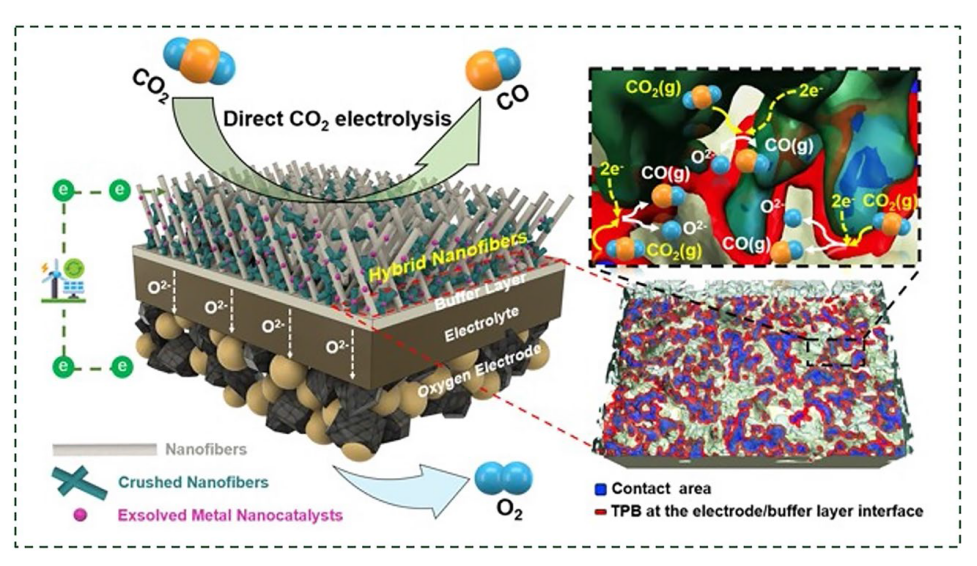
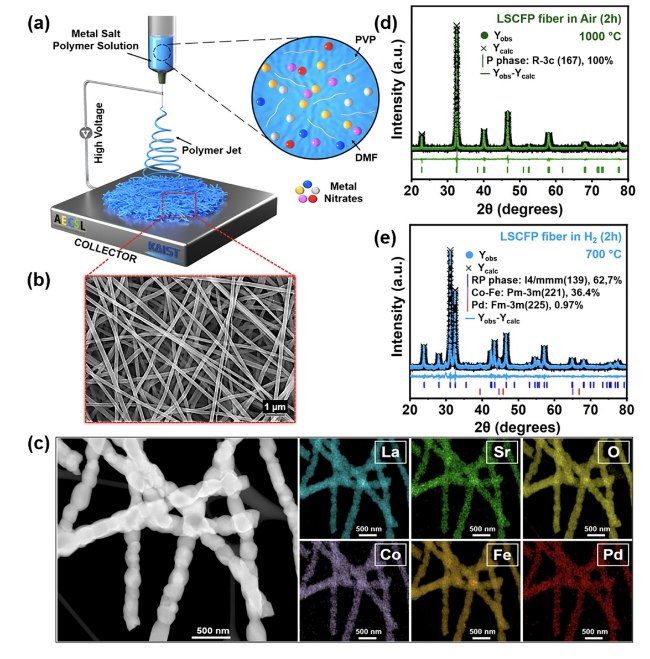
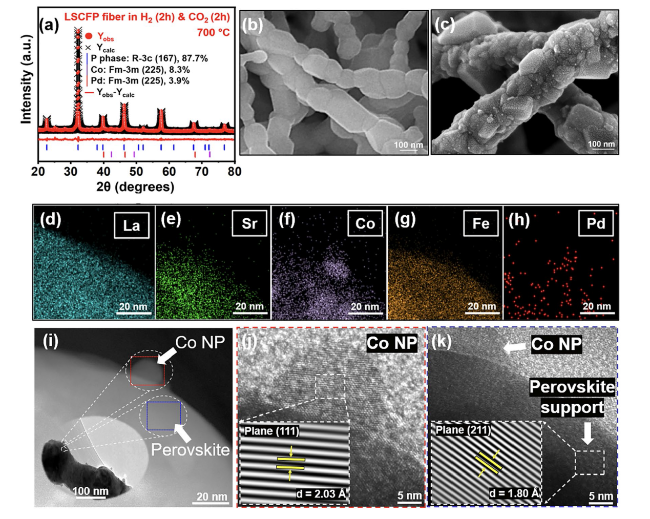
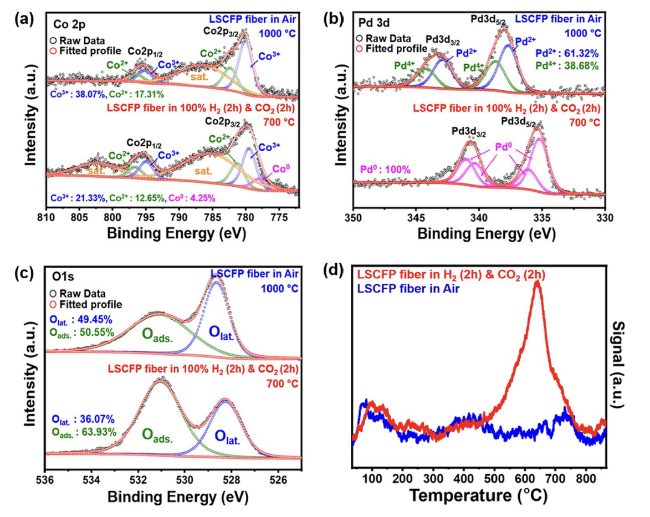
The performance of H-LSCFP and F-LSCFP fuel electrodes in CO
2 electrolysis was compared using LSGM electrolyte-supported single cells with identical LSCF-GDC oxygen electrodes. Prior to CO
2 measurements, both fuel electrodes were treated with 100% H
2 of 100 sccm at 700 °C for 2 h to promote the exsolution of metallic nanoparticles.
