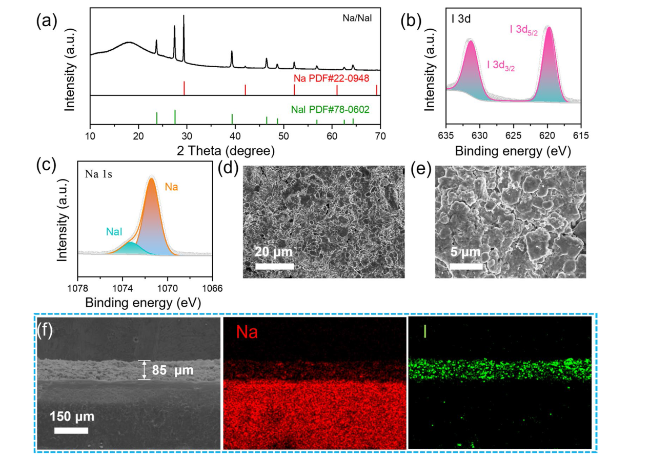
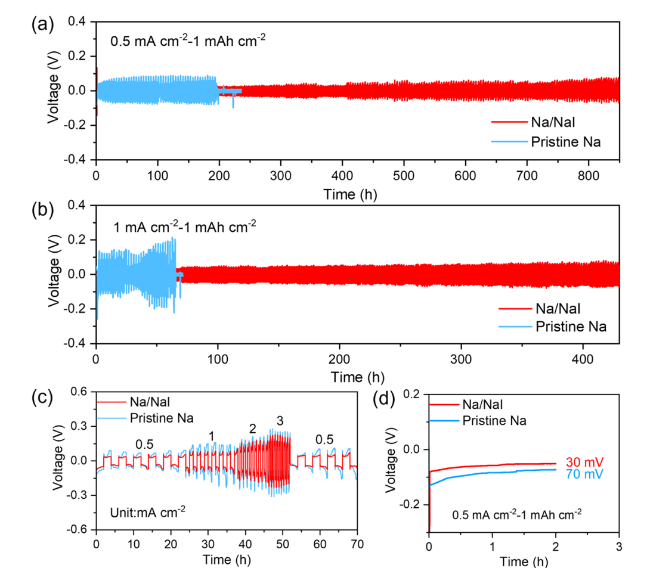
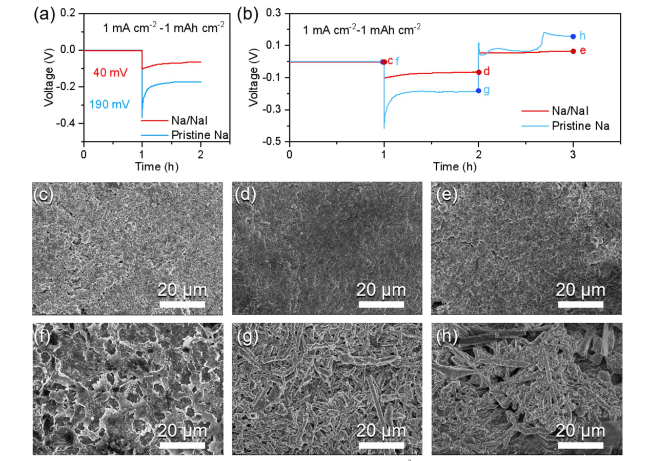
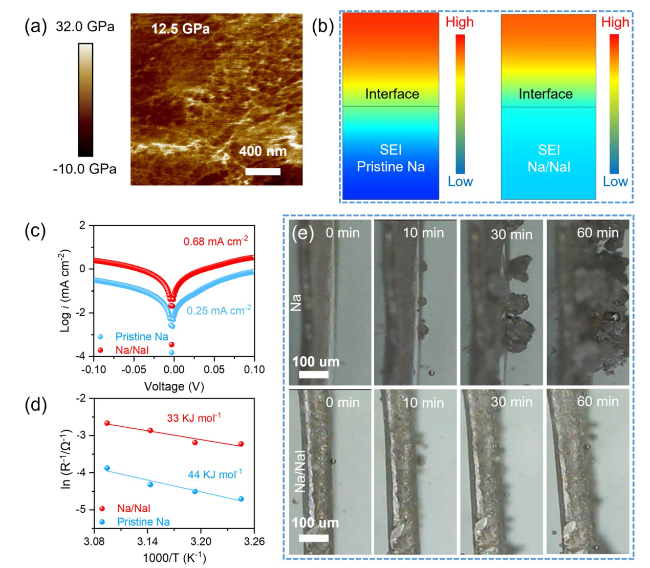
To further probe the role of the NaI protective layer in mitigating Na dendrites, AFM measurement was conducted to assess its strength. As displayed in
Fig. 4a, the Young’s modulus of the Na/NaI electrode can up to 12.5 GPa, which is much higher than that reported in previous studies for pristine and artificial Na electrodes (Table S1) [
40], indicating that the introduction of the NaI protective layer can enhance the mechanical strength of the electrode and further suppress the propagation of dendrites. In addition, the deposition behavior of Na on different electrode surfaces can be accurately simulated using finite element simulation in CMSOL multiphysics (
Fig. 4b). It is evident that, for the SEI of pristine Na electrode, the Na
+ concentration varies significantly at different depths, leading to an exacerbation of heterogeneous deposition. In contrast, the Na
+ concentration in the Na/NaI SEI is relatively low and gradually decreases without obvious fluctuations, which illustrates that the addition of a NaI layer facilitates the transfer of Na
+. Moreover, Tafel plot analysis was employed to verify the promotion of charge-transfer kinetics with the NaI layer. As showed in
Fig. 4c, the exchange current density (
j0) of pristine Na is 0.25 mA cm
-2, which is lower than that of Na/NaI (0.68 mA cm
-2). The charge transfer impedance of the Na/NaI symmetric cell at different temperatures displays smaller values (Figure S15). The fitting results, based on ln (1/
Rct) versus 1000/T, reveal that the activation energy (
Ea) of pristine Na and Na/NaI symmetric cells after 3 cycles is 44 and 33 KJ mol
-1, respectively (
Fig. 4d). The above results clearly reveal that the beneficial effect of introducing a NaI protective layer on the transport of Na ions.
Figure 4e provides a clearly in-situ optical observation of sodium deposition behaviors of pristine Na and Na/NaI electrodes. Initially, both of them appear flat. However, as the plating time increases to 10 min, noticeable differences start to emerge. Tiny particles begin to form on the Na surface and gradually grow over time. If left unchecked, these particles can lead to the formation of Na dendrites, which can negatively impact the lifespan of batteries in practical applications. In contrast, with the protection by the NaI layer, the surface remains flat without severe dendrites even after 60 minutes of plating (Figure S16).