1 Introduction
Table 1 Representative summary of photocatalytic CO2 reduction systems |
| Catalyst | Light source | Photosensitizer | Reaction media | Major products | Selectivity | Activity | Ref. |
|---|---|---|---|---|---|---|---|
| mCD/CN | Visible light | - | H2O | CH3OH | 99.6% | 13.9 μmol g−1 h−1 | Tang et al. 2020 [20] |
| MXene/Bi2WO6 | UV-Vis light | - | H2O, NaHCO3, H2SO4 | CH4 CH3OH | - | 1.78 μmol g−1 h−1 0.44 μmol g−1 h−1 | Yu et al. 2020 [21] |
| Cu2O@Cu3(BTC)2 | Visible light | - | H2O vapor | CH4 | 100% | 0.73 mmol (8 h) | Tang et al. 2021 [22] |
| Ni-SA-x/ZrO2 | UV-vis light | - | H2O | CO | 92.5% | 11.8 μmol g−1 h−1 | Zhang et al. 2020 [23] |
| CuPc/g-C3N4 | Visible light | - | H2O | CO CH4 | - | 1.5 μmol g−1 h−1 | Jing et al. 2020 [24] |
| Mn, C-ZnO CTSHS | UV-vis light | - | H2O | CO | 0.83 μmol g−1 | Yu et al. 2021 [25] | |
| CD/FAT | Visible light | - | H2O | CH3OH | 100% | 24.2 μmol g−1 h−1 | Tang et al. 2021 [26] |
| NiMn-COS-UCN | UV-vis light | - | H2O | CO CH4 | - | 13.85 μmol g−1 h−1 2.22 μmol g−1 h−1 | Jing et al. 2021 [27] |
| 1Au-6T/0.8M/PCN | Visible light | - | H2O | CH4 | 140 μmol g−1 h−1 | Jing et al. 2018 [28] | |
| BiVO4{010}-Au-Cu2O | Visible light | - | H2O | CO CH4 | - | 2.02 μmol g−1 h−1 3.14 μmol g−1 h−1 | Zhou et al. 2018 [18] |
| SrTiO3:La, Rh|Au|RuO2-BiVO4:Mo | Visible light | - | H2O, KHCO3 | HCOO- CO | 97% | 6.53 μmol g−1 h−1 | Demon et al. 2020 [29] |
| (001)TiO2-g-C3N4/BiVO4 | Visible light | - | H2O | CO CH4 | - | 5.18 μmol g−1 h−1 | Jing et al. 2021 [30] |
| Cu2O-Pt/SiC/IrOx | Visible light | - | H2O, FeCl3 | HCOOH | - | 896.7 μmol g−1 h−1 | Li et al. 2020 [31] |
| COF-318-TiO2 | Visible light | - | H2O | CO | - | 69.67 μmol g−1 h−1 | Lan et al. 2020 [32] |
| ZnPc/BVNS | Visible light | - | H2O | CO CH4 | - | 0.97 μmol g−1 h−1 | Jing et al. 2019 [33] |
| TiO2/C3N4/Ti3C2 Mxene | UV-vis light | - | H2O, H2SO4, NaHCO3, | CO CH4 | - | 4.39 μmol g−1 h−1 1.20 μmol g−1 h−1 | Yu et al. 202 [34] |
