1 Introduction
2 Methodology
2.1 Process description and simulation
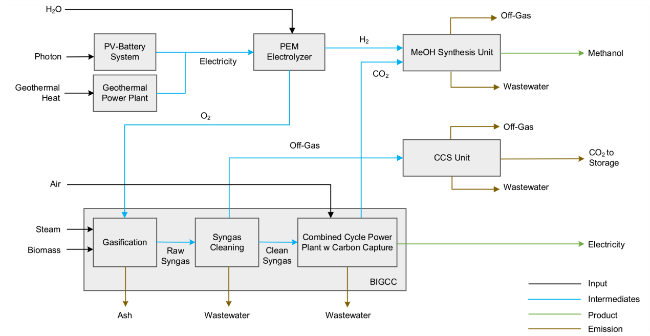
Fig. 1 Schematic diagram of the BECCS-PS for the co-production of green electricity and methanol |
2.1.1 The BIGCC unit
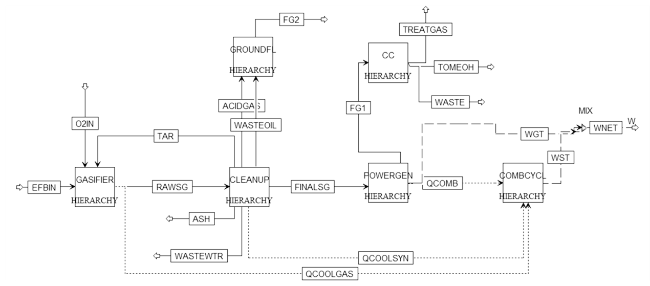
Fig. 2 Process flowsheet of the BIGCC |
Table 1 Proximate and ultimate analysis results of OPEFB [44] |
| Component | Value |
|---|---|
| Proximate Analysis (%wt) | |
| Moisture content | 5.18 |
| Volatile matter | 82.58 |
| Fixed carbon | 8.79 |
| Ash | 3.45 |
| Ultimate Analysis (%wt) | |
| C | 46.62 |
| H | 6.45 |
| N | 1.21 |
| S | 0.035 |
| O | 45.66 |
| Caloric Value (MJ/kg) | 17.02 |
2.1.2 Hydrogen production
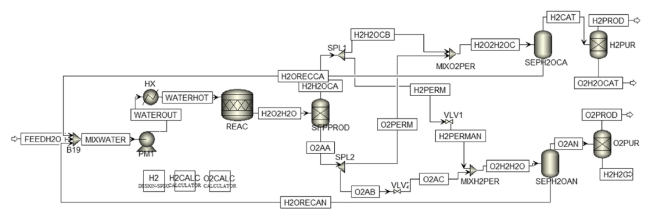
Fig. 3 Process flowsheet of the PEM electrolyser |
2.1.2.1 Solar photovoltaics
Table 2 PV-battery system specifications |
| Parameter | Value | Reference |
|---|---|---|
| PV efficiency (%) | 19.38 | [59] |
| PV dimension (mm) | 2008 × 1002 × 40 | [59] |
| PV capacity factor (%) | 12.7 | [60] |
| Global horizontal irradiation (kWh/m2/day) | 4.96 | [61] |
| Battery voltage | 380-505 | [62] |
| Round-trip efficiency (%) | 90 | [62] |
| Depth of discharge (%) | 100 | [62] |
| Battery dimension (mm) | 7168 × 1659 × 2522 | [62] |
2.1.2.2 Geothermal power plant
2.1.3 Methanol synthesis
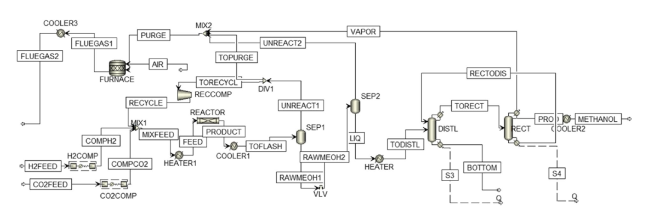
Fig. 4 Process flowsheet of methanol synthesis |
