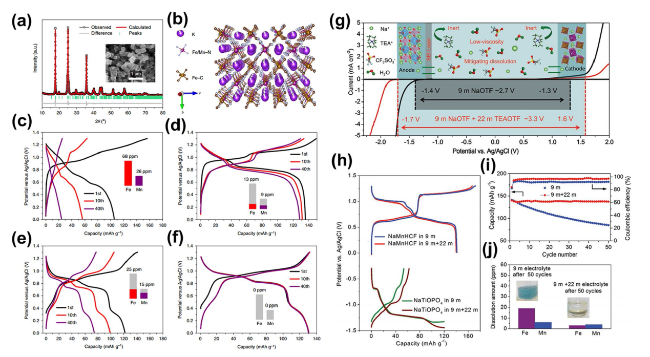
The utilization of economical electrolyte additives presents a more pragmatic and feasible approach compared to the high salt requisites of WiSE. Qian et al. have demonstrated that the introduction of the surfactant sodium dodecyl sulfate (SDS) into the aqueous medium effectively mitigates hydrogen or oxygen evolution at the electrode interface, thereby extending the electrochemical stability window of the electrolyte to approximately 2.5 V [
97]. In this context, SDS molecules adhere to the electrode surface via electrostatic interactions, effectively precluding the precipitation of hydrogen or oxygen. Employing Na
2MnFe(CN)
6 as the cathode and zinc sheets as the anode, the resulting aqueous rechargeable hybrid ion battery manifests an energy density peaking at 170 Wh kg
-1, maintaining capacity retention of 75% across 2000 cycles within an operational voltage of 2.0 V. Energy density computations, grounded in density flooding theory, underscore the pivotal role of SDS in curbing water splitting, manganese dissolution, and anode degradation, thereby enhancing cyclic durability and rate capacity. Empirical evidence suggests that the incorporation of surfactants into aqueous electrolytes augments the electrochemical stability window to 2.5 V. Adjusting the electrolyte composition to match the TM ions of the cathode alters dissolution equilibria, curbing continuous active material attrition, and the incorporation of TM ions within the cathode serves to fortify the structural integrity. Furthering this approach, Li et al. succeeded in bolstering the structural stability of Na
2CoFe(CN)
6 through the integration of specific electrolyte additives (1 wt% CoSO
4) within an aqueous 1 M Na
2SO
4 electrolyte [
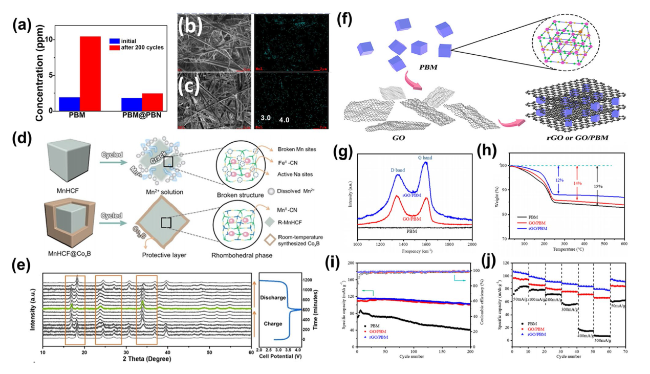
60]. The structural alterations of the electrode skeleton throughout the cyclic process have been examined via XRD, Fourier transform infrared spectroscopy (FTIR), and inductively coupled plasma atomic emission spectroscopy (ICP-AES). These analyses have elucidated that the presence of Co
2+ ions in the electrolyte effectively precludes both the disintegration of the CoHCF skeletal framework and the solubilization of TM ions within the structure during cycling. Similarly, Lu et al. instigated an in situ electrochemical cation substitution in the KMnF cathode by incorporating 0.2 M Fe(CF
3SO
3)
3 into a 21 M KCF
3SO
3 electrolyte [
32]. This innovative approach to surface modification markedly curtailed manganese dissolution, thereby enhancing both structural and surface chemical stabilities of the electrode. Investigations involving scanning transmission electron microscopy (STEM) electron energy loss spectroscopy line scans and elemental mapping were executed on the cross-sectional regions of both original and altered electrodes.
