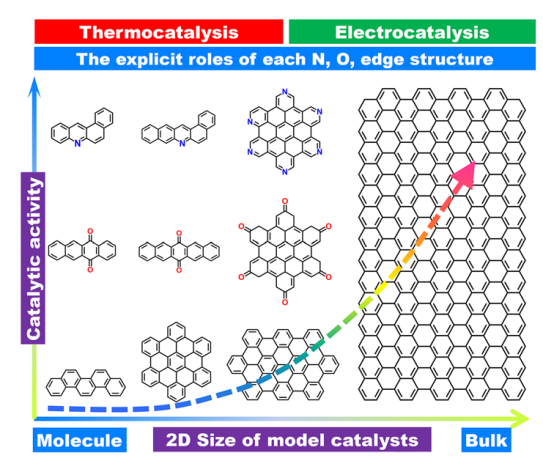
HIGHLIGHTS
1 Introduction
2 Fundamental Organic Molecule Models
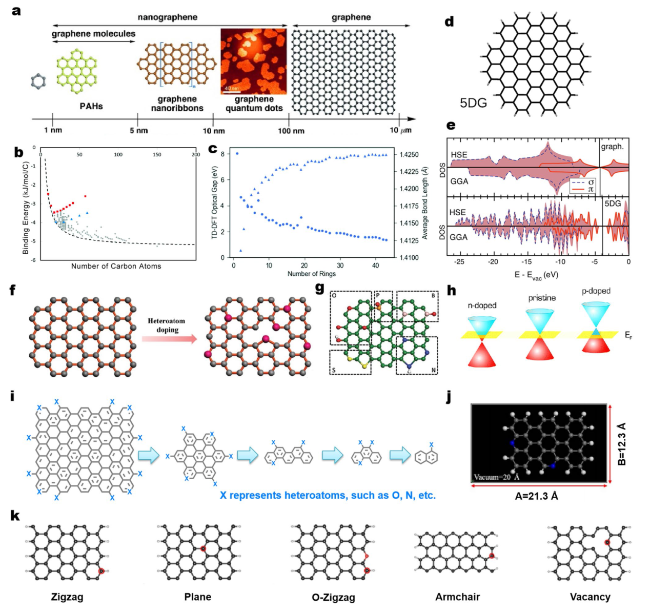
Fig. 1 a Schematic illustration of graphene terminology defined according to their size scale. Graphene nanomolecules are a subset of graphene with a size between 1 and 5 nm. Nanographene units are graphene fragments with diameters of < 100 nm, while graphene should exceed 100 nm in both directions. Reprinted with permission from Ref. [49]. Copyright 2012 Wiley-VCH. b Binding energy as a function of number of carbon atoms in the monomer for the complete set of benzenoid molecules studied. The dashed line depicts the empirical binding energy relationship. c TD-DFT optical gap (circles) and average bond length (triangles) as a function of number of rings for the circular catenation PAH series. Reprinted with permission from Ref. [52]. Copyright 2017 Royal Society of Chemistry. d, e Comparison of the density of states (DOS) for large PAHs 5DG with graphene, calculated within range-separated hybrid functional (HSE) (top) and, generalized gradient approximation (GGA) (bottom). The blue dashed and red solid lines represent π and σ states respectively. The straight line indicates the Fermi energy level. Reprinted with permission from Ref. [54]. Copyright 2015 Elsevier. f Heteroatom doping process on graphene. g Schematic representation of the model of different heteroatom (B, N, O, S, and P)-doped graphene. Here, green, pink, blue, red, yellow, and orange spheres stand for C, B, N, O, S, and P atoms, respectively. Reprinted with permission from Ref. [55]. Copyright 2019 Wiley-VCH. h Band structure of graphene showing p- and n-type doping regarding the Fermi level. Reprinted with permission from Ref. [56]. Copyright 2016 Royal Society of Chemistry. i Fragmentation of doped graphene into small heteroatoms-doped PAH molecules. j The common theoretical model regarding N-doped carbon-based structures. The sizes of vacuum containing unit cell are about 2 nm. Blue