Table 4 Design parameters of the two-step methanol distillation process |
| Parameter | Distillation I | Distillation II |
|---|---|---|
| Number of stages | 30 | 30 |
| Feed stage | 15 | 15 |
| Recycle stage | 1 | - |
| Condenser type | Partial-vapor | Partial-vapor-liquid |
| Reboiler type | Kettle | Kettle |
| Reflux ratio (mole) | 1.1 | 6.8 |
| Boil-up ratio (mole) | 2 | 5 |
| Pressure (bar) | 1 | 1 |
| Distillate vapor fraction (mass) | - | 0.01 |
2.1.4 Carbon capture and storage
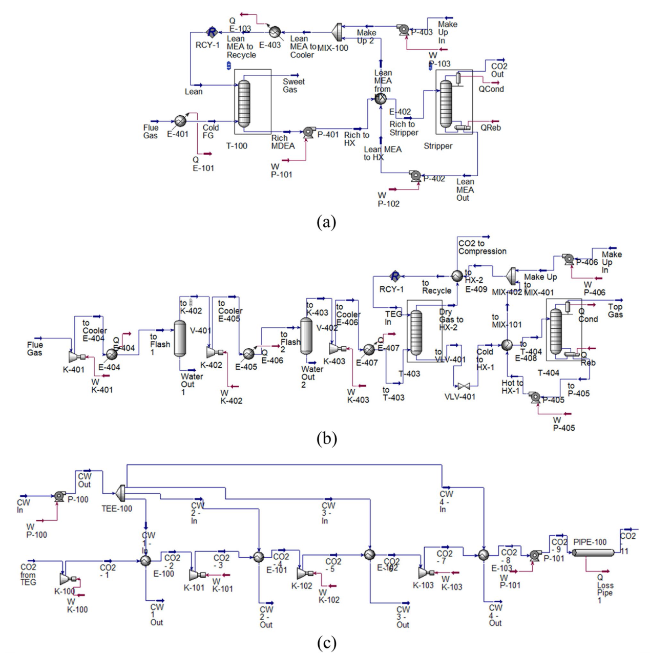
Fig. 5 Flowsheet of the CCS unit: a absorption, b dehydration, and c compression and transport |
Table 5 The input operating parameters for CO2 capture |
| Operating parameters | Value |
|---|---|
| Absorber | |
| Column diameter (m) | 2.5 |
| Height of packing (m) | 18 |
| Structured packing | Mellapak 250Y |
| Operating pressure (atm) | 1 |
| Number of stages | 15 |
| Stripper | |
| Column diameter (m) | 4.3 |
| Height of packing (m) | 20 |
| Structured packing | Mellapak 250Y |
| Operating pressure (atm) | 2 |
| Number of stages | 10 |
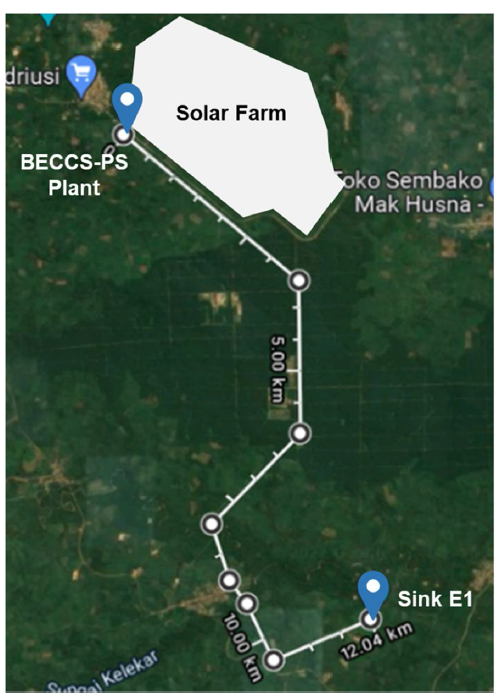
Fig. 6 BECCS-PS plant area |
2.2 System evaluation
2.2.1 Technical assessment
2.2.2 Life cycle assessment
Table 6 LCA inventory data |
| Activity | Value | Reference |
|---|---|---|
| OPEFB production | 0.168 kg CO2-eq/kg OPEFB | [79] |
| Biomass transportation | 29 g CO2-eq/km/ton | [80] |
| Deionized water production | 1.43 kg CO2-eq/m3 water | [81] |
