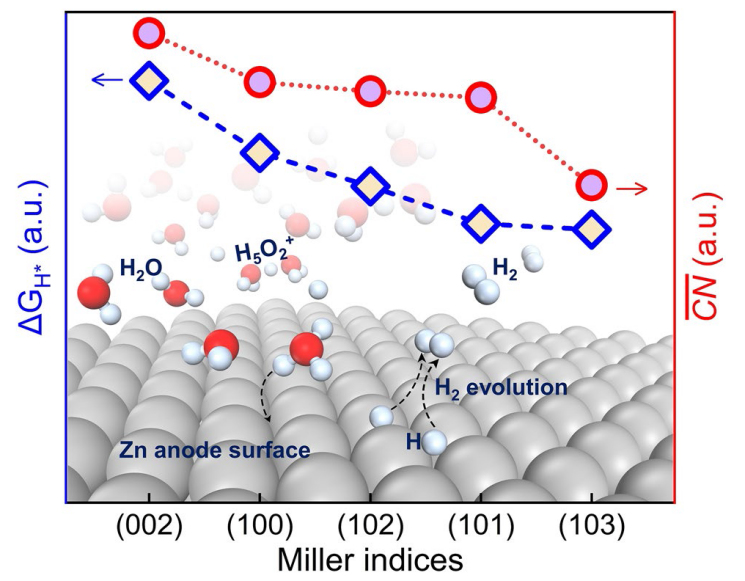
The hydrogen-adsorption free energy (
$\Delta {G}_{{{\text{H}}}^{*}}$) is used to predict the HER activity of catalysts from a thermodynamic aspect. According to Sabatier's principle [
31], a good HER activity of catalysts need to bond with the H atom neither too strongly nor too weakly. If the H atom bonded to the surface too weakly, the H adsorption step (Volmer) will be difficult to take place. In contrast, the desorption (Heyrovsky/Tafel) step will hard to be proceed when H bonded to the surface too strongly [
32].
$\Delta {G}_{{{\text{H}}}^{*}}$ ~ 0 is regarded as the optimal value for good HER activity from the thermodynamic point of view [
45]. Thus,
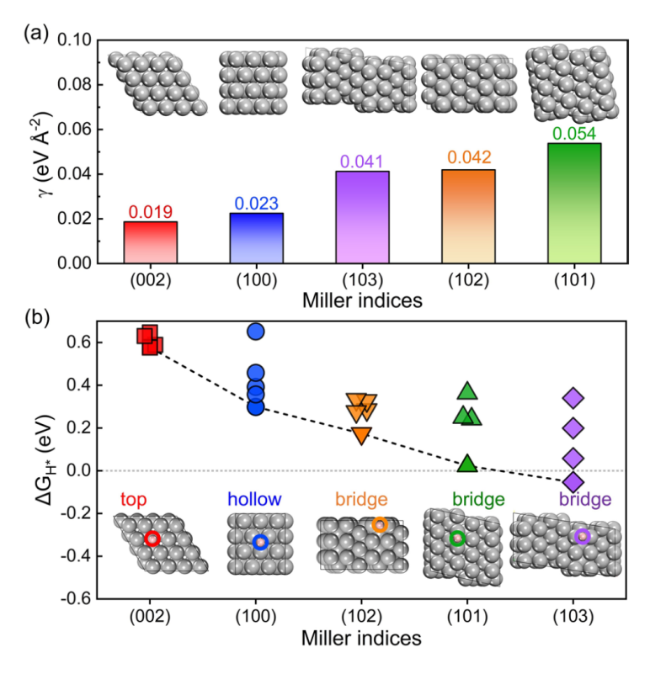
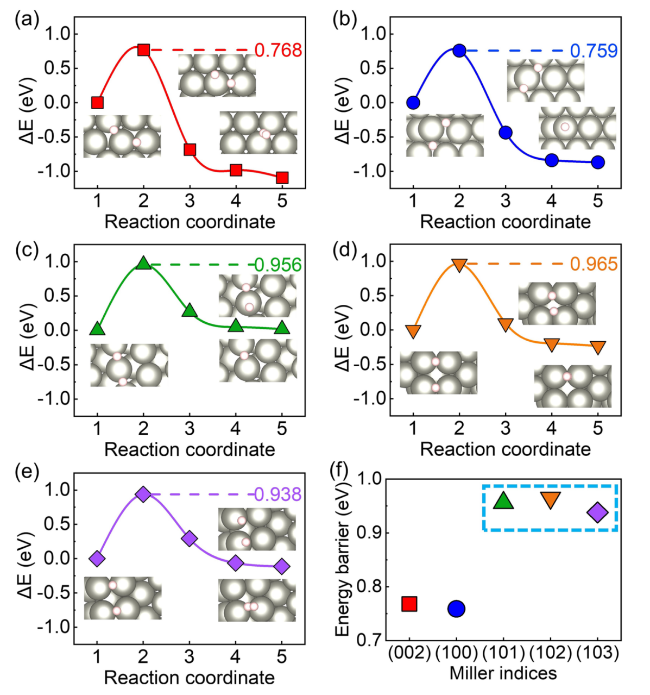
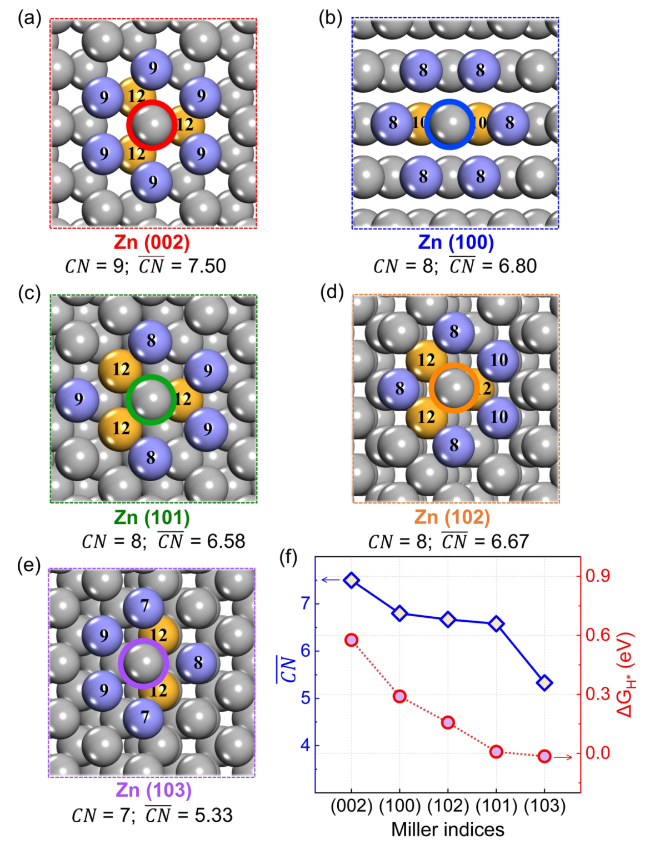
$\Delta {G}_{{{\text{H}}}^{*}}$ at different site of Zn (002), (100), (101), (102), and (103) surfaces were calculated to insight into the thermodynamic aspect of HER activity of Zn anode, as shown in
Fig. 1b. In addition, Van der Waals interactions with Grimme's D3 scheme have been tested for hydrogen-adsorption models (Fig. S2). The
$\Delta {G}_{{{\text{H}}}^{*}}$ values are slightly changed, while the relative values of
$\Delta {G}_{{{\text{H}}}^{*}}$ for several crystal surfaces are almost unchanged after considering the Van der Waals interactions. The most stable adsorption site for each crystal surface is identified, which is presented in the insert of
Fig. 1b. The adsorption energies of H atoms at bridge sites on the (101), (102), and (103) surfaces are significantly lower than those at other sites, suggesting that the bridge sites are the most stable adsorption site for (101), (102), and (103) surfaces. The detailed
