1 Introduction
2 Application of quantum dots in lithium- sulfur batteries
2.1 Positive electrode for lithium-sulfur batteries
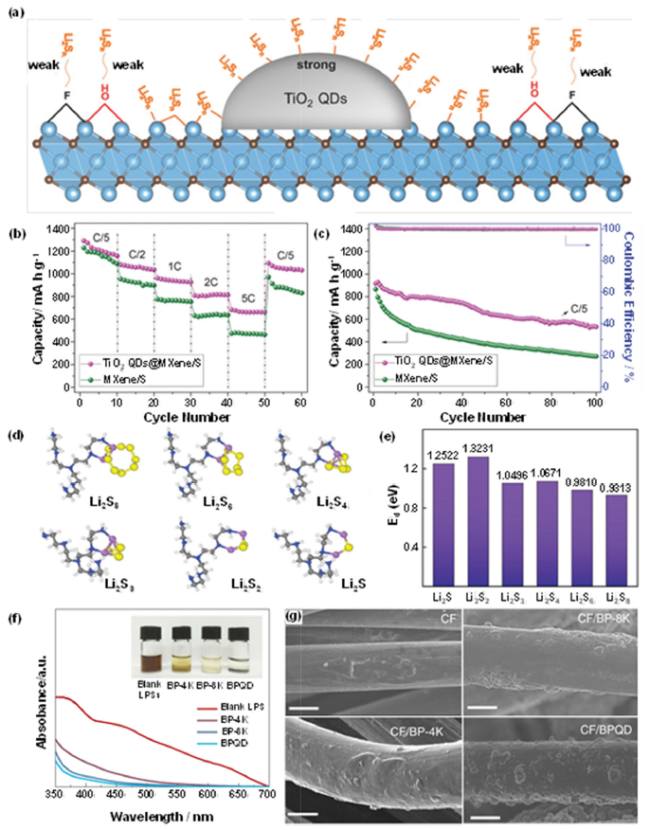
Fig. 1 A Schematic diagram of the principle of TiO2 quantum dots growing on MXene nanosheets to inhibit the polysulfide shuttle effect; B rate capabilities of TiO2 QDs@MXene/S and MXene/S cathodes (1C = 1675 mA g−1); C Comparisons of cycle behaviors at C/5 (sulfur loading = 5.5 mg cm−2) of TiO2 QDs@MXene/S and MXene/S cathodes;(Reprinted from Ref. [82]. Copyright 2018 Wiley-VCH.) D The calculated configurations of the LiPSs species (Li2S, Li2S2, Li2S3, Li2S4, Li2S6, and Li2S8) on a reducible molecular structure of the PEI surface functional groups; E The calculated binding energy between the PEI surface-active group and the LiPSs species; (Reprinted from Ref. [83]. Copyright 2019 Wiley-VCH.) F SEM images of Li2S precipitation on different substrates; G UV-vis spectra of LiPS with variation in color upon adsorption by different-sized BP flakes. (CF is carbon fiber, BP-4K is BP that was centrifuged at 4000 rpm, BP-8K was centrifuged at 8000 rpm, BPQD is black phosphorus quantum dot) (Reprinted from Ref. [81]. Copyright 2018 SPRINGER NATURE.) |
2.2 Quantum dot modified separator
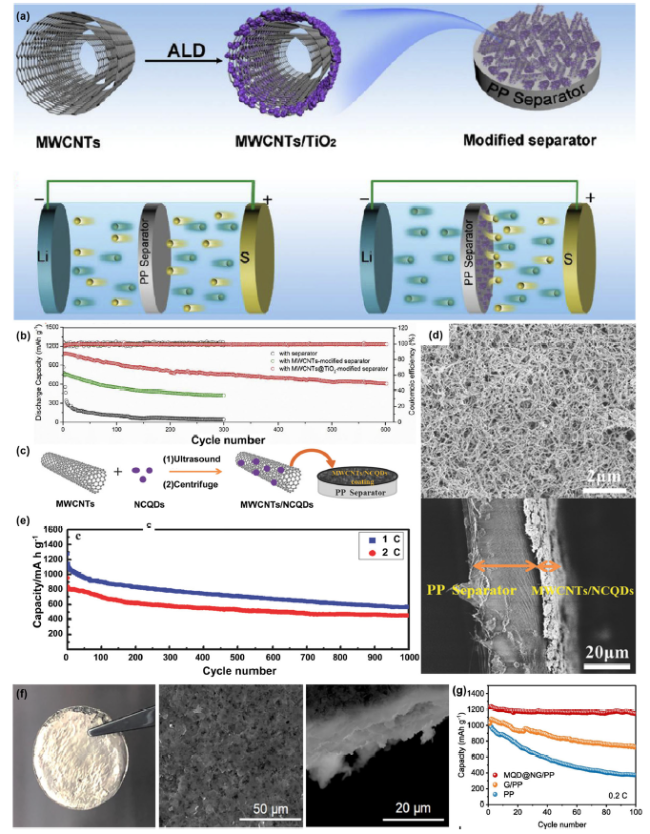
