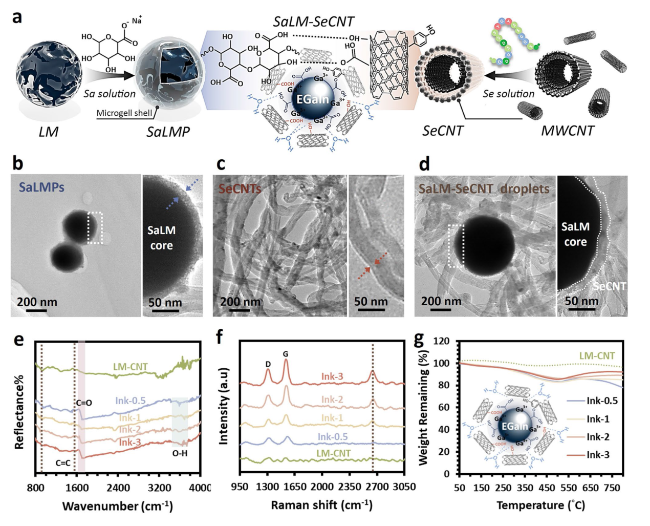
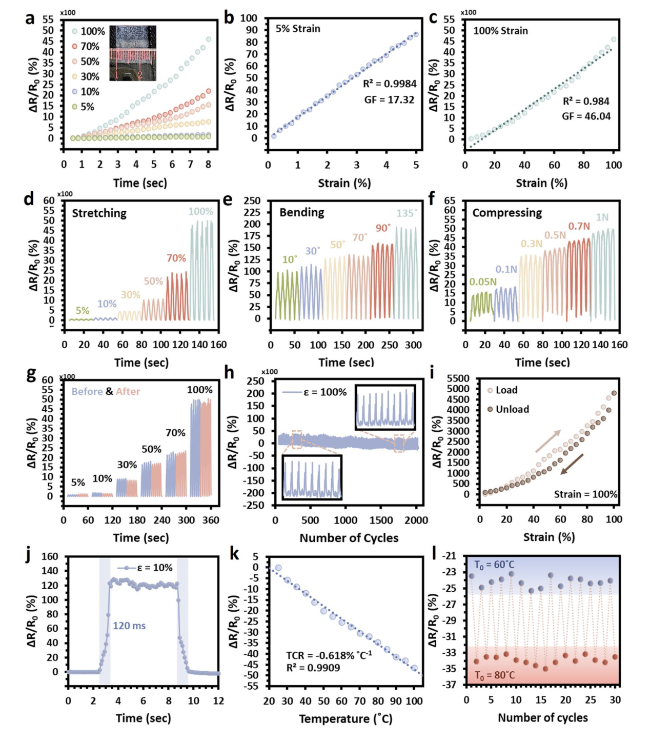
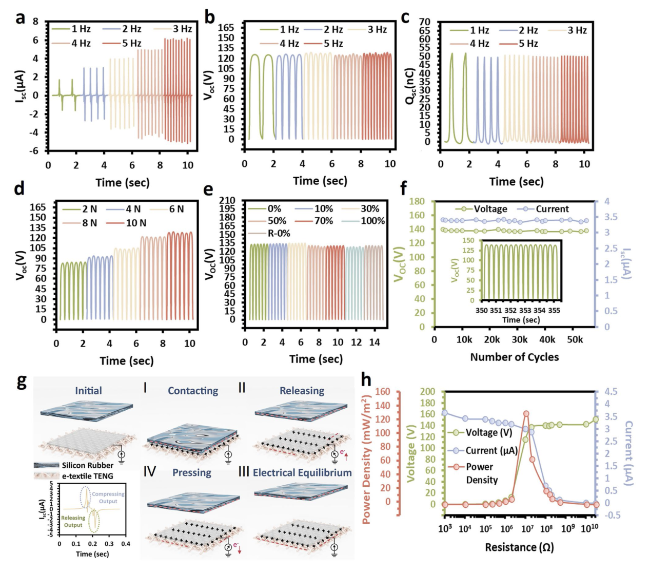
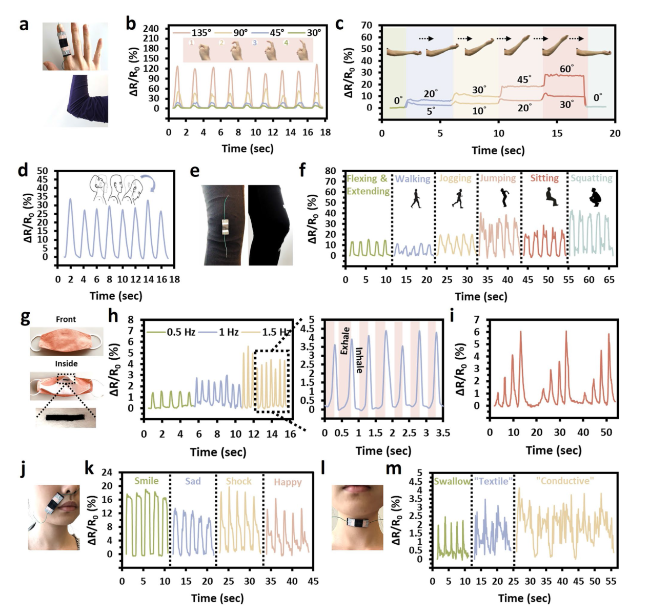
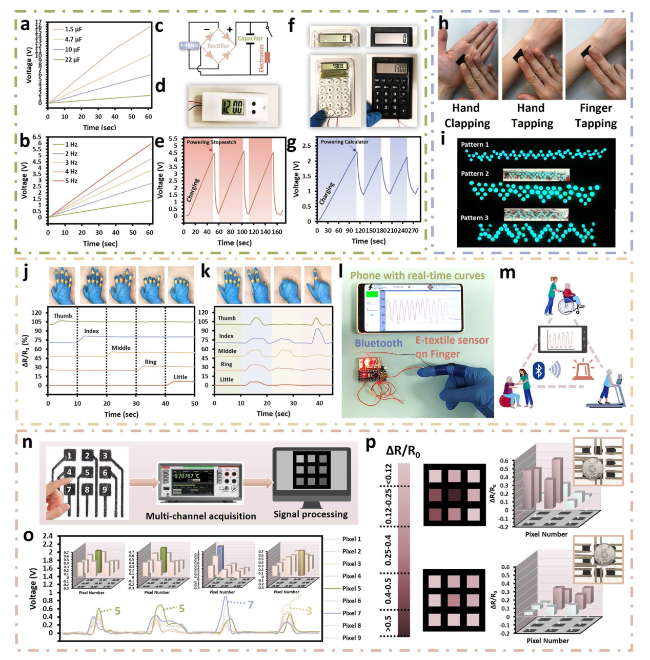
The hypothesis on the mechanisms underlying the structure of SaLM-SeCNT was preliminarily examined by transmission electron microscopy (TEM) images. Starting from bulk LM, it was micronized by powerful sonication in Sa aqueous solution and resulted from Sa-coated LM nanodroplets with polydispersity. The high carboxyl groups on the surface of Sa-coated LM nanodroplets were then combined with sericin-modified MWCNTs aqueous dispersion. The hydrophilic-rich groups of adsorbed sericin induced MWCNTs to form hydrogen bonds which could act as the “carbonaceous web” and form crosslinking networks with the rich carboxyl groups on SaLM nanodroplets (
Fig. 2a). The globoid LM nanodroplet is tightly coiled by SeCNTs, suggesting the presence of strong interactions between the surface of the LM nanodroplets and SeCNTs as seen in
Fig. 2b-d, validating that SaLM nanodroplet surfaces can be efficaciously encapsulated by SeCNT. The TEM image distinctly showed the core-microgel shell structure of the SaLM nanodroplet which was obtained spontaneously during the sonication (
Fig. 2b), and the Sa molecules were able to attach to the oxide layer of the LMs via metal coordination. Synchronously, uniformly dispersed SeCNTs with an interlayer spacing were also confirmed (
Fig. 2c), consistent with the previously reported diameter for the walls of the CNT and the adsorbed sericin [
52,
53]. To investigate the fact that there is a crosslinking between the SaLM nanodroplets and SeCNTs, a middle layer can be observed in