spheres represent N atoms. Reprinted with permission from Ref. [57]. Copyright 2019 Nature Publishing Group. k The common theoretical models regarding carbon-based structures involving zigzag, plane, O-containing zigzag, armchair, and vacancy configurations from Ref. [58]. The number of used carbon atoms in theoretical models ranges from 22 to about 100. All the edge carbon atoms are terminated with hydrogen atoms. (Color figure online) |
3 Brief Historical Perspective on the Application of Model Catalysts
4 Molecule Models in Thermocatalytic Reactions
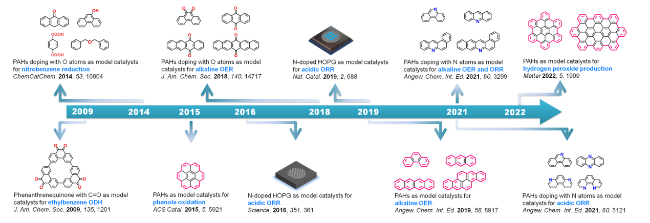
Fig. 2 Development of model catalysts in metal-free carbon catalysis. Here, ODH stands for oxidative dehydrogenation reactions |
4.1 Gas-Phase Oxidative Dehydrogenation Reactions
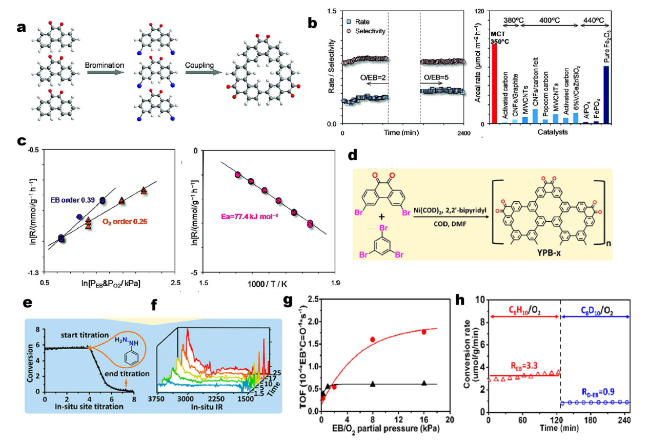
Fig. 3 a Schematic synthesis of the fused phenanthrenequinone (macrocyclic trimer, MCT) model catalyst toward ethylbenzene (EB) oxidative dehydrogenation (ODH) reaction. b Reaction rate (mmol g−1 h−1) and styrene selectivity on MCT oligomer (left side) and comparison of activities with typically reported catalysts (right side). c Dependencies of reaction rate on partial pressures of each reactant (left side) and temperature (right side). Reprinted with permission from Ref. [68]. Copyright 2008 American Chemical Society. d Schematic drawings of the synthesis procedure for model catalysts (YPB-x). e EB conversion as a function of time on stream during the steady-state activity measurement and in situ titration process. f In situ IR spectra of model catalysts upon the introduction of EB as a function of the reaction time. g ODH rate as a function of EB (circle) and O2 (triangle) partial pressure. h ODH rates with C8H10(EB)/O2 (empty triangle) and C8D10(deuterated EB)/O2 (empty circle) on model catalysts. Reprinted with permission from Ref. [73]. Copyright 2017 American Chemical Society |
4.2 Liquid-Phase Oxidation and Reduction Reactions
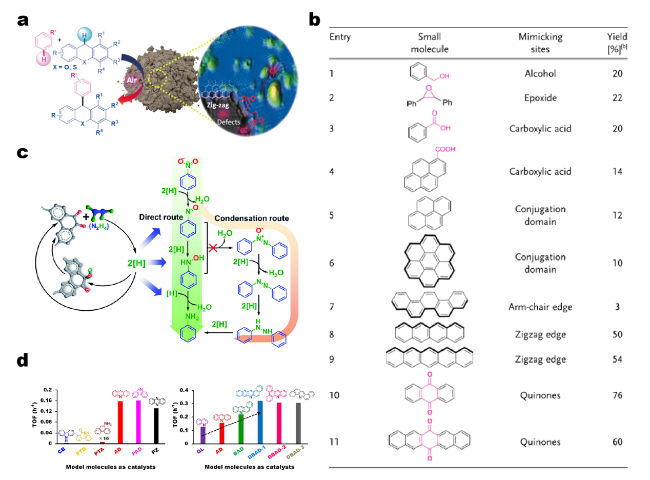