| [PMoV8MoVI4O35(OH)5Zn4]2 [Zn-TCPP][2H2O]·xGuest(NNU-13) | Visible light | - | H2O, TEOA | CH4 | 96.6% | 117 μmol g−1 h−1 | Lan et al. 2020 [35] |
| W18O49@Co | Visible light | Ru(bpy)32+ | H2O, MeCN, TEOA | CO | - | 21.18 mmol g-1 h-1 | Lou et al. 2021 [36] |
| IrQPY/CoPc | Visible light | Ir Ps | CH3CN, BIH, TEA | CO | 98% | - | Ouyang et al. 2021 [37] |
| 3DOM CdSQD/NC | Visible light | Co(bpy)32+ | H2O, MeCN, Benzylamine | CO | 89.6% | 5210 μmol g−1 h−1 | Wang et al. 2021 [38] |
| TiO2/CsPbBr3 | Uv-Vis light | Ru(bpy)32+ | H2O, MeCN, BIH | CO | - | 9.02 μmol g−1 h−1 | Yu et al. 2020 [39] |
| [Ni(tpy)2]2+ | Visible light | Ru(bpy)32+ | H2O, CH3CN, BIH | CO | 99% | - | Xiong et al. 2019 [40] |
| NiCoOP-NPs@MHCFs | Visible light | Ru(bpy)32+ | H2O, MeCN, TEOA | CO | - | 16.6 μmol h−1 | Lou et al. 2019 [41] |
| Au/CdS-HMCHPs | Visible light | Co(bpy)32+ | H2O, MeCN, TEOA | CO | 70.3% | 3758 μmol g−1 h−1 | Lou et al. 2019 [42] |
2 Basic principle and procedure of photocatalytic CO2 reduction
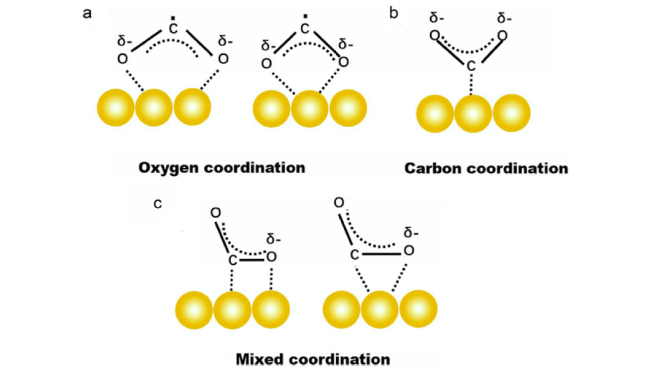
Fig. 1 The possible structures of adsorbed CO2δ•- species on catalysts [53] Copyright 2020, Elsevier |
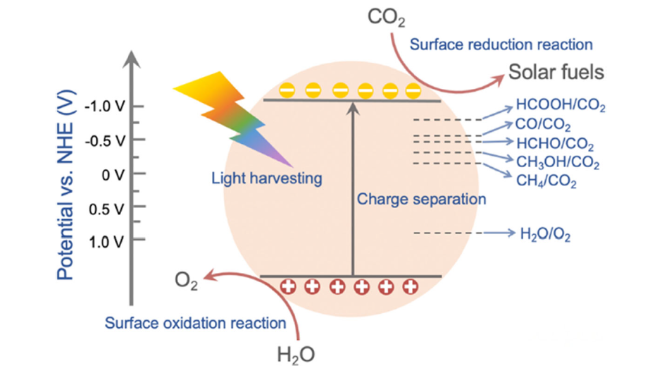
Fig. 2 Schematic illustration of photocatalytic CO2 reduction by water over a photocatalyst at pH = 7 |
3 Reaction systems and products detection for CO2 reduction
3.1 Reaction systems
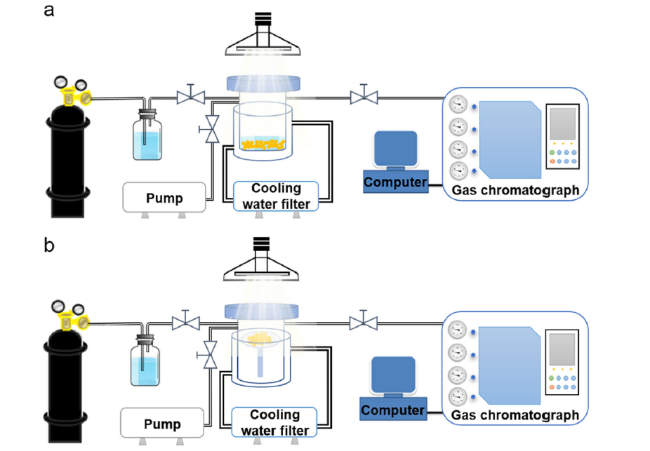
Fig. 3 Schematic diagram of the devices of the solid-liquid reaction system (a) and the gas-solid reaction system (b), respectively |
3.2 Products detection
3.2.1 Analysis of gaseous products
3.2.2 Analysis of liquid products
4 Strategies for improving photocatalytic performance of CO2 reduction by water
4.1 Co-catalysts engineering for CO2 adsorption and activation
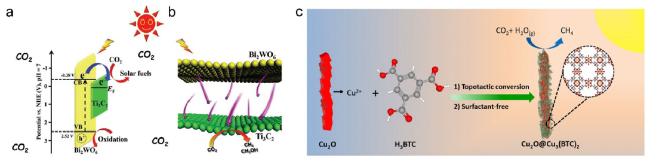
