An important trend in global agriculture is to utilize plant probiotics, which are microbes that can promote plant growth and/or increase plant resistance to stress conditions, to reduce the use of chemical fertilizers and pesticides while maintaining or increasing yield [
1,
2]. The global market size of plant probiotics for crops was valued at USD 5. 27 billion in 2021 and is projected to reach 15.71 billion by 2029 (
http://www.researchandmarkets.com/reports/5165433/agricultural-microbial-market-growth-trends). Probiotic-induced plant growth promotion commonly involves improvements in nutrient acquisition and the modulation of phytohormone homeostasis [
1], whereas the potential contributions of other cellular processes, such as the biogenesis of ribosomes, which translates mRNAs into proteins and accordingly has an essential role in the control of cell growth, have received little attention. In addition, unlike the apparent effects on plant growth, the effects of probiotics on plant secondary metabolites at the omics level, which are important for the quality of plant-based foods, are generally unclear.
Coriander (
Coriandrum sativum L.), commonly known as cilantro or Chinese parsley, is a worldwide culinary and medicinal plant with both nutritional and medicinal properties [
3,
4]. The leaves of juvenile coriander plants are popular in cuisine for their distinctive aroma. Little is known about how plant probiotics may promote growth or affect the food chemistry profile of coriander plants. The soil bacterium
Aeromonas sp. H1 is a recently isolated probiotic strain effective for many plant species [
5]. In this study, we investigated how the yield and quality of coriander may be affected by
Aeromonas sp. H1 using transcriptomic and metabolomic approaches.
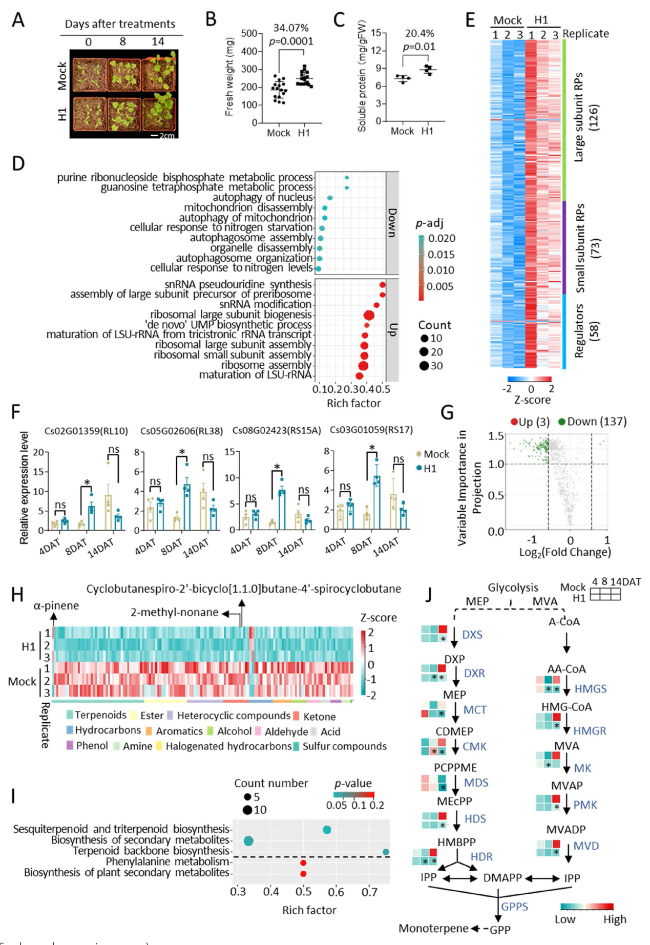
Aeromonas sp. H1 significantly increased the biomass of coriander, as indicated by a 34.1% increase in the aerial fresh weight at 14 days after treatment (DAT) (
Fig. 1A and B; Fig. S1A). The H1-treated coriander also presented increased levels of soluble proteins and soluble sugars but not chlorophylls or carotenoids (
Fig. 1C; Fig. S1B, C). To gain mechanistic insights into H1-induced growth promotion, we profiled the coriander transcriptome via mRNA sequencing of the aerial parts of plants at 8-DAT when growth promotion was about to become apparent, with the rationale that transcriptional regulation precedes the resultant morphological changes. A total of 1123 differentially expressed genes (DEGs, treated vs. control, fold change ≥ 1.5, FDR ≤ 0.05) were identified (Supplementary Table S1; Fig. S1D), including 405 downregulated genes and 718 upregulated genes. Gene Ontology (GO) analysis of the downregulated DEGs highlighted the biological process of autophagy (
Fig. 1D; Supplementary Table S2). This observation suggested that the H1-treated plants were healthier than the control plants, given that autophagy is responsible for recycling dysfunctional cellular components [
6]. GO analysis revealed that the DEGs were characterized by the upregulation of ribosome biogenesis (
Fig. 1D; Supplementary Table S2), which included the large subunit and small subunit ribosomal proteins and regulators of ribosome biogenesis (