Fig. 4 a Cross-dehydrogenative coupling of xanthene (or thioxanthene) and arenes catalyzed by carbon materials. Mechanistic studies suggest that, compared to oxygenated groups, zigzag edge sites play a greater role in carbocatalysis than previously supposed. b Performance evaluation of small molecules with various O species and edge configurations aiming to reveal the possible active sites. Reprinted with permission from Ref. [53]. Copyright 2018 Wiley-VCH. c Schematic drawings of model molecules with exclusive C=O group catalyzed nitro compounds reduction process. Reprinted with permission from Ref. [76]. Copyright 2016 Royal Society of Chemistry. d TOF values of model molecules with various N species (e.g., pyrrole, pyridine, lactam), gradually extended π-conjugated structure, and different structural configurations used as catalysts toward aerobic alcohol oxidation. Reprinted with permission from Ref. [77]. Copyright 2019 American Chemical Society |
5 Model Catalysts in Electrocatalytic Reactions
5.1 ORR
5.1.1 Four-Electron Process
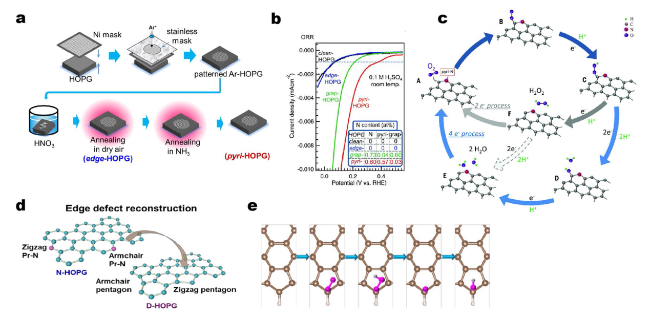
Fig. 5 a Schematic illustration for the preparation of edge-HOPG and pyri-HOPG model catalysts. b Performance evaluation of model catalysts toward acidic ORR. c Schematic reaction pathway for ORR on N-doped carbon materials. Reprinted with permission from Ref. [78]. Copyright 2016 American Association for the Advancement of Science. d Illustration of edge defect reconstruction aiming to remove original N species at elevated temperature. e The ORR pathway on pentagon edge configuration under acidic condition. Reprinted with permission from Ref. [57]. Copyright 2019 Nature Publishing Group |
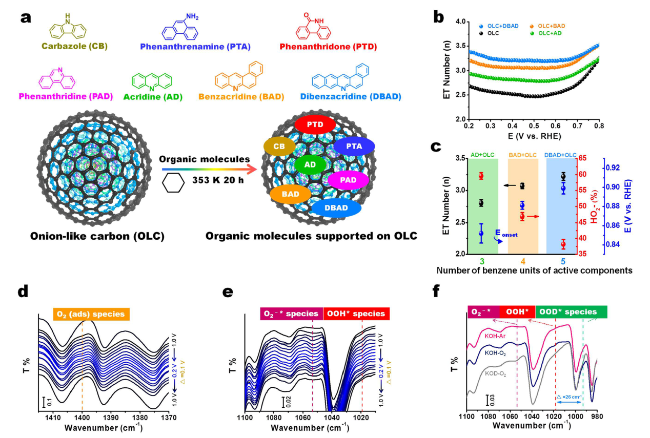
Fig. 6 a Structures of different model molecules with isolated N configurations (including pyrrolic, amine, lactam and pyridinic N) and the preparation of model molecules-supported on onion-like carbon (OLC) catalytic system using a solvothermal method. b Electron transfer (ET) numbers of three supported