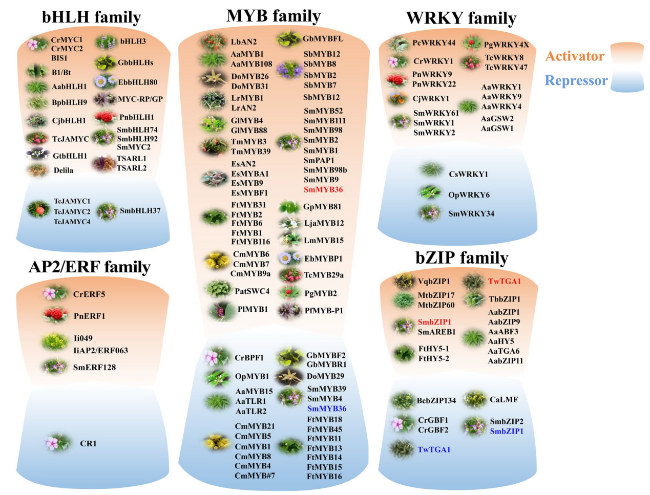
With the development and utilization of active SMs in medicinal plants, more research has shifted to medicinal plants for the biosynthesis and regulation of secondary metabolites. Currently, the regulation of SMs by MYBs in medicinal plants focuses on the biosynthesis of flavonoids, phenolic acids, and terpenoids. In
Scutellaria baicalensis,
SbMYB12 was found to activate the expression of the
SbCCL7-4,
SbCHI-2, and
SbF6H-1 genes and positively regulate the generation of baicalin and wogonoside (Wang et al.
2022a). Additionally, the
GlMYB4 and
GlMYB88 from
Glycyrrhiza uralensis could positively regulate flavonoid synthesis in licorice cells induced by MeJA (Li et al.
2020).
Ginkgo biloba is another medicinal plant rich in flavonoids in which
GbMYBF2 and
GbMYBFL play opposite roles in regulating flavonoid biosynthesis as a repressor and an activator, respectively (Xu et al.
2014; Zhang et al. 2018b). In a series of research studies on
Epimedium sagittatum, Huang et al. identified several MYB proteins that promote flavonoid biosynthesis, such as
EsMYB9,
EsMYBA1,
EsAN2, and
EsMYBF1 (Huang et al.
2013; Huang et al. 2016a; Huang et al. 2016b; Huang et al.
2017). Information on MYB TFs involved in flavonoid synthesis in other medicinal plants is shown in
Fig. 4 and Supplementary Table 2. Two MYB proteins (SmMYB1 and SmMYB2) from
Salvia miltiorrhiza were proven to upregulate the expression of the
CYP98A14 gene and significantly promote salvianolic acid accumulation (Zhou et al.
2021; Deng et al. 2020a, b), and
SmMYB9b and
SmMYB98b positively regulate tanshinone biosynthesis in the hairy roots (Liu et al.
2020).
SmMYB4, on the other hand, functions as a repressor in the biosynthesis of phenolic acids and tanshinones (Tian et al.
2022). An R2R3 type MYB transcription factor
LmMYB15 gene from
Lonicera macranthoides was isolated and characterized by Tang et al. (2021), who found that it might facilitate chlorogenic acid biosynthesis via direct transcriptional activation of the
4CL gene. The transcriptional regulation of terpenoids is mainly found in monoterpene, sesquiterpene, and triterpene saponins. As a kind of sesquiterpene lactone with significant antimalarial effects, artemisinin is synthesized and stored in the glandular trichome of
Artemisia annua leaves.
AaMYB1 could positively regulate trichome initiation and artemisinin biosynthesis, while
AaMYB15 led to a significant decline in the expression levels of the
AaADS,
AaCYP,
AaDBR2, and
AaALDH1 genes and decreased the artemisinin contents in
A. annua (Wu et al.
2021).
AaTLR1 and
AaTLR2 also reduced artemisinin levels by inhibiting trichome development (Lv et al.
2022). In
Panax ginseng,
PgMYB2 was reported to improve ginsenoside production by promoting
PgDDS gene expression (Liu et al.
2019a). Similarly,
PnMYB2 isolated from
Panax notoginseng was considered likely to regulate the biosynthesis of ginsenoside, but its specific functions were still unclear (Xia et al.
2022).
GpMYB81 can bind to the promoters of the
GpFPS1 and
GpCHS genes and activate their expression, acting as a “dual-function” regulator of gypenoside and flavonol biosynthesis in
Gynostemma pentaphyllum (Huang et al.
2022). Furthermore,
OpMYB1 from
Ophiorrhiza pumila and
CrBPF1 from
Catharanthus roseus had the function of regulating alkaloids biosynthesis (Rohani et al.
2016).
EbMYBP1 from
E.breviscapus was a activator involved in the regulation of flavonoid accumulation (Zhao et al.
2022). These results will be helpful for further research on the complex regulatory mechanism of secondary metabolite formation in medicinal plants.