Fig. 2 A schematic diagram of the preparation process of MWCNTs@TiO2 quantum dots and Schematic diagram of li-S cell with commercial PP diaphragm and MWCNTs@TiO2 quantum dot coated diaphragm; B Long life cycle tests of Celgard, MWCNTs/Celgard and MWCNTs@TiO2 quantum dots /Celgard. (Reprinted from Ref. [75]. Copyright 2018 Elsevier Ltd.) C Schematic diagram of BPQD/TNS composite and HRTEM image of BPQD anchored on TNS surface; D Photographs of one side of the MWCNTs/NCQDs-coated separator and cross-section of MWCNTs/NCQDs-coated separator; E Long-term cycling performance at 1 and 2 C of the Li-S batteries with MWCNTs/NCQDs-coated separator.(Reprinted from Ref. [88]. Copyright 2018 Wiley-VCH.) F Optical photograph of the lithium plate after plating/stripping with MQD@NG/PP separators for 200 cycles. Corresponding SEM image of the lithium plate with MQD@NG/PP separator. cross-section of the lithium plate after cycles. G Cycle performance of PP, G@PP, MQD@NG/PP batteries at 0.2c. (Reprinted from Ref. [89]. Copyright 2020 Elsevier Ltd.) |
3 Progress of quantum dots in supercapacitors
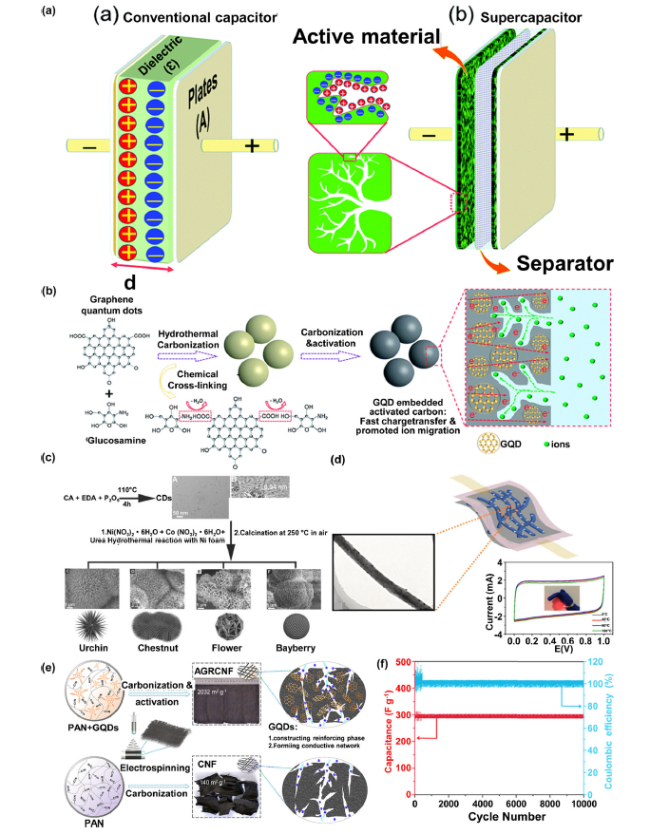
Fig. 3 A Schematic representation of a conventional capacitor and a supercapacitor; (Reprinted from Ref. [96]. Copyright 2019 Royal Society of Chemistry.) B Illustration of the N-GQDs//MoS2-QDs asymmetric MSCs; (Reprinted from Ref. [97]. Copyright 2019 Royal Society of Chemistry.) C The CDs are synthesized hydrothermally with uniform sizes and employed to prepare C-F) the CDs/NiCo2O4 composites with different morphologies; (Reprinted from