catalysts with isolated pyridinic N species based on rotating ring-disk electrode measurements. c The relationships between the number of benzene units of model molecules with single pyridinic N species and ET number, HO2− or onset potential (Eonset). d-f In situ ATR-IR spectra coupling with isotopic labeling experiments for identifying and monitoring dynamic evolution of the involved intermediate oxygen species including adsorbed O2, O2−* and OOH* species. Reprinted with permission from Ref. [79]. Copyright 2021 Wiley-VCH |
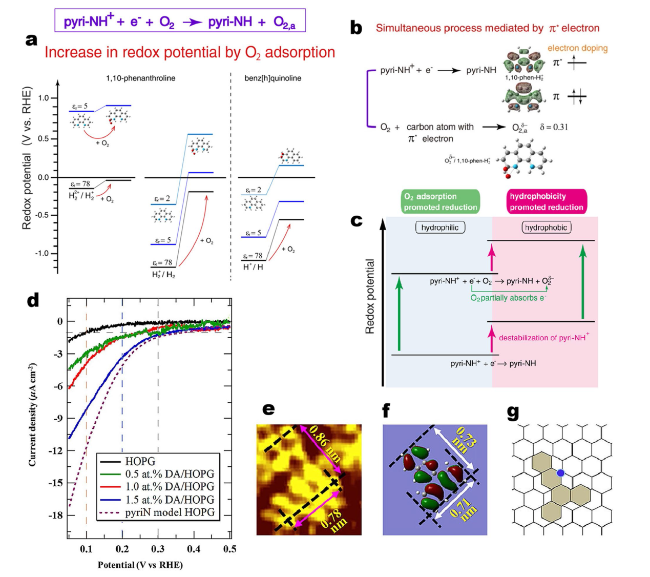
Fig. 7 a Calculated redox potentials of pyri-NH+/pyri-NH with and without O2 adsorption for 1,10-phenanthroline and benz[h]quinoline in water at different relative permittivities. b π* electron-mediated simultaneous reduction, where electron is supplied into π*orbital of 1,10-phenanthroline, which was used for O2 energy gain due to the adsorption energy of O2. c Promotional effects of O2 conditions on the reduction of pyri-NH+. Reprinted with permission from Ref. [80]. Copyright 2021 Wiley-VCH. d ORR results for various concentrations of DA. The ORR results for pristine HOPG and the pyridinic-nitrogen HOPG model catalyst (pyri N model HOPG) are also shown. Electrolyte conditions: 0.1 M H2SO4, room temperature. e Magnified STM image of the DA molecule on HOPG. f HOMO of isolated DA. g DA molecule in the model structure in HOPG. Reprinted with permission from Ref [81] |
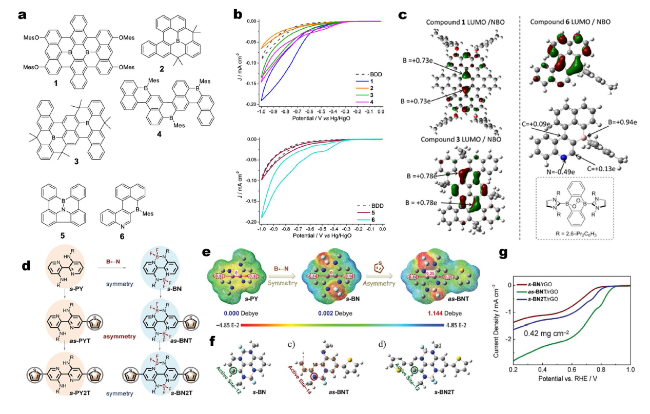
Fig. 8 a Well-defined doped PAHs used herein and the comparison compounds quinone A and perylene (compound B). Mes = mesityl, a boron “protecting group”. b CVs of 1 − 4 and 5 − 6 on a BDD electrode in O2-saturated 0.1 M KOH (aq) at a scan rate of 50 mV s−1. c LUMO (at isosurface value = 0.04) and select NBO charges for compounds 1, 3, and 6. The inset shows the product from the reaction of a NHC-stabilized 9,10-dibora-anthrene with O2 (NHC = N-heterocyclic carbene). Reprinted with permission from Ref. [82]. Copyright 2019 