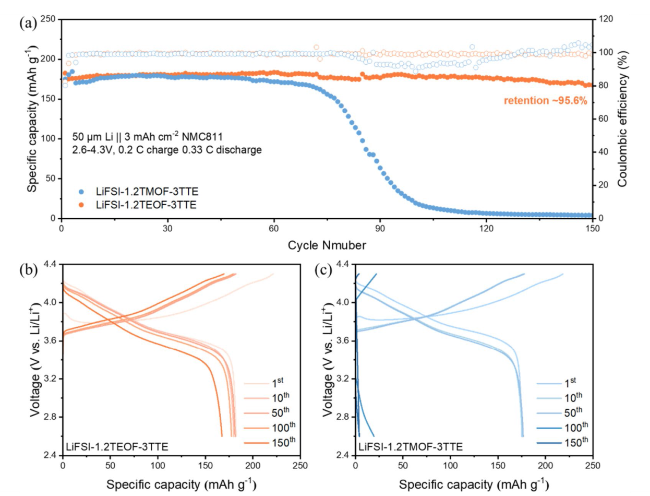
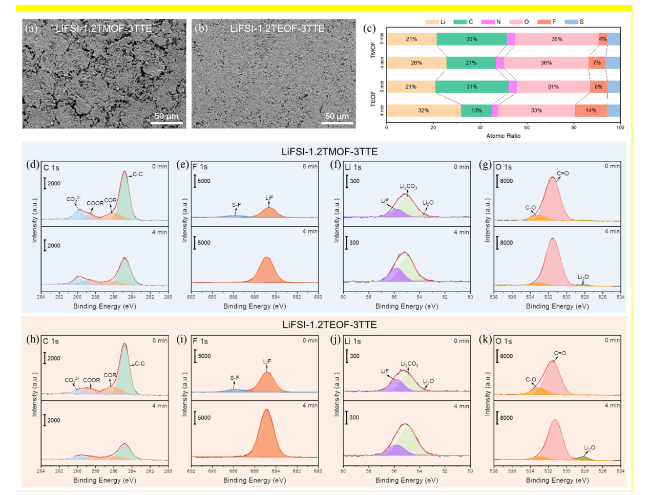
SEM and X-ray photoelectron spectroscopy (XPS) characterizations were conducted to investigate the morphology evolution and interfacial chemistry of the Li metal anode cycled in Li||NMC811 cells after 50 cycles. The cycled Li metal in the LiFSI-1.2TMOF-3TTE electrolyte presents extensive cracks (
Fig. 6a, Fig. S12), which results in the continuous penetration of electrolyte to occur in continuous side reactions with fresh Li metal. By contrast, the Li metal anode cycled in LiFSI-1.2TEOF-3TTE exhibits a uniform and smooth surface without any cracks (
Fig. 6b). The distinct morphologies of the cycled Li metal origin from the different properties of SEI, which were regulated by the different solvation structures in two electrolytes [
51,
52,
53]. XPS characterizations were further applied to investigate the compositions of SEI. As shown in
Fig. 6c, although the similar atomic ratio of C element can be observed in the outer layer of SEI formed in two electrolytes (30% for LiFSI-1.2TMOF-3TTE, 31% for LiFSI-1.2TEOF-3TTE), after argon ion cluster sputtering for 4 min, more content of C can be detected in the inner layer of SEI formed in LiFSI-1.2TMOF-3TTE (21%) than LiFSI-1.2TEOF-3TTE (13%). Furthermore, apparent signals of C-C (~ 284.8 eV), C-O (~ 286 eV), C = O (~ 288.5 eV), and CO
32− (~ 289.8 eV) can be observed in the SEI formed in two electrolytes (
Fig. 6d h). The increased content of C-containing organic species in the SEI formed in LiFSI-1.2TMOF-3TTE indicates the major contribution of solvent molecules on interfacial reactions, which matches well with the results of solvation structures. More importantly, the Li metal anode cycled in the LiFSI-1.2TEOF-3TTE (7% and 14%) electrolyte shows an increase in the atomic ratio of F element in the outer and inner layer of SEI than that cycled in LiFSI-1.2TMOF-3TTE (4% and 8%). In the F 1s spectra, higher intensity of LiF signal (~ 684.7 eV) and S-F species (~ 687.8 eV) can be detected in the SEI formed in LiFSI-1.2TEOF-3TTE (
Fig. 6e and
i), which matches with the trend of LiF signal in the Li 1s spectra (
Fig. 6f and
j). After 4 min sputtering, the Li
2O (~ 528.2 eV) peak intensity in the O 1s spectra in LiFSI-1.2TEFO-3TTE is slightly higher than that in LiFSI-1.2TMOF-3TTE (
Fig. 6g and
k). The enrichments of LiF and Li
2O are critical to improving the compatibility of LiFSI-1.2TEOF-3TTE towards the Li metal anode. In addition, the interfacial chemistry of cathode cycled in Li||NMC811 cells after 50 cycles were also characterized (Figs. S13, S14). The cathode cycled in LiFSI-1.2TEOF-3TTE shows an increased peak intensity of the LiF signal, which improves the cycling stability of the high-voltage NMC cathode. Combined with the steric hindrance effect, we Found that the anion-dominant solvation structures in the LiFSI-1.2TEOF-3TTE electrolyte contribute to the formation of inorganic-rich SEI and CEI, which are beneficial in improving the cycling stability of high-voltage Li||NMC811 cells.