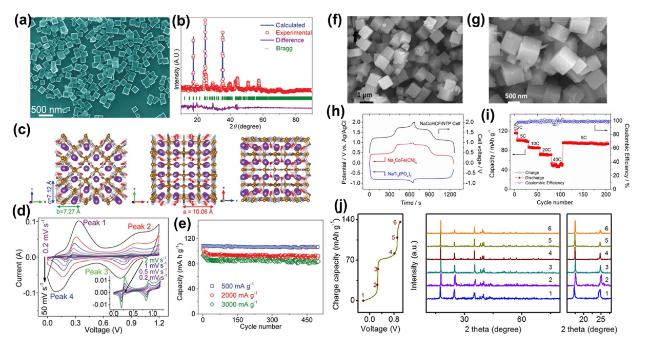
Figure 6a and b displays intensity contour plots and elemental compositions, demonstrating a uniform presence of Fe and Mn in both initial and modified electrodes. Notably, the surface of the altered electrode showed a substantial relative increase in Fe in contrast with Mn, solidifying the conclusion that Fe
3+ is integrated into the electrode framework during cycling in the amended electrolyte. Comprehensive characterizations indicated that Fe present in the modified electrolyte compensates for the Mn vacancies generated in situ within the KMnF structure. Consequently, the KMnF electrode revealed exceptionally prolonged cyclical stability, sustaining a capacity of roughly 120 mAh g
-1 following 130,000 cycles at 2500 mA g
-1 without notable capacity attrition per cycle (
Fig. 6c). In a more nuanced approach, Wang and associates ingeniously incorporated Na
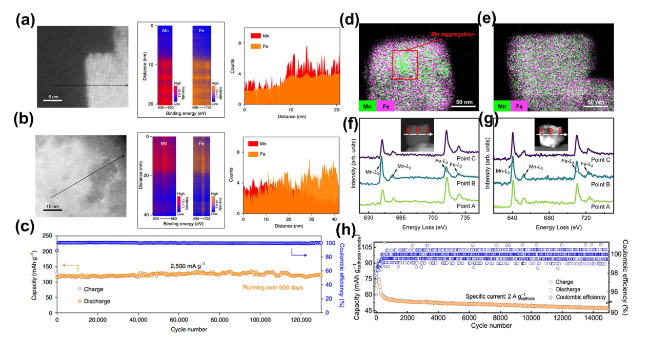
4Fe(CN)
6 as a supplemental salt in the concentrated aqueous NaClO
4-based electrolyte solution, aiming to occupy the Mn vacancies emergent on the surface [
98]. Addressing the lattice expansion ensuing from Na
+ extraction, the [Fe(CN)
6]
4− anion was employed to engage with dislocated or displaced Mn, thereby rectifying Mn vacancies in situ and fortifying the surface's chemical stability. Post 300 cycles utilizing diverse electrolytes,
Fig. 6d illustrated pronounced Mn accumulation within individual particles when subjected to the standard electrolyte. Conversely, Mn distribution remained uniform across particles in the presence of the modified electrolyte (
Fig. 6e), denoting the efficacious curtailment of Mn dissolution. Subsequent analyses employed electron energy-loss spectroscopy (EELS) on the particles' cross-sections harvested from the cycling electrode (
Fig. 6f and
g). Within the unmodified electrolyte system, the Mn L
3/L
2 intensity ratio surged markedly from 4.02 (point A) to 5.18 (point B). This alteration in ratio underscored the Mn's inhomogeneous valence state within discrete particles and the persistence of an elevated valence state, potentially linked to amorphous compounds emanating from structural degradation. In stark contrast, the Mn L
3/L
2 intensity ratio within the modified electrolyte system exhibited negligible fluctuation, signifying a consistent valence state of Mn across the particles. As delineated in
Fig. 6c, the capacity retention of the PTCDI||NaFeMnF stood at 73.4% following 15,000 cycles at 2 A g
-1, with the specific capacity sustained at 47.5 mAh g
-1, and the coulombic efficiency approached a near-perfect 100%.