Fig. 2d. Of note, the structural attributes and chemical composition further verified the presence of the hydrogen bond crosslinking networks between SaLM-SeCNT. It was taken by comparing pure LM-CNT to four SaLM-SeCNTs samples with different conductive loading ratios. The former (i.e., pure LM-CNT) was constructed with no addition of Sa and Se whereas the latter four were prepared with the addition of Sa and Se. Aiming to study the effect of SeCNT fillers in generating stable non-covalent bonds, four SaLM-SeCNTs samples were conducted following four different weight ratios of SaLM to SeCNT from 0.5, 1, 2, to 3 and were labeled as Ink-0.5, Ink-1, Ink-2, and Ink-3, respectively. Detailed information is provided in Table S1. The signal of metallic Ga and Ga
2O
3, the carbon bonds (C=C), and carbon-oxygen bond (C=O and O-C=O) in LM-CNTs and SaLM-SeCNTs can be defined by FTIR spectra in
Fig. 2e. The characteristic peak attributable to the Ga and Ga
2O
3 located at 950 cm
−1 was decreased significantly upon the increase of SeCNTs. This phenomenon was mainly ascribed to the formation of a complexation reaction between Ga
3+ ions with carboxyl functional groups on the CNT [
36,
54]. The double carbon bond (C=C) located at 1670 was also decreased by increasing the addition of SeCNTs spectra. A new absorption peak corresponding to the carbonyl group (C=O/O-C=O) motif at 1710 cm
−1 in the SaLM-SeCNT proved the carbonyl group and bonding in the SaLM-SeCNT droplet which was shifted to low-wavenumbers due to the strengthened interaction between sericin and CNT with the increasing content of CNTs. In addition, the stretching vibration peak of O-H also appeared significantly broad and with higher intensity in SaLM-SeCNT which was shifted from 3466 to 3585 cm