Fig. 1E; Supplementary Table S1); together, these DEGs accounted for 22.9% of all the DEGs and 34.8% of all the ribosome biogenesis genes in coriander; these ratios were remarkable since the ratio of all DEGs to all genes in the coriander genome was only 4.6%. The transcriptional elevation of ribosome biogenesis DEGs was observed at 8-DAT but not 4-DAT or 14-DAT (
Fig. 1F), indicating that transcriptional regulation is a hallmark event at the onset of H1-induced growth promotion. In addition to the ribosome biogenesis DEGs, the DEGs involved in tRNA synthesis, translation initiation, and translation elongation were also predominantly upregulated (Fig. S1E, Supplementary Table S1). Thus, the H1-induced growth promotion in coriander was underpinned by the bacterial enhancement of ribosome biogenesis and protein production.
Coriander leaves have a distinctive aroma and are enriched in a variety of secondary metabolites [
3,
4,
7]. To determine whether H1 altered the quality of coriander, we profiled the volatile metabolome of coriander leaves at 14 DAT. The volatile organic compounds (VOCs) were collected from the samples by using solid-phase microextraction (SPME), followed by gas chromatography coupled with mass spectrometry (GC‒MS) analysis. A total of 855 VOC components were identified in the coriander metabolome; 3 and 137 of these compounds exhibited increased and decreased levels, respectively (fold change ≥ 1.5, VIP ≥ 1), as a result of H1 inoculation (
Fig. 1G; Fig. S1F; Supplementary Table S3). Terpenoids accounted for the largest group (30.7%) of the decreased metabolites (
Fig. 1H); moreover, Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis revealed that the decreased metabolites were particularly enriched in terpenoid biosynthesis (
Fig. 1I), including a group of sesquiterpenoids and triterpenoids (Fig. S1G). Terpenoid backbones are synthesized from two universal precursors, isopentenyl pyrophosphate (IPP) and its isomer dimethylallyl pyrophosphate (DMAPP) [
8]. Consistent with the metabolic repression of terpenoid backbone biosynthesis, the H1-treated coriander at 14-DAT presented repressed gene expression of DXR (1-deoxy-D-xylulose 5-phosphate reductoisomerase) and HMGR (hydroxymethylglutaryl-CoA reductase) (
Fig. 1J), which are rate-limiting enzymes in the biosynthetic pathways for IPP and DMAPP, respectively [
8]. H1-treated coriander also showed transcriptional repression of isoprene synthase (Fig. S1H, I), which produces isoprene, although isoprene was not detected in the metabolome, likely because isoprene biosynthesized in situ by isoprene synthase is immediately released and not stored in the leaf [
9]. Interestingly, despite the overall suppression of terpenoid biosynthesis (
Fig. 1J), H1-treated coriander plants exhibited significantly increased levels of α-pinene (
Fig. 1H), a monoterpene of intense medicinal interest due to its antimicrobial, antioxidant, anti-inflammatory, and neuroprotective activities [
10]. In coriander, unsaturated aldehydes, mainly decanal and dodecanal, are described by many people as fruity, green and pungent, while (E)-2-alkenals, mainly (E)-2-decenal and (E)-2-dodecenal, taste soapy to some other people due to genetic variants in olfactory receptors [
11]. The H1-treated coriander plants produced similar levels of both fruity and soapy metabolites as did the control plants (Fig. S1J).