Fig. 4 (a) Energy level structure diagram of Bi2WO6 and Ti3C2 (b) Photo-induced electron transfer process at the interface of the hybrids [21] Copyright 2018, Wiley-VCH (c) Illustration of the synthetic route towards Cu2O@Cu3(BTC)2 core-shell nanowires through a surfactant-free topotactic conversion strategy for selective photocatalytic carbon dioxide reduction to methane [22] Copyright 2021, Wiley-VCH |
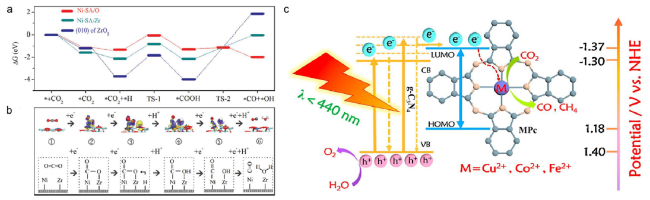
Fig. 5 (a) Energy profile for the elementary steps of CO2 reduction to CO over (010) facets of ZrO2, Ni-SA/Zr, and Ni-SA/O (b) Differential charge density diagrams and intermediates during CO2 reduction to CO over Ni-SA/O model [23] Copyright 2020, Wiley-VCH (c) Schematic of HLEEs transfer process in the fabricated MPc/CN heterojunction [24] Copyright 2020, Elsevier |
4.2 Accelerated water oxidation kinetics
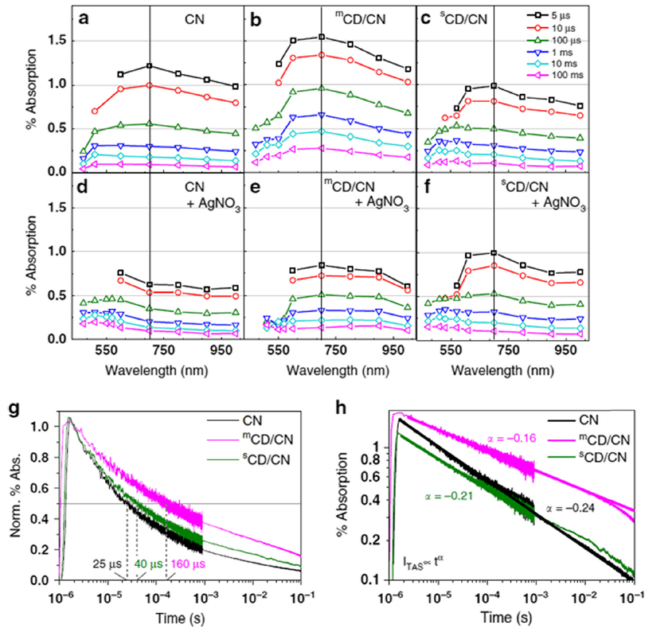
Fig. 6 Diffuse reflectance TAS spectra for samples with (a-c) and without (d-f) 10 mM AgNO3 (g) μs-TAS decay kinetics of CN, mCD/CN and sCD/CN in water monitored at 700 nm and excited by pulsed 355 nm excitation (460 μJ/cm2) and (h) fitted α parameters indicated in the same color as the associated trace [20] Copyright 2020, Springer Nature |
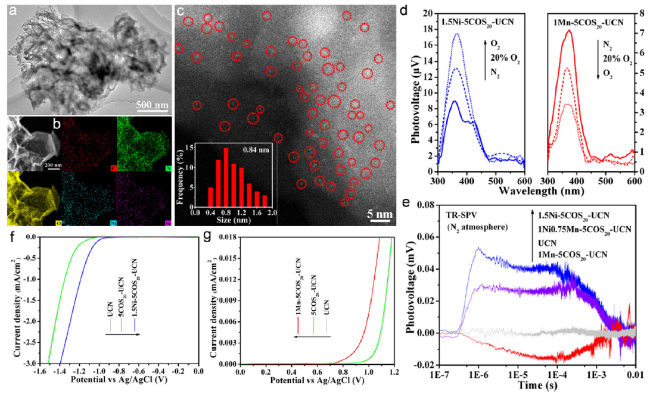
Fig. 7 (a) TEM image, (b) STEM image and the corresponding EDX mapping images of C, N, O, Ni, and Mn elements, and (c) HAADF-STEM image of 1Ni0.75Mn-COS20-UCN (d) SPS responses of 1.5Ni-COS20-UCN (left) and 1Mn-COS20-UCN (right) in different atmospheres (e) TS-SPV responses of UCN, 1Mn-COS20-UCN, 1.5Ni-COS20-UCN, and 1Ni0.75Mn-5COS20 UCN in the N2 atmosphere (f) EC reduction curves of UCN, 5COS20-UCN and 1.5Ni-COS20-UCN in the CO2-bubbled system (g) EC oxidation curves of UCN, 5COS20-UCN and 1Mn-COS20-UCN samples in the N2-bubbled system [27] Copyright 2021, Wiley-VCH |
4.3 Promoting charge separation by constructing Z-scheme heterojunctions
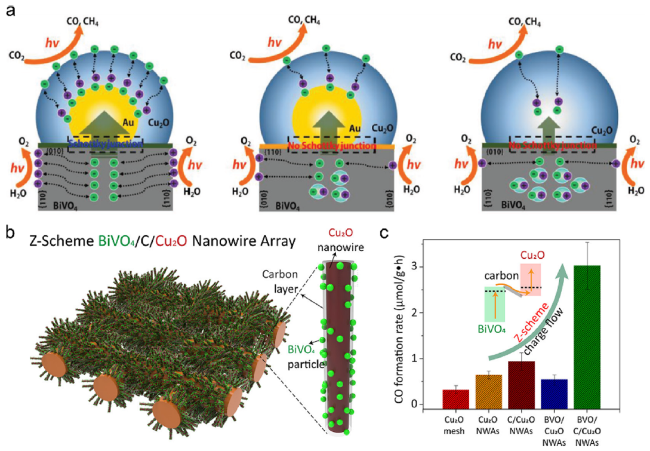
