Core
Gene and accession numbers
Introduction
Results
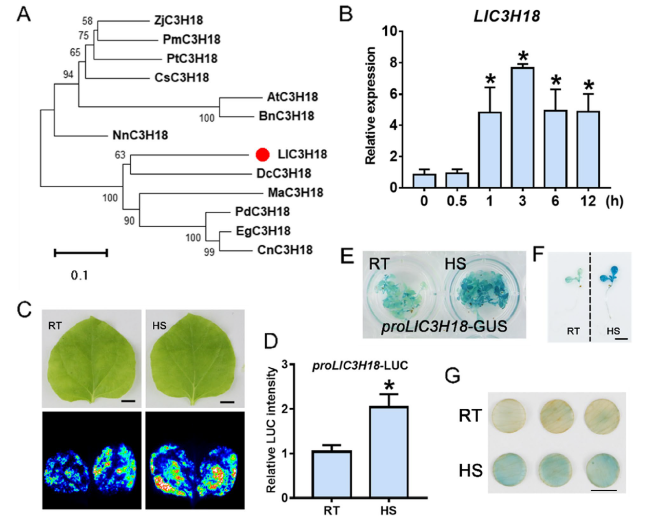
Lily LlC3H18 encodes a non-TZF CCCH protein that is activated by high temperature
Fig. 1 LlC3H18 is a heat-inducible CCCH-type protein. A Phylogenetic tree analysis of LlC3H18 and its homologs from other plant species. The evolutionary tree was assembled in MEGA 7.0 via the neighbor-joining method (bootstrap replicates, n = 1,000). PdC3H18 (Phoenix dactylifera, XP_008800632.1); EgC3H18 (Elaeis guineensis, XP_010907182.1); DcC3H18 (Dioscorea cayenensis, XP_039146376.1); CnC3H18 (Cocos nucifera, KAG1342178.1); NnC3H18 (Nelumbo nucifera, XP_010270169.1); MaC3H18 (Musa acuminata, XP_009394377.1); ZjC3H18 (Ziziphus jujuba, XP_048327362.1); CsC3H18 (Camellia sinensis, XP_028094358.1); AtC3H18 (Arabidopsis thaliana, AT2G05160); PtC3H18 (Populus trichocarpa, XP_024441083.1); PmC3H18 (Prunus mume, XP_008229903.1); BnC3H18 (Brassica napus, XP_048619457.1). B The expression of LlC3H18 in lily leaves under heat stress conditions for different time durations. HS, heat stress, 37°C. Bars indicate the mean ± SD from three replicates (Student’s t-test, * P < 0.05, all treatments compared with 0 h). C The LUC reporter assay of LlC3H18 promoter activity in tobacco leaves at room temperature (RT, 22°C) and under HS (37°C, 3 h). One representative image based on three independent experiments. Scale bar = 1 cm. D Quantification of LUC intensity in panel C. All values shown are the mean ± SD of three replicates (Student’s t-test, * P < 0.05). E The activity of LlC3H18 promoter in proLlC3H18-GUS transgenic Arabidopsis at RT (22°C) and under HS (37°C, 3 h). One representative image based on three replicates. F The single plant of proLlC3H18-GUS transgenic Arabidopsis in (E). Scale bar = 1 cm. G LlC3H18 promoter activity in proLlC3H18-GUS that was transiently expressed lily petal discs at RT (22°C) and under HS (37°C, 3 h). One representative image based on three independent experiments. Scale bar = 1 cm |
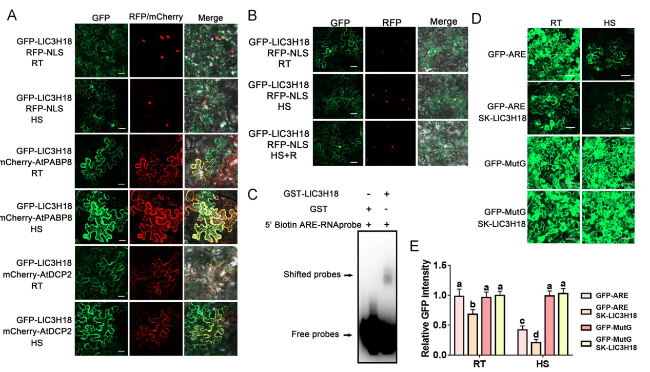
LlC3H18 localizes in cytoplasmic foci in response to heat stress
Fig. 2 Subcellular localization assay of LlC3H18. A Detection of fluorescence signals in tobacco leaf cells co-transfected with GFP-LlC3H18, the nuclear marker RFP-NLS, the PB marker RFP-AtDCP2, the SG marker RFP-AtPABP8 at room temperature (RT, 22°C) and under HS (37°C, 3 h). Scale bar = 50 µm. B Detection of fluorescence signals in tobacco leaf cells co-transfected with GFP-LlC3H18, and the nuclear marker RFP-NLS at room temperature (RT, 22°C), under HS (37°C, 3 h), and