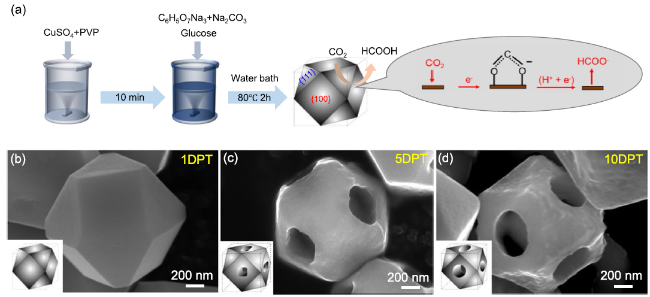
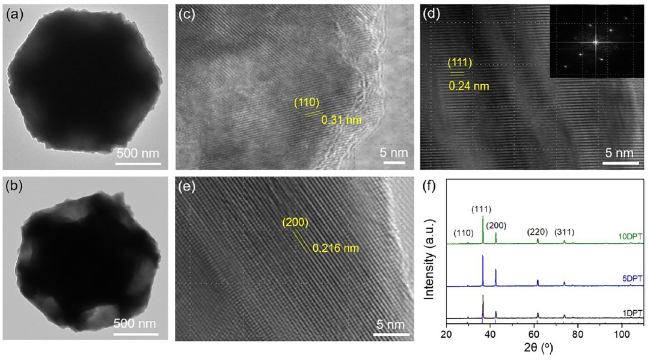
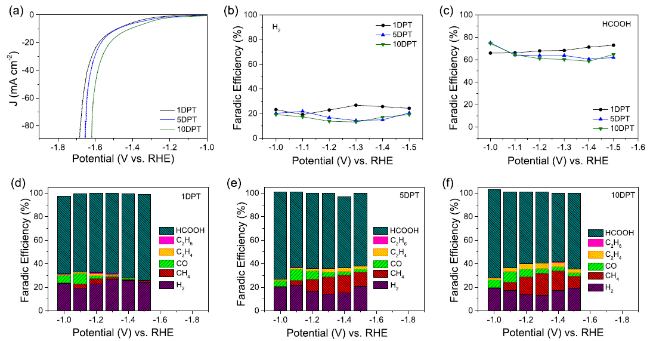
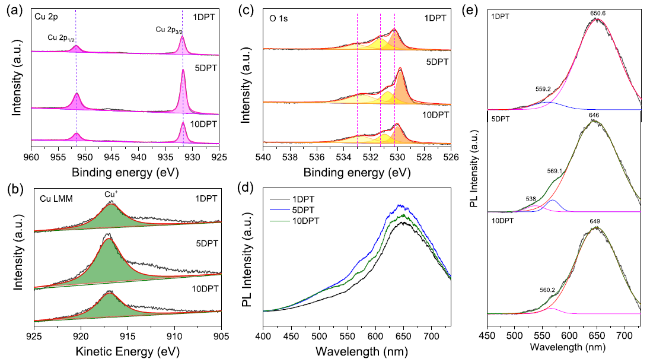
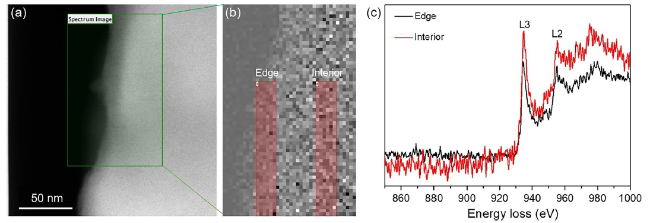
Formic acid (HCOOH) is an essential CO
2RR product, which has been commercialized due to its feasibility and large applicability spectra [
11]. It is proved to be a satisfying fuel applied in fuel cells [
12] and can serve as an energy-storage medium with high stability [
13]. Different catalysts were utilized to generate HCOOH, including Sn, Pb, Cu
2O and some metal-complex catalysts [
11]. It has been reported that during the CO
2RR process catalyzed by Cu
2O, many oxygen atoms are precipitated in the catalyst in the reduction process [
5]. Meanwhile, some of the remaining atoms can generate a portion of Cu(Cu
+) with positive valence to remain on the surface of the catalyst. During the surface reduction and reconstruction of the catalyst, Cu
+ and Cu in adjacent positions are favorable for coupling C-C and C-H to generate multi-carbon products with added value. The Cu-based compounds with metastable structure, crystal interface, high density and high specific surface area can expose more catalytic active sites, have higher catalytic activity, stabilize the intermediates in the reduction process, and reduce the overpotential in the CO
2RR process, thus reducing energy consumption [
10,
14,
15,
16,
17].