Fig. 8 (a) Scheme to describe the carrier migration behavior from BiVO4 to Cu2O with and without Au particles deposited on the different facets of BiVO4 [18]. Copyright 2018, Wiley-VCH (b) Schematic of the synthesized BVO/C/Cu2O nanowire (c) Photocatalytic performance for CO2 reduction of various catalysts [78]. Copyright 2018, American Chemical Society |
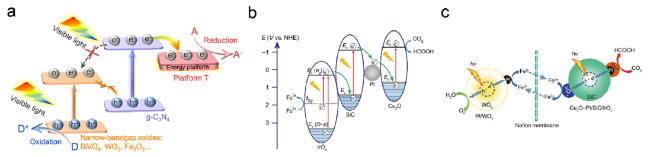
Fig. 9 (a) Schematic representation of the proposed cascade Z-Scheme mechanism of photogenerated charge transfer under visible light for efficient photocatalysis. T refers to (001)TiO2, which can feasibly be replaced by other wide bandgap semiconductors like SnO2 [30] Copyright 2021, Wiley-VCH (b) The electron transfer processes in Cu2O-Pt/SiC/IrOx under light illumination (c) The proposed mechanism of the separated system for the efficient CO2 reduction and O2 evolution [31] Copyright 2020, Springer Nature |
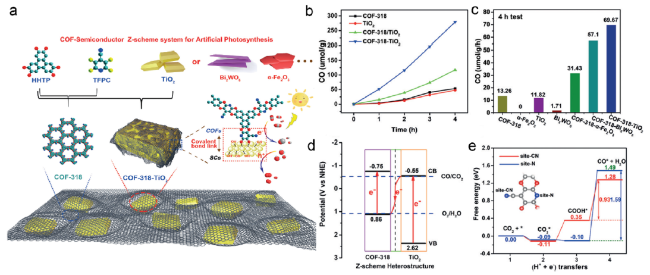
Fig. 10 (a) Schematic representation of the preparation of COF-318-SCs via the condensation of COF-318 and semiconductor (b) Photocatalytic CO2 to CO performances of COF-318-TiO2 for gas-solid CO2 reduction compared with COF-318, TiO2 and COF-318/TiO2 (c) Photocatalytic performances of various COF-318-SCs and bulk COF-318, TiO2, Bi2WO6 and α-Fe2O3 (d) Schematic illustration of the charge transfer process under light irradiation with Z-scheme model (e) CO2 RR process on COF-318-TiO2 based on DFT calculations [32] Copyright 2020, Wiley-VCH |
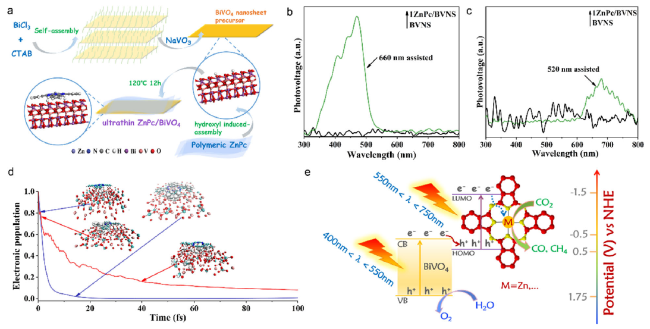
Fig. 11 (a) Schematic illustration of the preparation of ultrathin ZnPc/BVNS heterojunction. SPS responses of 1ZnPc/BVNS and BVNS in N2 atmosphere assisted with a (b) 660 nm and (c) 520 nm monochromatic beam (d) The time-dependent survival probability (TDSP) curves of the excited electrons transfer between partly hydroxylated BVNS and ZnPc during the injection process (Red: from ZnPc to BVNS; Blue: from BVNS to ZnPc) (e) Proposed cascade Z-scheme mechanism of photogenerated charge transfer on MPc-coupled BVNS. M represents a transition metal [33] Copyright 2019, Wiley-VCH |