after recovery 1 h from HS (HS + R, 22°C). Scale bar = 50 µm. C RNA-EMSA assay of GST- LlC3H18 protein and ARE sequence. One representative image based on three independent experiments. D GFP-ARE or GFP-MutG co-transformed with LlC3H18. GFP-MuG (replace the A residue in ARE with G) as a negative control. RT, room temperature, 22°C; HS, heat stress, 37°C, 3 h. Scale bar = 50 µm. E The GFP intensity in (D) is measured. Data are presented as the mean ± SD of three replicates, with different letters indicating statistically significant difference (Student-Newman-Keuls test, P < 0.05) |
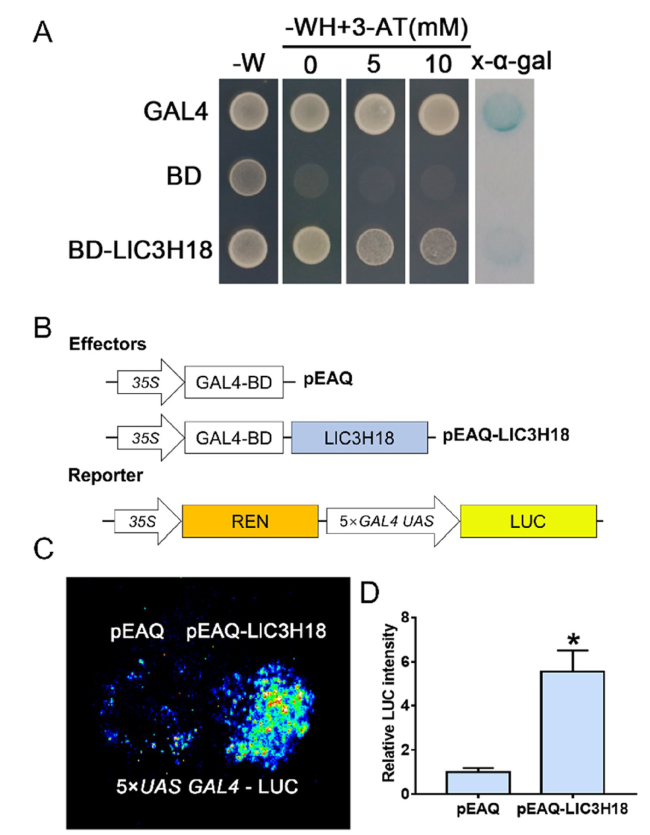
LlC3H18 exhibits transactivation activity in yeast and plant cells
Fig. 3 Transactivation assay of LlC3H18. A Transactivation activity assay in the yeast AH109 strain. The transformants were screened on SD-W medium (lacking Trp) while the growth of transformants was detected on SD-WH medium (lacking Trp/His) containing 3-amino-1,2,4-triazole (3-AT). The color reaction associated with x-α-gal degradation was used as a readout for β-galactosidase activity in the transformants. Representative image based on three replicates. B The constructs for the LUC reporter assay. C Detection of the LUC signal in infiltrated tobacco leaves. The image is representative of three independent experiments. Scale bar = 1 cm. D Measurement of LUC intensity in the reporter assay (Student’s t-test, * P < 0.05) |
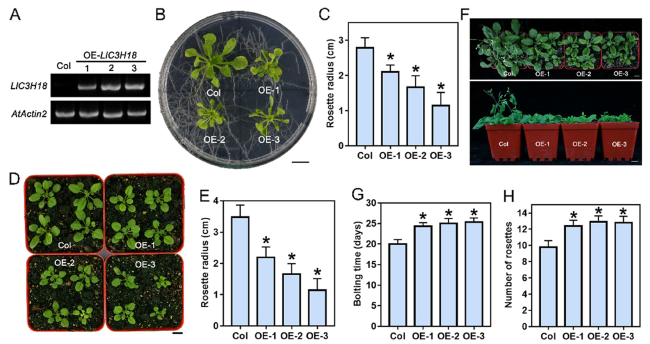
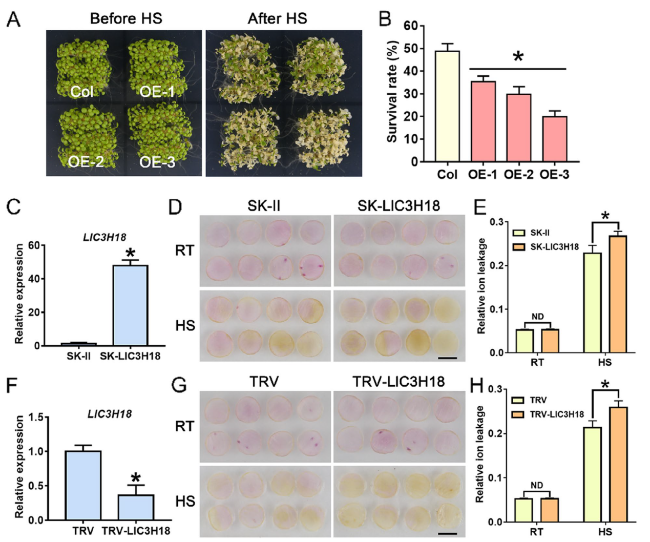
Overexpression of LlC3H18 causes growth defects in transgenic plants
Fig. 4 Overexpression of LlC3H18 causes growth defectives. A Detection of LlC3H18-overexpression lines by RT-PCR. The 5-day-old seedlings were used to detect the expression of LlC3H18 in transgenic Arabidopsis lines. PCR of the endogenous control and test gene was performed with 28 and 30 cycles, respectively. AtActin2 was used as an endogenous control. B Seedlings of wild-type and transgenic lines grown on MS medium for 3 weeks. Scale bar = 1 cm. C Rosette radii of the plants which grown on MS medium for 3 weeks were counted. Bars are means ± SD of the tested plants (n = 9). D The 10-days-old seedlings were transferred from agar plates to soil for two weeks. The representative picture based on three replicates. Scale bar = 1 cm. E Rosette radii of the plants which grown on the soil for two weeks were counted. Bars are means ± SD of the tested plants (n = 9). F The 10-days-old seedlings were transferred from agar plates to soil for three weeks. Scale bar = 1 cm. The representative picture based on three replicates. G Bolting time for wild-type and transgenic lines. Bars are means ± SD of three independent experiments (n = 9, Student’s t-test, *P < 0.05). H The number of rosettes of the bolting transgenic and wild-type plants. Bars are means ± SD of three independent experiments (n = 9, Student’s t-test, *P < 0.05) |
