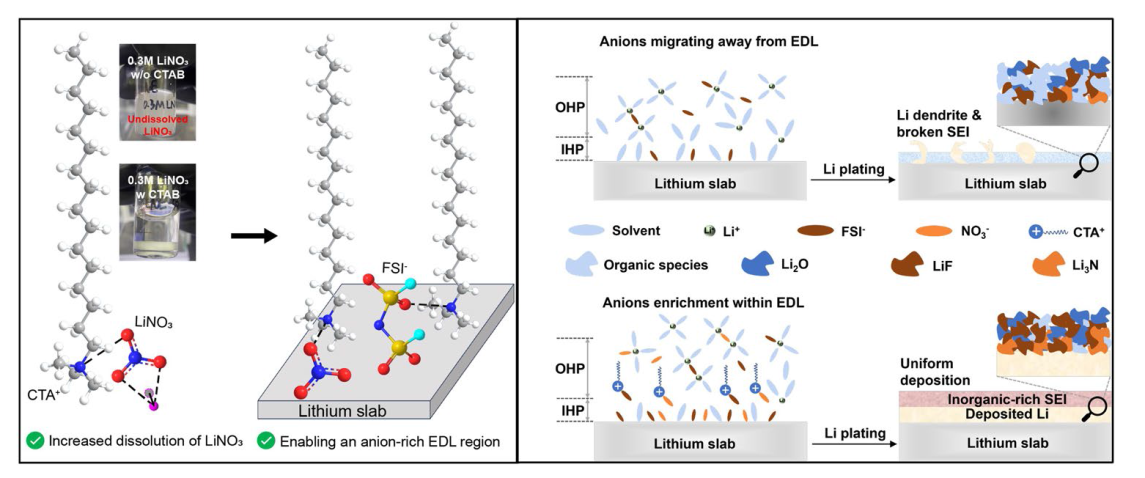
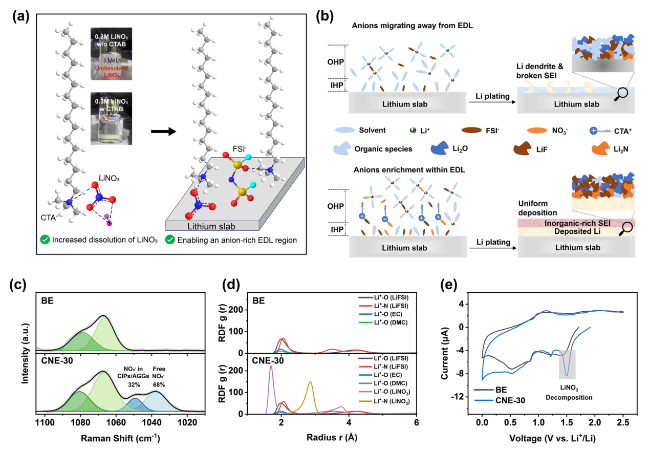
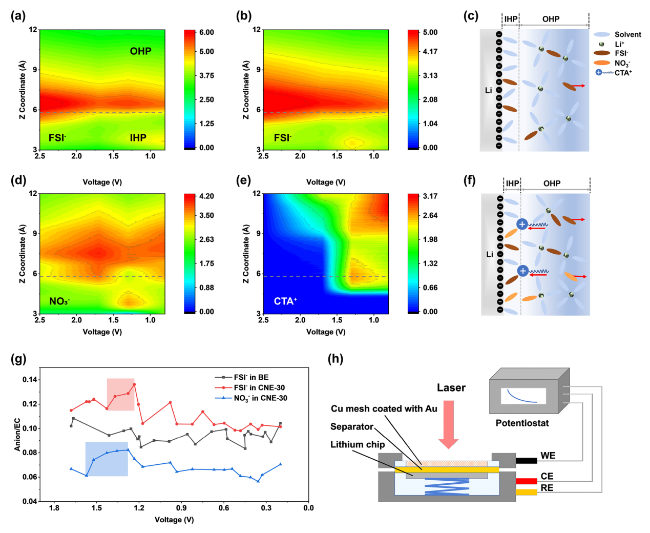
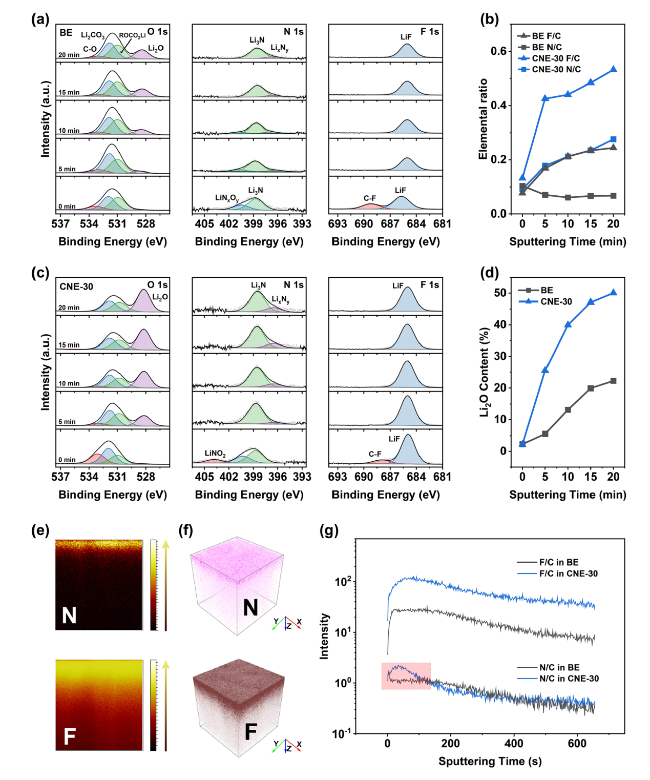
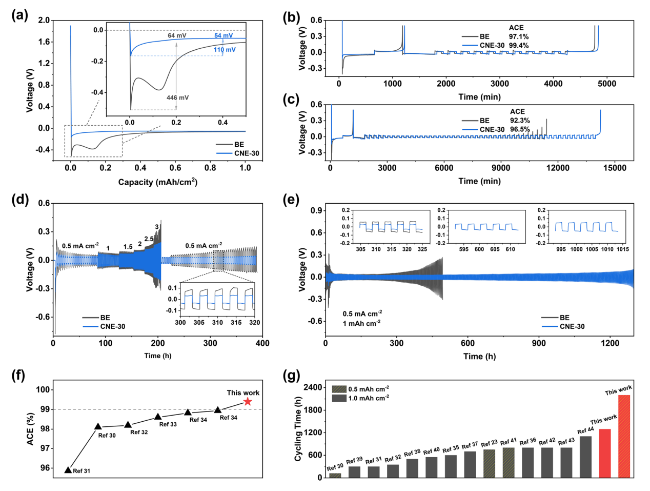
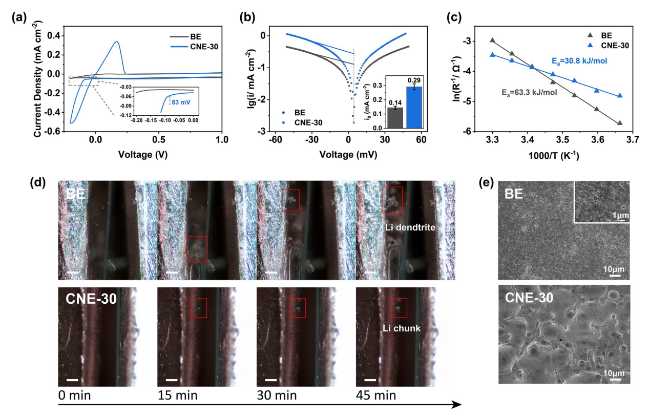
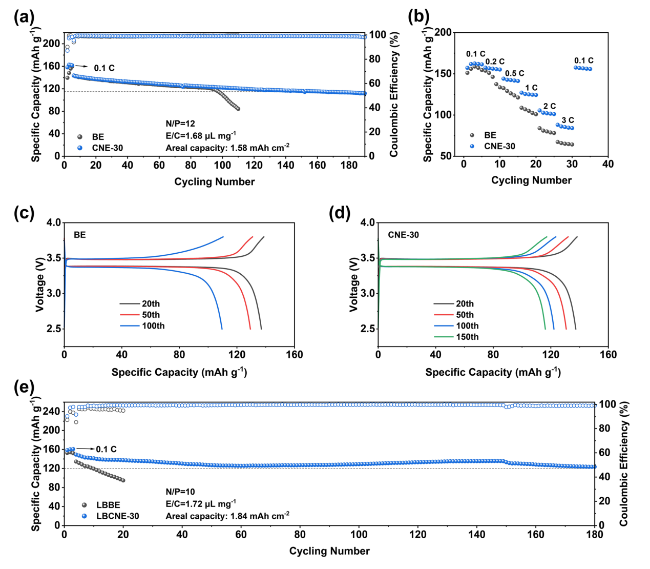
In addition to the Li
+-solvation structure in the bulk electrolyte, the evolution of the component distribution in the EDL region has significant influence on the initial SEI formation, which will be further explored by MD simulations and in situ EC-SERS under various voltages. As shown in
Fig. 2a, b and S6, FSI
−/EC ratios in the BE and the CNE-30 electrolyte were plotted as a function of distance (Å) from graphene electrode under different applied voltages (V). Typically, the Inner Helmholtz plane (IHP) is thinner than 1 nm within EDL region, which can only hold single ion/molecular layer on electrode surface [
13]. In this work, the thickness of IHP is determined to be 5.8 Å according to a peak position of solvent nearest to graphene electrode (Fig. S7). Along with voltage drop from 2.5 to 0.8 V, the component distribution in EDL region is