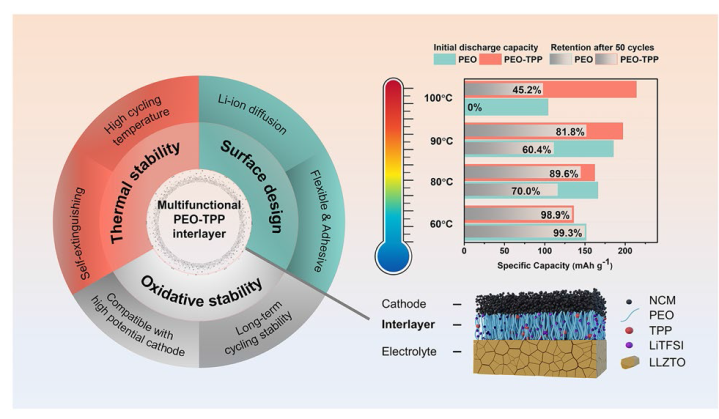
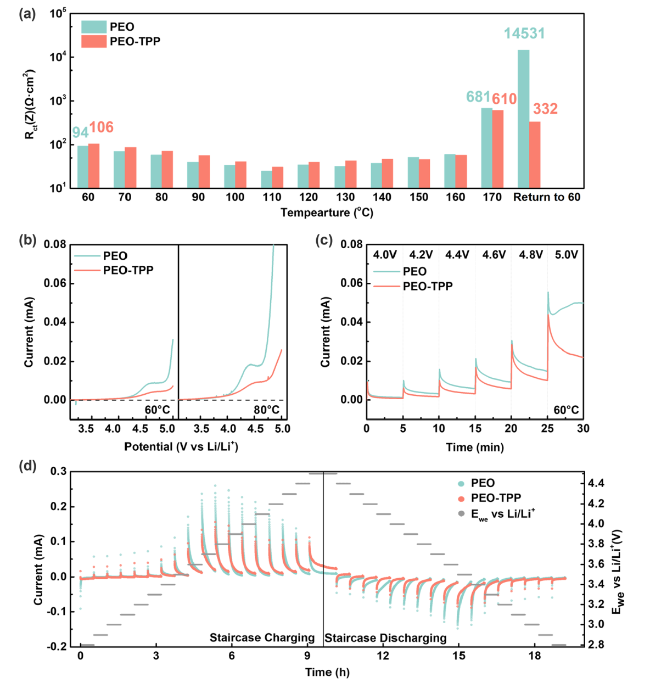
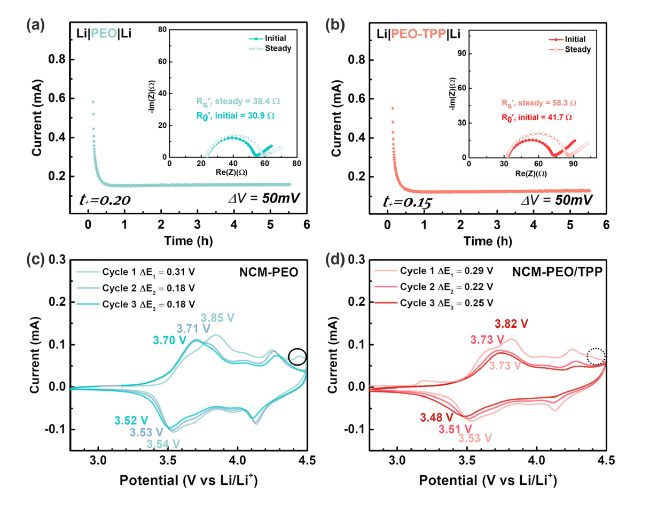
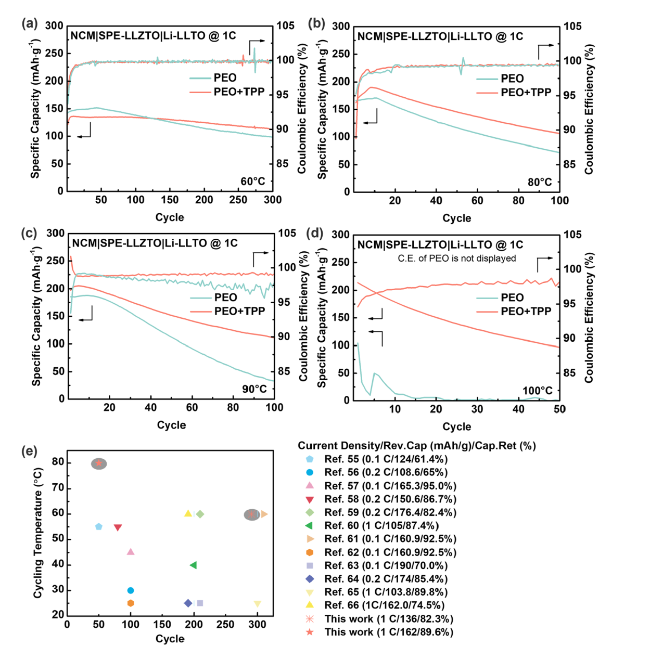
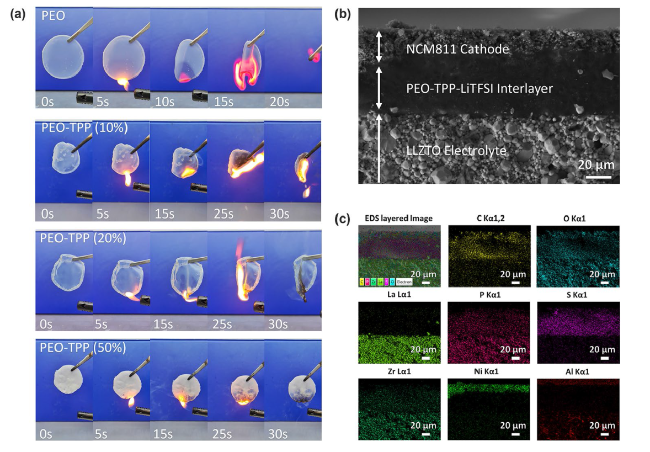
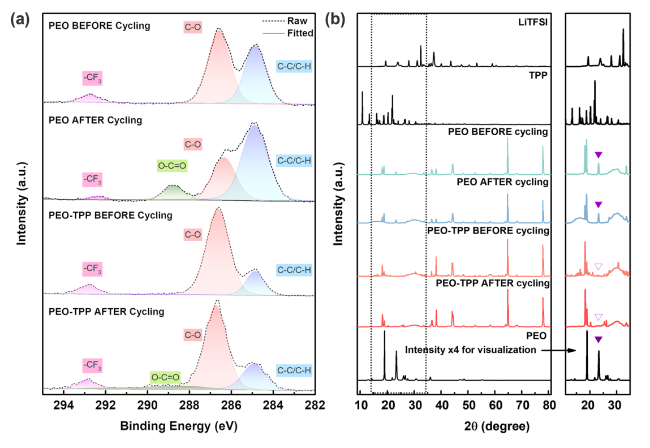
It was reported that some functional additives could facilitate the formation of cathode-electrolyte interphase (CEI) [
28]. During electrochemical oxidation, such additive decomposes prior to the other electrolyte components to form a kinetically protective CEI layer, which suppresses further decomposition of the electrolyte at the electrode. Certain flame retardants (F- and P-based) used in LIBs can decompose at the electrode to form a protective layer in the same way to prevent thermal runaway [
29,
30,
31,
32,
33,
34,
35,
36,
37,
38]. For example, Cui et al. incorporated TPP into PVDF-HFP to substitute the conventional membrane separator in conventional LIBs, thereby introducing flame-retardant properties triggered by thermal decomposition of PVDF-HFP [
39]. Recently, we have used liquid-state trimethyl phosphate (TMP) as wetting agent to realize highly stable anodic and cathodic interfaces with improved physical contact and chemical stability [
35]. In this study, we selected TPP as an additive to enhance the electrochemical and thermal stability of the PEO-based catholyte considering its widespread use, cost effectiveness, and efficient performance as a phosphorus-based flame retardant [
40]. With its distinct solid-state nature at RT, TPP plays a crucial role in tackling thermal stability challenge as a flame-retardant additive and contributes to the realization of an all-solid-state battery solution. As depicted in