Overexpression of LlC3H18 damages thermotolerance of transgenic plants
Fig. 5 Thermotolerance analysis of LlC3H18-overexpressed and -silenced petal discs, and LlC3H18-overexpressed Arabidopsis plants. A The 5-d-old seedlings were directly exposed to 45℃ conditions for 1 h to detect their thermotolerance ability; the figure is a photo image taken after 7 days of recovery at 22℃. B The survival rate, measured after 7 days of heat stress (HS). Bars are the mean ± SD of three independent experiments (Student’s t-test, *P < 0.05). C Detection of LlC3H18 expression in the LlC3H18-overexpressed petal discs. Data are presented as the mean ± SD of three replicates (Student’s t-test, * P < 0.05). D Phenotypes of lily petal discs under room temperature conditions (RT, 22 °C) and after exposure to heat stress (HS, 40 °C, 12 h). Representative image came from three experiments. Scale bar = 1 cm. E Relative ion leakage (%) of discs at 22 °C (RT) and after HS (40 °C, 12 h). Data are presented as the mean ± SD of three replicates (Student’s t-test, * P < 0.05; ND, no significant difference; the SK-LlC3H18 was compared with the SK-II control under the RT or HS condition, respectively. F Expression of LlC3H18 in TRV-VIGS lily petals. Data are presented as the means ± SD of three replicates (Student’s t-test, * P < 0.05). G Phenotypes of lily petal discs at RT (22 °C) and after HS (40 °C, 12 h). Representative image based on three experiments. Scale bar = 1 cm. (H) Relative ion leakage (%) of discs at RT and after HS (40 °C, 12 h). Data are presented as the mean ± SD of three replicates (Student’s t-test, *P < 0.05; ND No significant difference, TRV-LlC3H18 was compared with the TRV-control under the RT or HS condition, respectively) |
Silencing of LlC3H18 reduces thermotolerance in lily
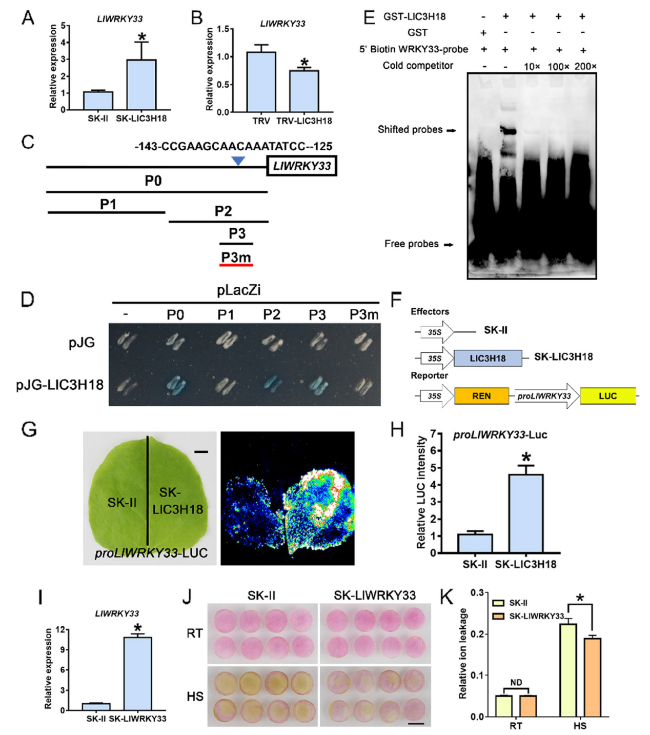
LlC3H18 binds to the promoter of LlWRKY33 and activates its expression