dramatically varied, greatly influencing the subsequent SEI formation. Generally, with the increase in density of electron on graphene electrode, anions will be repulsed away from the IHP while the cations attracted to the IHP. In the BE electrolyte (
Fig. 2a), the FSI
−/EC peak displays attenuated intensity in the IHP region with the decrease in voltages, indicating that FSI
− anions are migrating away from the IHP. However, in the CNE-30 electrolyte (
Fig. 2b, d), significant enrichment of the FSI
− and NO
3− can be observed in the IHP region when the voltage drops below 1.5 V. Besides, the decrease in the FSI
−/EC intensity is obviously mitigated compared to that in the BE electrolyte. Interestingly, as shown in
Fig. 2e, CTA
+ tends to accumulate in the OHP region and the boundary between the OHP and the IHP when the voltage drops from 2.5 to 1.6 V, exhibiting a pattern of enrichment analogous to that of anions in the CNE-30 electrolyte. The accumulation of CTA
+ in the EDL region can be attributed to the applied electric field force, which expulses anions but attracts cations with the decrease in voltages. Due to the steric hindrance, CTA
+ cannot enter the IHP but stay at the outer boundary of IHP. Consequently, the electrostatic interaction between CTA
+ and anions (i.e., NO
3−, FSI
−) dramatically relieves the migration of anions away from the EDL and induces the enrichment of anions in the IHP at specific voltage. One snapshot taken from MD simulations can also verify the enrichment of both NO
3−/FSI
− and CTA
+ near LMA (Fig. S8). Based on the preceding simulation results, the illustrative diagram of anion enrichment within EDL region is presented in
Fig. 2c, f
. To be specific, the force exerted by the electric field, pushing anions away from the anode, is shielded by CTA
+ in the CNE-30 electrolyte, resulting in a higher retention of anions within the EDL region. Therefore, NO
3− and FSI
− undergo preferential reduction to render a dense and inorganic-rich SEI on the surface of LMA. In contrast, driven by electric field force, anions move away from the anode in the BE electrolyte, leading to a greater occupancy of solvent molecules within the EDL region. Consequently, the SEI formed in the BE electrolyte is mainly derived from solvent reduction.