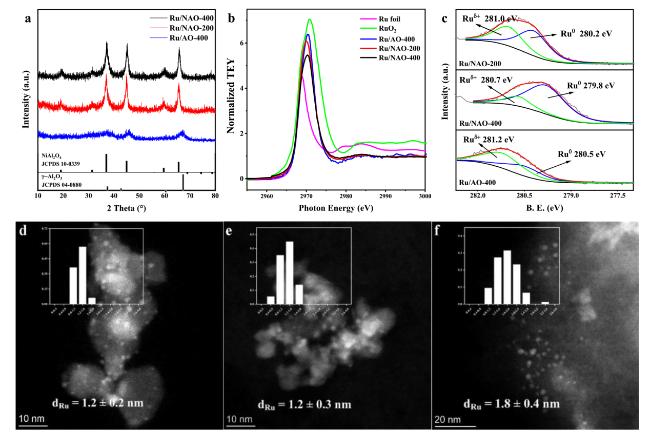
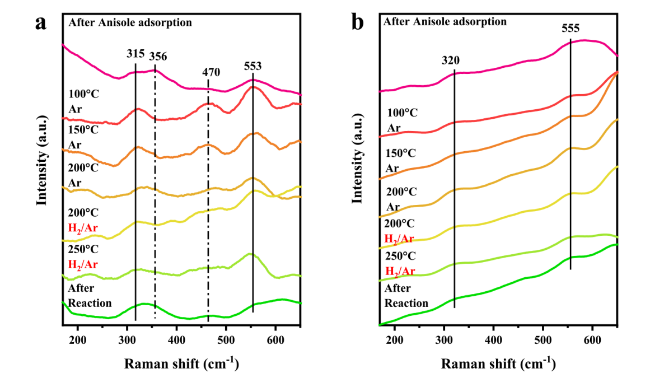
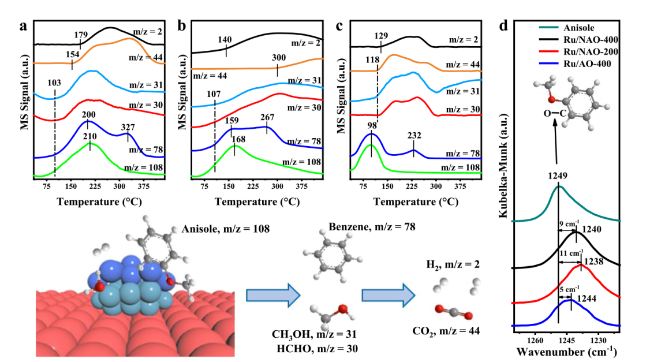
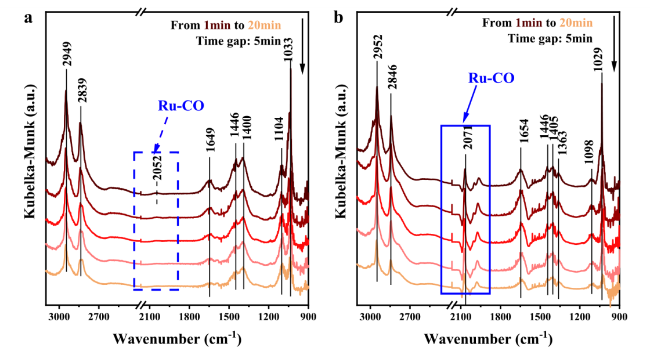
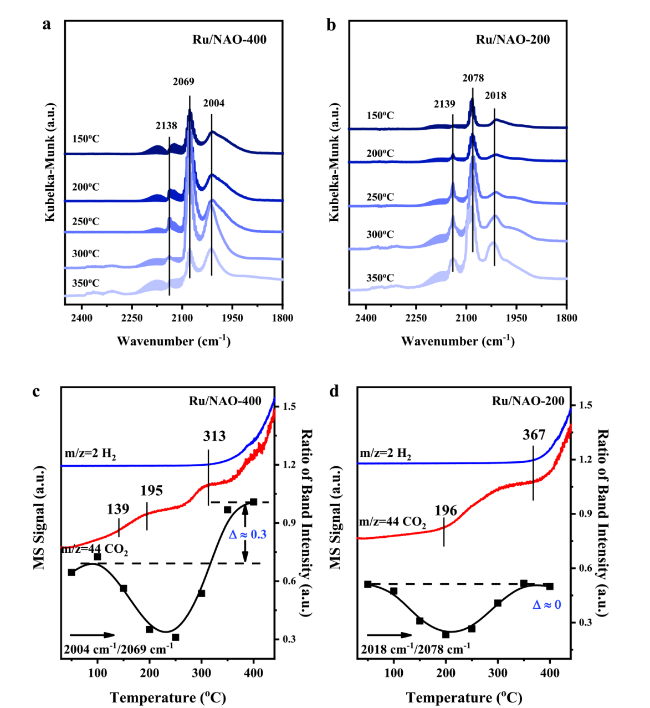
Figure 3a and
b show that the generation temperatures of methanol were quite close over two NiAl
2O
4-based catalysts, which were 103 °C and 107 °C over Ru/NAO-400 and Ru/NAO-200, respectively, suggesting they might possess similar activation ability for C-O bond cleavage. This speculation could also be confirmed via the in-situ DRIFT spectra of anisole adsorption. The bands corresponded to methoxy C-O bond at 1240 cm
−1 and 1238 cm
−1 in
Fig. 3d were both red-shifted compared with gaseous anisole, indicating the existence of strong interaction between the substrate and the catalyst surface. As a contrast in
Fig. 3c, the demethoxylation over Ru/AO-400 initiated from 115 oC, whereas only a slight redshift of 5 cm
−1 in DRIFT was observed, indicating that the activation of C-O bond was less effective over Ru/AO-400. More interestingly, the adsorption of anisole on Ru/AO-400 was weak since the desorption peak of anisole was at 98 °C, right before the starting point of demethoxylation. Besides Ru/AO-400, the desorption peaks of anisole also vary between the other two catalysts, which were 210 oC and 168 oC over Ru/NAO-400 and Ru/NAO-200, respectively, which could be ascribed to the fact that the chemical compositions of Ru, namely the proportion of Ru
0 species, play an important role in the adsorption of the aromatic ring in anisole, which was also mentioned by the previous work [
50]. However, a higher concentration of Ru