Fig. 6 LlC3H18 binds the promoter of LlWRKY33 and activates its expression. A Detection of the expression level of LlWRKY33 in LlC3H18-overexpressed lily petals. Data are presented as the mean ± SD of three replicates (Student’s t-test, * P < 0.05). B Detection of the expression level of LlWRKY33 in LlC3H18-silencing lily petals. Data are presented as the mean ± SD of three replicates (Student’s t-test, * P < 0.05). C Diagram of the LlWRKY33 promoter. The W-box elements are marked with blue triangles. The truncated fragments used for the yeast one-hybrid (Y1H) assay are marked with black lines. The mutant fragment used for the Y1H assay is marked with a red line. D A Y1H assay for LlC3H18 and the promoter of LlWRKY33. Fragment activity was analyzed by a color change on Ura-/Trp-deficient SD medium following the addition of x-gal. One representative image based on three replicates. E An electrophoretic mobility shift assay (EMSA) of GST-LlC3H18 and the potential elements from the LlWRKY33 promoter. One representative image based on three replicates. F Constructs used in the dual-luciferase reporter assay. G Detection of the LUC signal in tobacco leaves. One representative image based on three replicates. Scale bar = 1 cm. H Measurement of LUC intensity in the dual-luciferase reporter assay. Data are presented as means ± SD of three replicates (Student’s t-test, * P < 0.05). (I) Detection of LlWRKY33 expression in the LlWRKY33-overexpressed petal discs. Data are presented as the mean ± SD of three replicates (Student’s t-test, * P < 0.05). (J) Phenotypes of lily petal discs under room temperature conditions (RT, 22 °C) and after exposure to heat stress (HS, 40 °C, 12 h). Representative image came from three experiments. Scale bar = 1 cm. (K) Relative ion leakage (%) of discs at 22 °C (RT) and after HS (40 °C, 12 h). Data are presented as the mean ± SD of three replicates (Student’s t-test, * P < 0.05; ND, no significant difference; the SK-LlWRKY33 was compared with the SK-II control under the RT or HS condition, respectively |
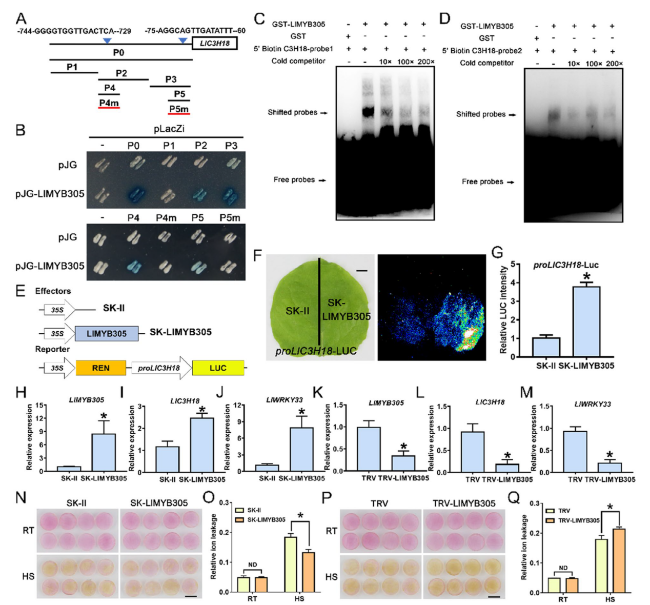
LlMYB305 binds to the promoter of LlC3H18 and activates its expression
Fig. 7 LlMYB305 binds the promoter of LlC3H18 and activates its expression. (A) Diagram of the LlC3H18 promoter. The W-box elements are marked with blue triangles. The truncated fragments used for the yeast one-hybrid (Y1H) assay are marked with black lines. The mutant fragment used for the Y1H assay is marked with a red line. (B) A Y1H assay for LlMYB305 and the promoter of LlC3H18. Fragment activity was analyzed by a color change on Ura-/Trp-deficient SD medium following the addition of x-gal. One representative image based on three replicates. (C and D) An electrophoretic mobility shift assay (EMSA) of GST-LlMYB305 and the potential elements from the LlC3H18 promoter. The probe 1 and 2 came from the core sequence of P4 and P5 fragments, respectively. One