| Mono-cSi PV manufacturing | 45 g CO2-eq/kWh | [82] |
| Li-ion battery manufacturing | 56.3 g CO2-eq/kWh | [83] |
| Geothermal power plant emission | 3.88 g CO2-eq/kWh | [84] |
| BIGCC electricity production | Simulation result | |
| Hydrogen production | Simulation result | |
| Methanol production | Simulation result | |
| CCS unit | Simulation result | |
2.2.3 Economic assessment
3 Results and discussion
3.1 Technical analysis
3.1.1 The BIGCC and CCS unit
Table 8 Technical performance of the BIGCC unit |
| Parameter | Unit | Value |
|---|---|---|
| Ash content in syngas | mg/m3 | 0 |
| H2S content in syngas | ppmv | 18.15 |
| Tar content in syngas | mg/m3 | 0 |
| Carbon conversion efficiency (CCE) | % | 99% |
| Cold gas efficiency (CGE) | % | 60.40% |
| H2/CO ratio of syngas | - | 0.59 |
| LHV of syngas | Btu/scf | 277.13 |
| Gas turbine efficiency | % | 35.63% |
| Combined cycle total efficiency | % | 49.14% |
| Overall efficiency | % | 29.68% |
Table 9 The final CO2 specification for pipeline |
| Component | Unit | Value |
|---|---|---|
| CO2 | %v/v | 99.7 |
| SO2 | ppmv | 1661.10 |
| Nitrogen | %v/v | 0.006 |
| Oxygen | ppmv | 1.22 |
| H2O | ppmv | 37.63 |
3.1.2 Hydrogen production and methanol synthesis
Table 10 Technical performances of the hydrogen production and methanol synthesis processes |
| Parameter | Unit | Value |
|---|---|---|
| Hydrogen production | ||
| Energy usage | kWh/kg H2 | 53.75 |
