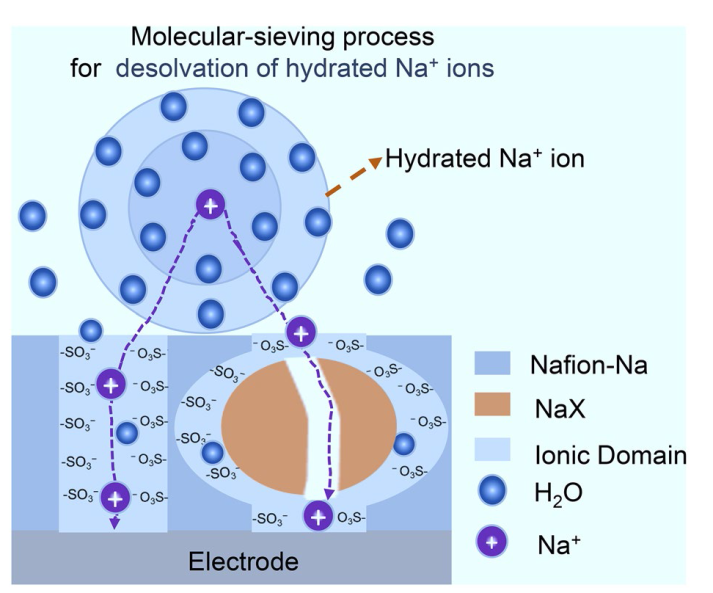
The NaX zeolite was synthesized according to previous reports [
31] and details are provided in the Supporting Information. After high-temperature (450 °C) vacuum drying, NaX was subjected to infrared characterization (Fig. S6). NaX exhibits typical adsorption bands at around 3400 and 3700 cm
−1, revealing the presence of hydroxyl groups (-OH) on the skeleton [
44]. As shown in Fig. S2, the element composition of NaX was verified by inductively coupled plasma-optical emission spectrometry (ICP-OES, Si/Al/Na = 1.0/2.09/1.66). The XRD pattern of synthesized NaX is well matched with PDF#38-0237 (standard figure); N
2 adsorption-desorption analyses reveal the high Brunner − Emmet − Teller (BET) surface area (488.06 m
2 g
−1) and the pore width (0.39 nm) of obtained NaX (Figs. S3 − S5). To avoid the potential side reactions, the sulfonic acid moieties of Nafion were neutralized by NaOH, and the product was named Nafion-Na. As verified by attenuated total reflectance infrared (ATR-FTIR) spectra (
Fig. 1a), after being mixed with NaX, the peak belonging to the -SO
3- site of Nafion-Na shifted from 1060 to 1062 cm
−1 along with peak broadening, which is ascribed to the hydrogen-bonding between -SO
3- of Nafion-Na and hydroxyl groups of NaX [
31]. TEM images provide visible evidence for the ionic domains (hydrophilic regions) formed by the aggregation of sulfonic groups and NaX nanoparticles (both are dark regions) in the samples stained with Pb
2+. The ionic domains in Nafion-Na are around 5 nm in diameter [
29] (
Fig. 1b), whereas the ionic domain sizes decrease with the incorporation of NaX (
Fig. 1c), indicating a less distinct phase separation in the Nafion portion of the Nafion-Na/NaX composite. NaX particles (the dark regions with a diameter of tens of nanometers in
Fig. 1c) are evenly distributed in the composite, verifying the polymer-zeolite compatibility. The small angle X-ray scattering (SAXS) profile of Nafion-Na (saturated with 2 m aqueous NaOTF) exhibits a scattering peak of the ionic domains in line with previous reports of Nafion under a fully hydrated condition [
29] (
Fig. 1d); however, this peak for Nafion-Na/NaX shifts to a higher
q value, corresponding to a lower
d-spacing (recognized as the average diameter of ionic domains, about 3.9 nm in width) (
d = 2π/
q), confirming the formation of downsized ionic domains. It is clear from ATR-FTIR, TEM and SAXS results that the hydrogen-bonding interaction between Nafion-Na and NaX induces an orientation of -SO
3- along the NaX surface [
45,
46], reducing the concentration of hydrophilic regions in the bulk phase (Fig. S6). This hinders the self-assembly process of -SO
3- and prevents the formation of large ion channels, as illustrated in Fig. S7, which may bring molecular selective permeance due to the size-sieving effect.