American Chemical Society. d The chemical structures of symmetric (s-PY, s-PY2T, s-BN, and s-BN2T) and asymmetric (as-PYT and as-BNT) organic molecular catalysts. e The electrostatic potentials and dipole moments of s-PY, s-BN, and as-BNT. f The optimized molecular structures of s-BN, as-BNT and s-BN2T models. g LSV curves of s-BN/rGO, as-BNT/rGO, and s-BN2T/rGO. Reprinted with permission from Ref. [83]. Copyright 2022 Wiley-VCH |
5.1.2 Two-Electron Process
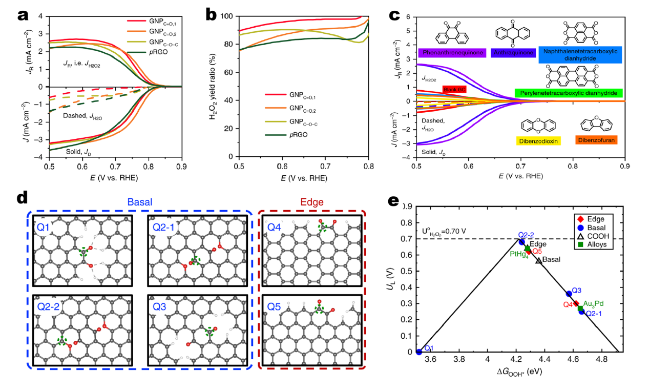
Fig. 9 The performance characterization and mechanism study of electrosynthesis H2O2 on O-enriched graphitic nanoplatelets (GNP). a, b The activity and yield of H2O2, respectively. c The electrosynthesis H2O2 performance of model molecules with different O groups. d Theoretical structures of different oxygenated groups at basal and edge sites. e Theoretical H2O2 activity volcano plot. Horizontal dashed line corresponds to the thermodynamic equilibrium potential for H2O2 electrosynthesis (U0 = 0.70 V). Reprinted with permission from Ref. [87]. Copyright 2020 Springer Nature |
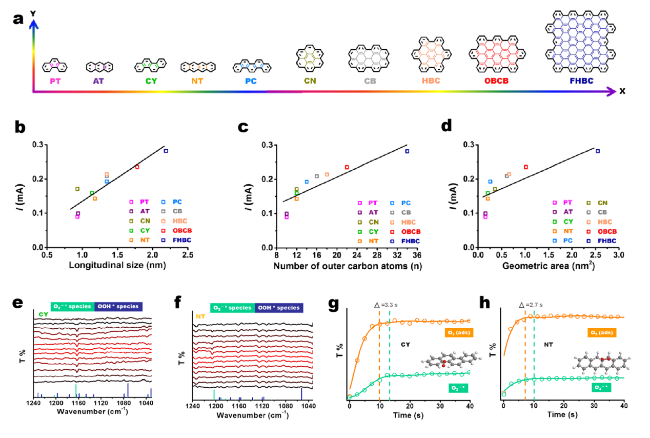
Fig. 10 Structure/size/area-function relationships and electrochemical performance of PAH catalysts for electrosynthesis H2O2. a Trends of gradient structures of PAH catalysts. b-d The correlations among the longitudinal sizes, the number of exposed outer carbon atoms and the geometric areas of PAHs and the disk current at 0.6 VRHE. e, f In situ ATR-IR spectra for monitoring dynamic evolution of the involved intermediate oxygen species on CY and NT catalysts, respectively. The vertical solid lines represent the theoretical vibration band positions of O2−* (cyan) and OOH* (dark blue) and their relative intensities. g, h Time-resolved in situ ATR spectra over CY and NT, respectively. The data were shown as dots and the fitted lines were also presented. The peak positions were determined by the maximum intensity of O2 (ads) and O2−* bands. Reprinted with permission from Ref. [88]. Copyright 2022 Elsevier. (Color figure online) |