| H2O usage | kg/kg H2 | 9.39 |
| Energy efficiency | % | 62 |
| PV capacity | MWp | 1,894 |
| Battery capacity | MWh/day | 3,496 |
| PV land requirement | m2/kWp | 6.33 |
| Geothermal heat requirement | MW | 2,072.46 |
| Methanol production | ||
| Electricity usage | kWh/kg MeOH | 0.218 |
| CO2 usage | kg/kg MeOH | 1.41 |
| H2 usage | kg/kg MeOH | 0.193 |
| Overall CO2 conversion | % | 97.64 |
| Overall H2 conversion | % | 100 |
| Energy efficiency | % | 82.93 |
3.1.3 Overall technical performance
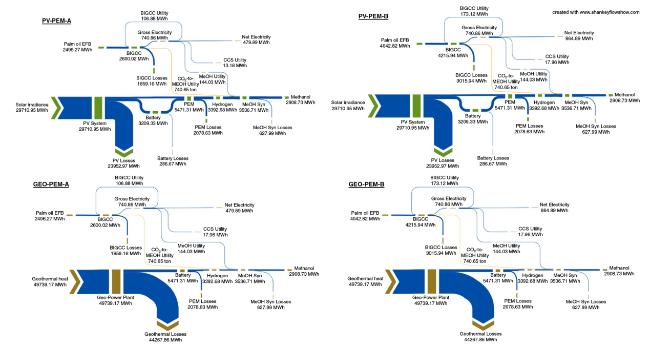
Fig. 7 Sankey diagram of the energy flow for all scenarios |
3.2 Life cycle analysis
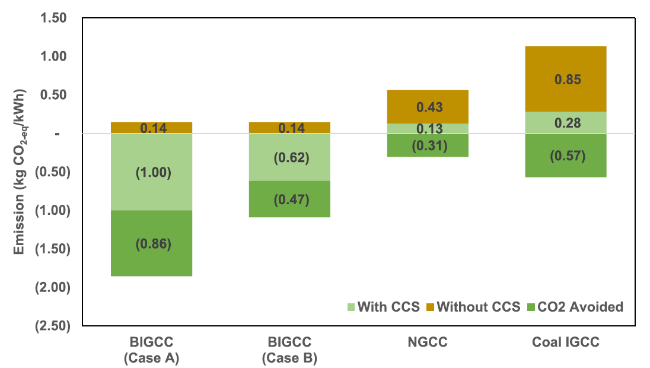
Fig. 8 The CO2-eq emissions from different power plants |
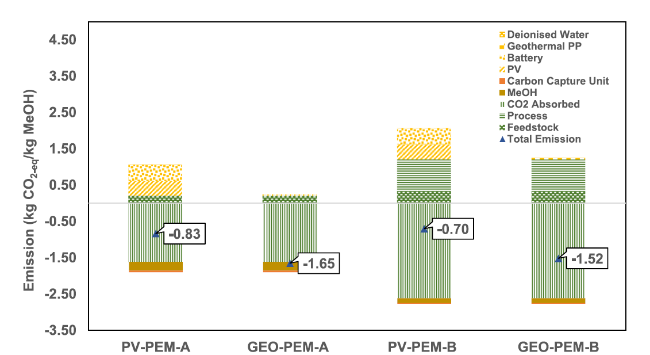
Fig. 9 The CO2-eq emissions for methanol synthesis in all scenarios |
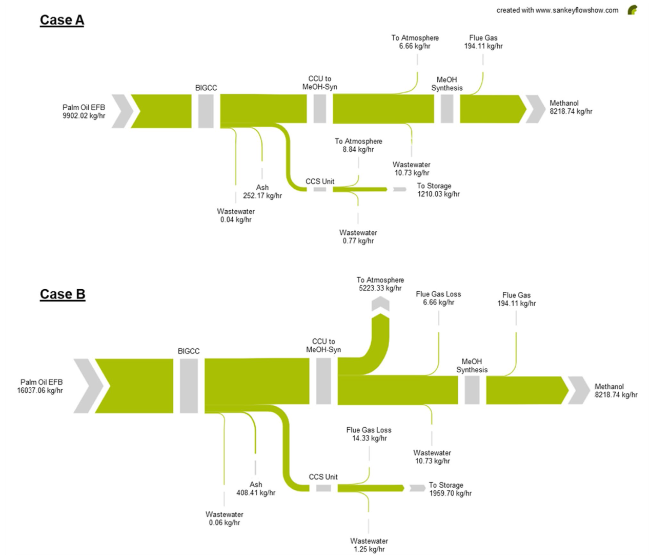
Fig. 10 Sankey diagrams of the carbon flow through the polygeneration system |
3.3 Economic analysis
3.3.1 The LCOE and CAC
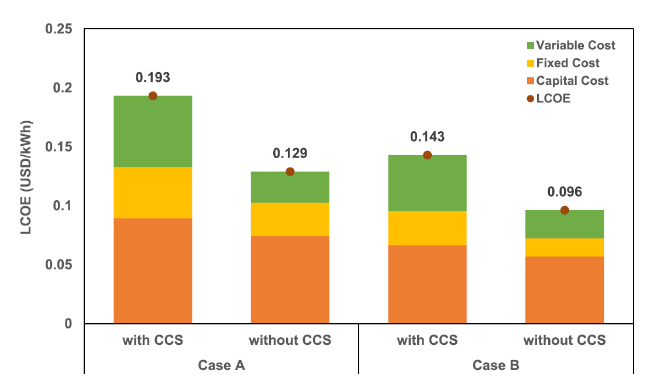
Fig. 11 Levelized cost of electricity for all cases in the BIGCC unit |
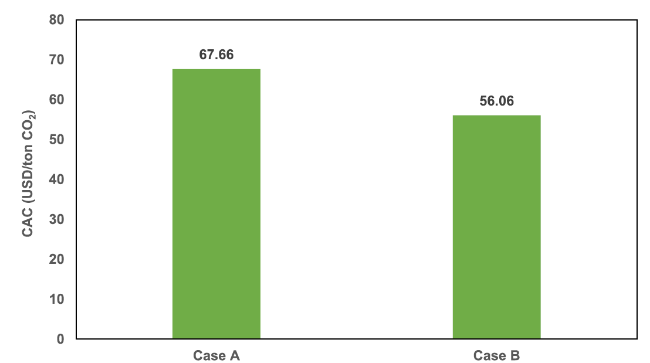
Fig. 12 The CAC for both cases in the BIGCC unit |
3.3.2 LCOM
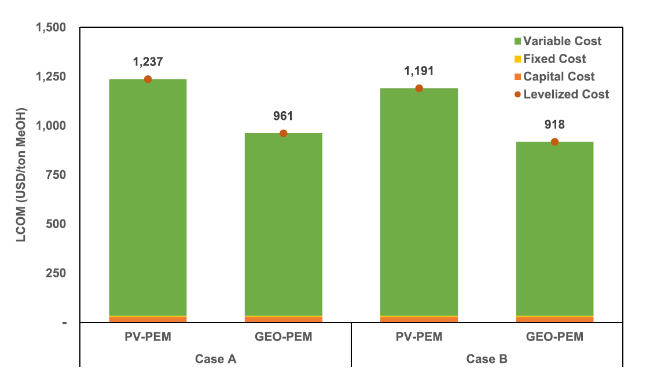
Fig. 13 Levelized cost of methanol in all scenarios |
3.3.3 Projection of levelized cost
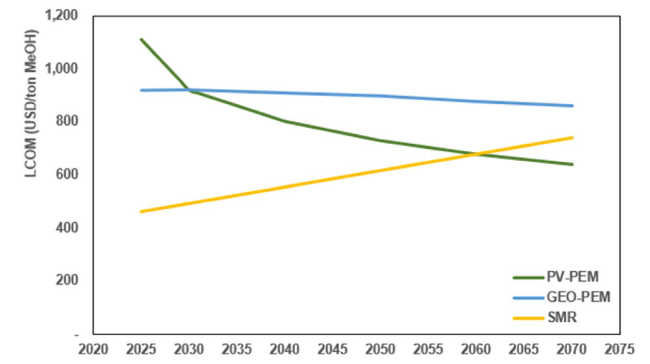
Fig. 14 Projected levelized cost of methanol |