0 species can enable the stronger adsorption of the reactant, but may also make it more difficult for the desorption of produced aromatics, which will hinder the recovery of Ru
0 species. These influences can be seen from the second desorption peaks of fragment at m/z = 78 at, 327 °C, 267 °C and 232 °C over Ru/NAO-400、Ru/NAO-200和Ru/AO-400, respectively. Hence, combined with the Raman results, an appropriate Ru
0/Ru
δ+ ratio played vital role in demethoxylation, and Ru/NAO-200 catalyst likely possessed a good ratio of Ru
0/Ru
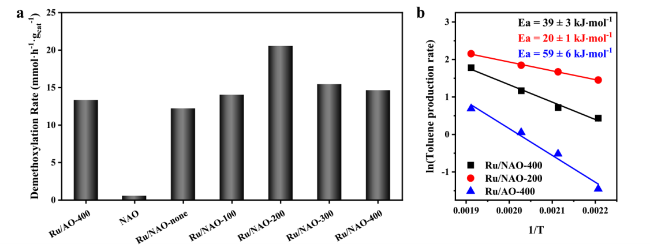
δ+. This analysis is in good agreement with catalytic performance summarized in
Fig. 2. In addition, we also observed the formation of H
2 (m/z = 2) and formaldehyde (m/z = 30) when we raised the temperature, suggesting the decomposition of generated methanol, or methoxy, that later initiated the WGS according to the uplift of CO
2 (m/z = 44) [
34,
38,
51]. Interestingly, the generation temperature of CO
2 was at 300 °C over Ru/NAO-200 in
Fig. 3b, higher than 154°C and 118°C over Ru/NAO-400 and Ru/AO-400, respectively, which could be ascribed to the low WGS rate over Ru/NAO-200. The difference between Ru/AO-400 and Ru/NAO-400 was also thought to be related to the acidity of the support, and the more acidic Ru/AO-400 had lower desorption temperature. Combining the activity evaluation, in-situ Raman, DRIFT and TPSR-MS, we can conclude that the synergy between Ru
0 and Ru
δ+ is crucial for the demethoxylation reaction, i.e., Ru
0 is in charge of the dissociation of H
2 and suitable adsorption of the aromatics, while Ru
δ+ is responsible for the activation of C-O bond.