representative image based on three replicates. (E) Constructs used in the dual-luciferase reporter assay. (F) Detection of the LUC signal in tobacco leaves. One representative image based on three replicates. Scale bar = 1 cm. (G) Measurement of LUC intensity in the dual-luciferase reporter assay. Data are presented as means ± SD of three replicates (Student’s t-test, * P < 0.05). (H-J) Detection of the expression level of LlMYB305 (H), LlC3H18 (I), and LlWRKY33 (J) in LlMYB305-overexpressed lily petals. Data are presented as the mean ± SD of three replicates (Student’s t-test, * P < 0.05). (K-M) Detection of the expression level of LlMYB305 (K), LlC3H18 (L), and LlWRKY33 (M) in LlMYB305-silencing lily petals. Data are presented as the mean ± SD of three replicates (Student’s t-test, * P < 0.05). (N) Phenotypes of lily petal discs under room temperature conditions (RT, 22 °C) and after exposure to heat stress (HS, 40 °C, 12 h). Representative image came from three experiments. Scale bar = 1 cm. (O) Relative ion leakage (%) of discs at 22 °C (RT) and after HS (40 °C, 12 h). Data are presented as the mean ± SD of three replicates (Student’s t-test, * P < 0.05; ND, no significant difference; the SK-LlMYB305 was compared with the SK-II control under the RT or HS condition, respectively. (P) Phenotypes of lily petal discs at RT (22 °C) and after HS (40 °C, 12 h). Representative image based on three experiments. Scale bar = 1 cm. (Q) Relative ion leakage (%) of discs at RT and after HS (40 °C, 12 h). Data are presented as the mean ± SD of three replicates (Student’s t-test, *P < 0.05; ND, no significant difference; TRV-LlMYB305 was compared with the TRV-control under the RT or HS condition, respectively) |
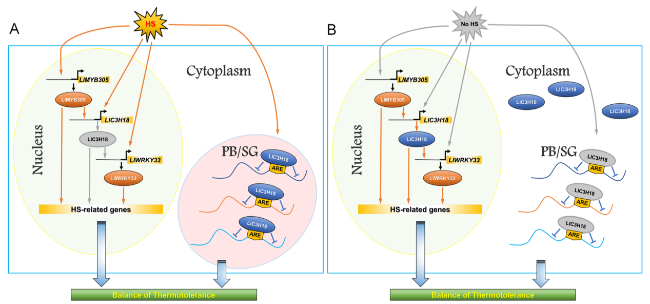
Discussion
Fig. 8 A simple working model of the LlC3H18-mediated regulatory mechanism in lily in response to heat stress. A Under HS conditions, LlC3H18 is a heat-inducible CCCH gene, which can be directly activated by LlMYB305; LlC3H18 locates in the cytoplasm foci and acts as RNA binding protein to form mRNP granules, thus balancing the thermotolerance. B At the recovery period without HS, LlC3H18 can be transformed from cytoplasm foci to localize in the nucleus, which promotes it to act as a trans-activator, directly activating the expression of LlWRKY33, thus forming a heat-inducible LlMYB305-LlC3H18-LlWRKY33 regulatory module, and sustaining the heat stress response. The gray arrow indicates a closed state, while the yellow arrow indicates a working state. HS Heat stress, PB Processing body, SG Stress granule |