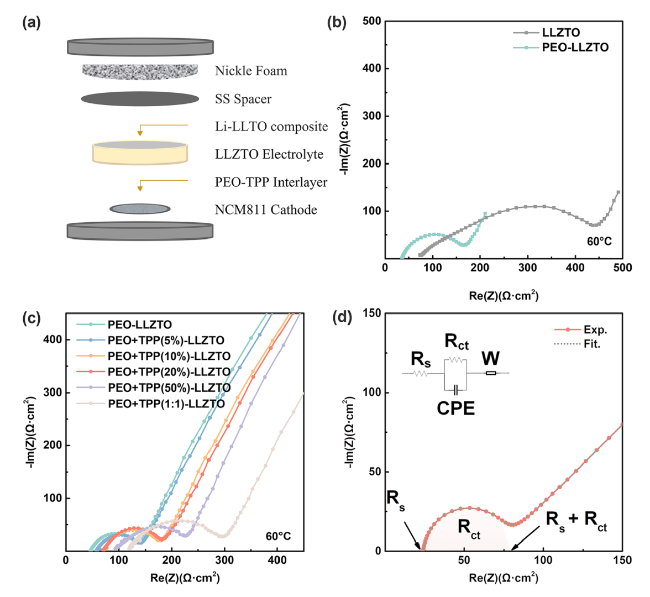
Fig. 1c, the introduction of TPP into SPEs results in an escalation of charge transfer resistance (
Rct) at the interface, and the trend becomes more pronounced when TPP content reaches a mass ratio of 1:1 with PEO electrolyte. This phenomenon may be related to the high viscosity of TPP and its influence on the solvation of lithium-ion charge carriers [
41], indicating the need for content optimization. It was found the obvious fire-retardant effect was demonstrated when TPP content is 20% or higher, which will be discussed in more detail later. The
Rct (charge transfer resistance at the interface) and
Rs (bulk resistance of electrode and electrolyte material) of the electrolyte can be estimated by fitting the Nyquist plot with equivalent circuit model as demonstrated in
Fig. 1d. Although the addition of 20 wt% TPP into PEO increased bulk resistance of the entire PEO-LLZTO system from 38.5 to 71.9 Ω·cm
2, there is minimal effect on charge transfer resistance (107.3 Ω·cm
2 for PEO-LLZTO and 119.5 Ω·cm
2 for PEO/TPP-LLZTO) at 60 °C. Conversely, increasing TPP content to a 1:1 mass ratio with PEO electrolyte not only raises
Rb from 38.5 to 119.6 Ω·cm
2, but also results in a much larger
Rct (177.8 Ω·cm
2) compared to pristine PEO-LLZTO. In light of achieving a suitable equilibrium between fire retardancy (as demonstrated in
Fig. 5a) and interfacial resistance, we selected a composition of 20 wt% TPP within the PEO-TPP system as the optimized catholyte for subsequent experimental demonstrations.