$\Delta {G}_{{{\text{H}}}^{*}}$ values and the corresponding structures of H adsorbed at (101), (102), and (103) surfaces are shown in Tables S2–S4 in the supporting information, respectively. And H prefers to be absorbed on the top and hollow sites of (002) and (100) surfaces, respectively. All the adsorption sites and the correspongding
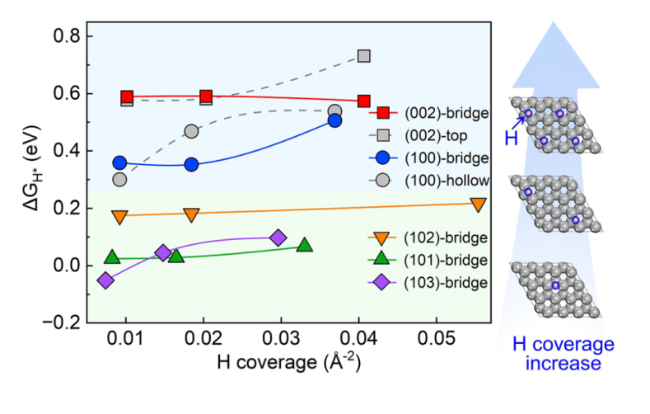
$\Delta {G}_{{{\text{H}}}^{*}}$ values at (002) and (100) surfaces that were considered here are shown in Tables S5, S6 in the supporting information, respectively. However, the bridge site adsorption becomes more stable as the H coverage increases, which will be discussed in the following part. The lowest value of
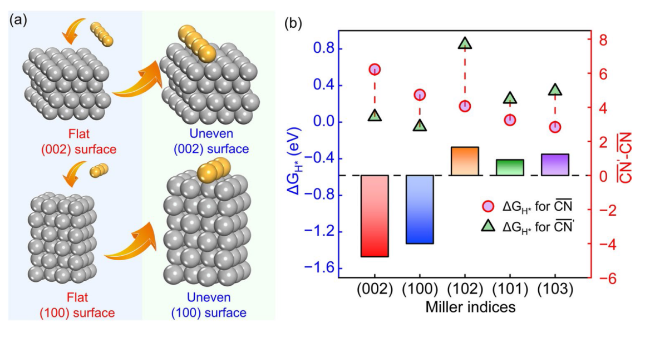
$\Delta {G}_{{{\text{H}}}^{*}}$ for the Zn (002) surface is the highest one among several crystal surfaces. The Zn (002) and (100) surfaces show higher
$\Delta {G}_{{{\text{H}}}^{*}}$ for the most stable adsorption site than (101), (102), and (103) surfaces. Keep in mind that the above values of
$\Delta {G}_{{{\text{H}}}^{*}}$ are calculated at a low hydrogen coverage, and the hydrogen coverages are not the same for different crystal surfaces of Zn metal. Besides, H
2O adsorption energies on several crystal surfaces have been calculated (Fig. S3). The adsorption energy differences are within 0.1 eV. Generally, the trends of H
2O adsorption energies are similar to the H adsorption energies. The (002) surface shows the highest H
2O adsorption energy, indicating that H
2O molecules are less likely to adsorbed at (002) surface.