Ref. [98]. Copyright 2016 Wiley-VCH.) D Schematic diagram of graphene quantum dots modified carbon nanofiber felt flexible supercapacitor; (Reprinted from Ref. [99]. Copyright 2021 Royal Society of Chemistry.) E Schematic diagram of GQD-reinforced electrospun carbon nanofiber fabrics(AGRCNF) prepared by electrostatic spinning, carbonization and chemical activation; F Cycle performance of the AGRCNF-3//AGRCNF-3 supercapacitor at 50 A g-1. (Reprinted from Ref. [100]. Copyright 2020 ACS Publications) |
Table 1 Application of quantum dots in supercapacitors |
| Electrode materials | Specific capacitance | Cycling life | Electrolyte | Energy density | Ref |
|---|---|---|---|---|---|
| NiCo2O4/GQDs | 481.4Fg-1 at 0.35 Ag-1 | 65.88% (300) | 1M KOH | - | [103] |
| GQDs | 332 Fg−1 at 0.5 Ag−1 | - | 1M H2SO4 | 6.4 Wh kg-1 | [99] |
| CQDs/Ni3S2 | 1130 Fg−1 at 2 Ag−1 | 80% (3000) | 1M KOH | 18.8 Wh kg-1 | [104] |
| NCH/NCQDs | 727C g-1 at 1 A g-1 | 87.5% (8000) | 3M KOH | 49.1 Wh kg-1 | [105] |
| NiCo2O4/CQDs | 856 F g-1 at 1A g-1 | 98.75% (10000) | 2M KOH | 13.1 Wh kg-1 | [106] |
| V2O5/GQDs | 572 F g-1 at 1A g-1 | 92% (10000) | 0.1 M H2SO4 | 31.25 Wh kg-1 | [107] |
| ONCDs/Porous Hydrogels | 400 F g-1 at 1A g-1 | 100% (10000) | 1M H2SO4 | 13.5 Wh kg-1 | [108] |
| MnO2/CQDs | 340 F g-1 at 1A g-1 | 80.1% (10000) | 1M Na2SO4 | 33.6 Wh kg-1 | [109] |
| GQDs/Ultra microporous | 3 F cm-2 at 0.5 A g-1 | 100% (50000) | 6M KOH | 9.38 Wh kg-1 | [110] |