−1. The shift of these peaks attested to hydrogen bonds between SaLM and SeCNT. Additionally, the interface strength between SaLM cores and SeCNT wrappings could be further evaluated by Raman spectrum. The peaks of the D band (
ID) and G band (
IG) for the SeCNT web in the SaLM-SeCNT with different ratios are summarized in
Fig. 2f, which reflects the evolution of crosslinking networks to some extent. The
ID was related to the disorder degree of the system, whereas the G peak was attributed to the carbon atoms of CNTs having a complete hexagonal structure (
sp2 hybridization) [
55], which could measure the defect concentration of SeCNTs and determine the interface strength [
56]. As presented in
Fig. 2f, the
ID/
IG of LM-CNT and SaLM-SeCNT is shifted from 1339 to 1347 cm
−1 and 1554 to 1568 cm
−1, respectively, in which the
ID to
IG ratio increases from 0.144 to 0.383, showing an enhancement of > 100%. Moreover, the
D peak and
G peak also increased from 110.0 of the Ink-0.5 up to 219.5 of the Ink-3, and 128.4 of the Ink-0.5 up to 345.7 of the Ink-3, respectively. It reveals that more defective structures on SeCNT made it easier to form a strong interface interaction with SaLM, thereby obtaining a better performance on the composites which will be discussed later. Additionally, the content of Sa and Se coating in the SaLM-SeCNT nanodroplet was roughly evaluated by thermogravimetric analysis (TGA). It notes that pure LM-CNT nanodroplets demonstrated relatively stable thermal stability due to the lack of a polymer layer. As for SaLM-SeCNT, a continuous weight loss of 20 wt% is observed till 500 °C, corresponding to the thermal degradation of the Sa and Se layer (
Fig. 2g). These results manifest that the interaction of SeCNT on the surface of SaLM cores could be efficiently constructed based on the proposed strategy.