5.2 OER
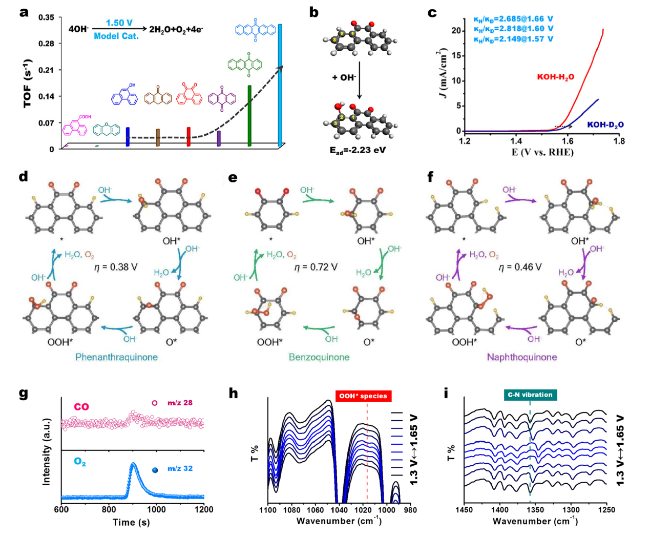
Fig. 11 a Performance assessment of various model molecules with different O species (including quinone, hydroxy, ether, and carboxyl groups), edge configurations, and flexibly extended conjugated structure. b Adsorption energy and structure of OH− species on model molecule having C=O group. Color code: carbon is gray, hydrogen is white, and oxygen is red. c KIE values calculated by current density ratio 0.1 M KOH dissolved in H2O and D2O (99.9%) solution. Reprinted with permission from Ref. [89]. Copyright 2018 American Chemical Society. d-f Consistency of theoretical and experimental activity and stability on phenanthraquinone, benzoquinone, and naphthoquinone, respectively. The gray, red, and yellow balls represent C, O, and H atoms, respectively. Reprinted with permission from Ref. [62]. Copyright 2018 American Chemical Society. g Mass spectra profiles of the produced CO (m/z 28) and O2 (m/z 32) with model molecules with isolated pyridinic N species. h-i In situ ATR-IR spectra for monitoring dynamic evolution of the involved intermediate *OOH and identifying the active sites on model molecules with isolated pyridinic N species. Reprinted with permission from Ref. [79]. Copyright 2021 Wiley-VCH. (Color figure online) |
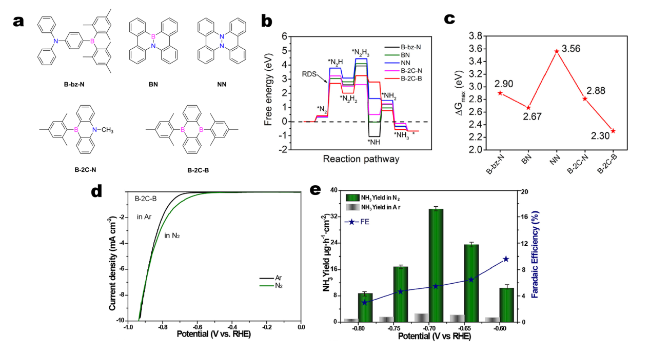
5.3 NRR
Fig. 12 a Molecular structures of five polycyclic aromatic hydrocarbons: B-bz-N, BN, NN, B-2C-N and B-2C-B. b Full free energy diagrams of all these five PAHs. c The maximal Gibbs free energy changes. d LSV curves of B-2C-B in Ar (black curve) and N2 saturated electrolytes (green curve). e The NH3 production rates (left y-axis) and FE NH3 (right y-axis) of B-2C-B in Ar (grey column) or N2 saturating (green column) electrolytes at various applied potentials between − 0.6 and − 0.8 V. The error bars represent the average of three independent measurements. Reprinted with permission from Ref. [90]. Copyright 2020 Elsevier. (Color figure online) |