| N-O GQDs | 325 F g-1 at 0.8 A g-1 | 82.6% (5000) | 1M H2SO4 | 8.0 μWh cm-2 | [111] |
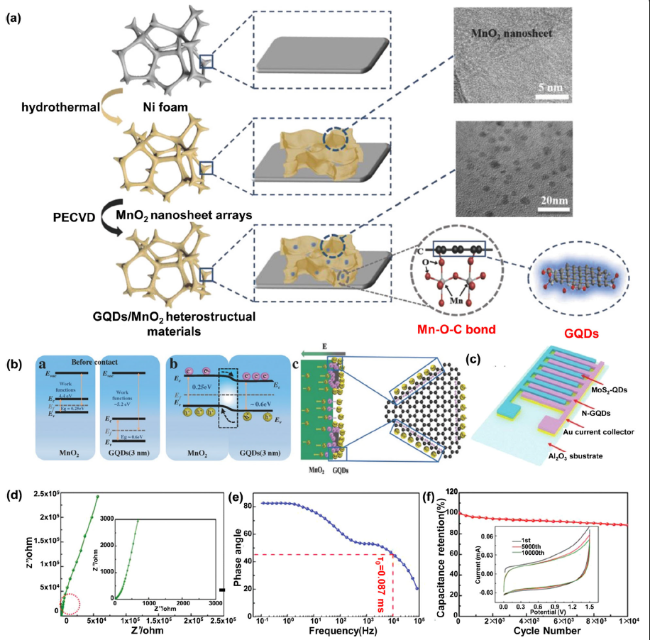
Fig. 4 A Fabrication process of GQD/MnO2 heterostructural materials; B Energy diagram of MnO2 and GQDs before contact, energy diagram of the interface between MnO2 and GQDs after the formation of a heterojunction and the schematic diagram of free electrons accumulating near the GQDs surface; (Reprinted from Ref. [94]. Copyright 2018 Wiley-VCH.) C General preparation route of the GQD embedded activated carbons; D Nyquist plot of the MSCs showing the imaginary part versus the real part; E Plot of impedance phase angle versus frequency; F Capacitance retention of the MSCs as a function of cycle number measured at the scan rate of 1 V s−1 with 10 000 cycles. (Reprinted from Ref. [115]. Copyright 2020 Wiley-VCH.) |
4 Photocatalytic Hydrogen Evolution Applications of Quantum Dots
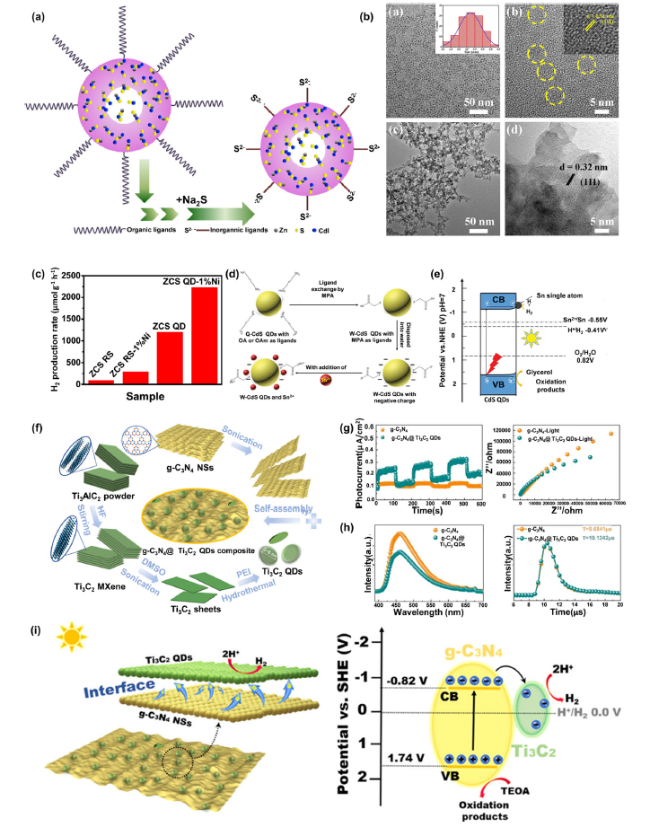
Fig. 5 A The schematic illustration of the ligand-exchange process for ZCS QD; B The TEM images of ZCS quantum dots; C Photocatalytic H2-production rates of ZCS RS, RS-1% Ni, QD, and QD-1% Ni; (Reprinted from Ref. [138]. Copyright 2021 Elsevier Ltd.) D A schematic illustration of ligand exchange and adsorption of Sn2+ on the surface of CdS QDs; E H2 production and separation of CdS QDs with Sn2+ or Sn atom under visible light irradiation; (Reprinted from Ref. [139]. Copyright 2020 Wiley-VCH.) F Schematic Illustration of the Preparation of g-C3N4@Ti3C2 QD Composites; G Steady photoluminescence (PL) and time-resolved fluorescence decay spectra of g-C3N4 and g-C3N4@Ti3C2 QDs-100 mL composites, λex = 325 nm; H Photocurrent responses and EIS of g-C3N4 NSs and g-C3N4@Ti3C2 QDs-100 mL composites; I Schematic Photocatalytic Mechanism of g-C3N4@Ti3C2 QD Composites. (Reprinted from Ref. [140]. Copyright 2019 ACS Publications) |
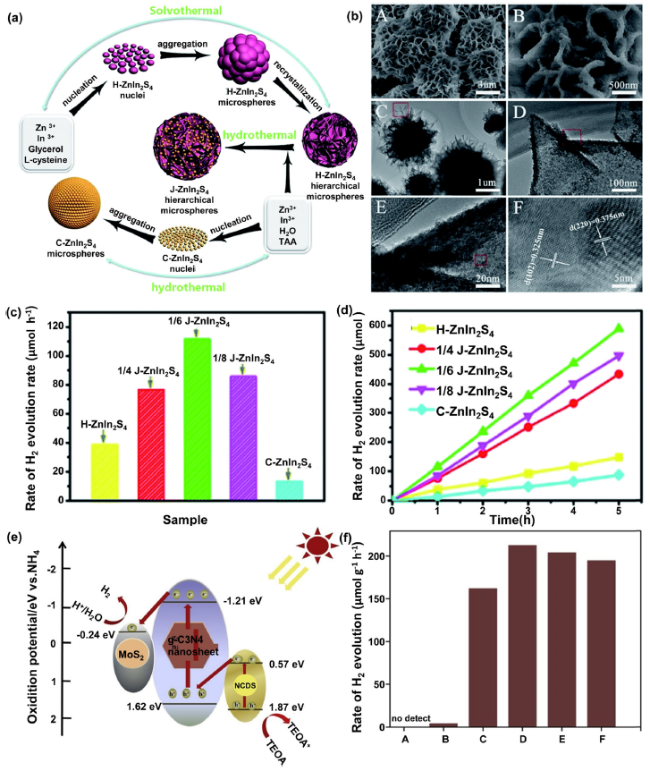
Fig. 6 A Schematic illustration of the formation process of the cubic quantum dot/hexagonal microsphere ZnIn2S4 heterophase junctions; B SEM images, TEM image, and the HRTEM image of the cubic quantum dot/hexagonal microsphere ZnIn2S4 heterophase junction; C, D Comparison of the photocatalytic H2 evolution rate of H-ZnIn2S4, C-ZnIn2S4 and J-ZnIn2S4 samples with different cubic/hexagonal mole ratios (1:4, 1:6, and1:8) and different reaction times; (Reprinted from Ref. [152]. Copyright 2017 Royal Society of Chemistry.) E Proposed photocatalytic mechanism for hydrogen evolution over g-C3N4/NCDS/MoS2 under visible light irradiation; F Photocatalytic reaction of g-C3N4/NCDS/MoS2 composite photocatalyst under visible Light (λ≥420nm) for 3h. (A: bulk g-C3N4. B: g-C3N4/MoS2-3%. C: g-C3N4/NCDS/MoS2-2%. D: g-C3N4/NCDS/MoS2-3%. E: g-C3N4/NCDS/MoS2-4%. F: g-C3N4/NCDS/MoS2-5%). (Reprinted from Ref. [153]. Copyright 2019 Elsevier Ltd.) |