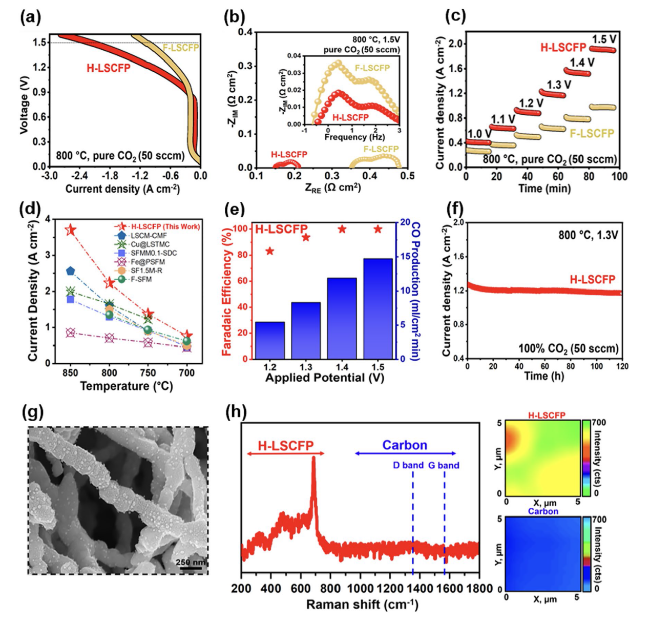
Figure 5a shows the current density of the SOEC with the H-LSCFP and F-LSCFP fuel electrodes measured in 100% CO
2 of 50 sccm at 800 °C with an applied potential scanned from open-circuit voltage (
Voc) to 1.6 V. The H-LSCFP cell exhibited a maximum current density of 2.2 A cm
−2 at 1.5 V, approximately twice that of the F-LSCFP cell (1.09 A cm
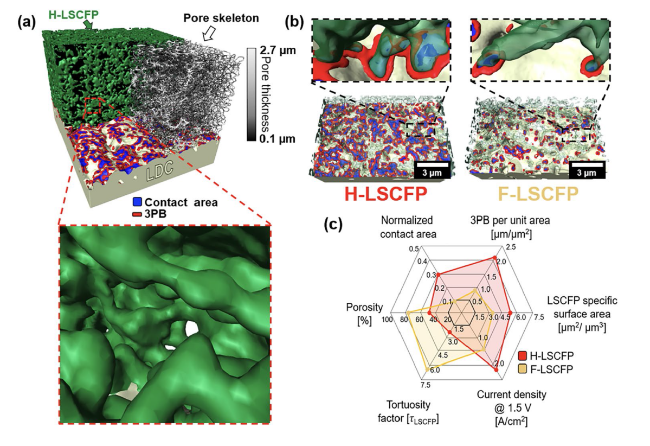
−2). To demonstrate the broader applicability of the H-LSCFP electrode, solid oxide cells (SOCs) with H-LSCFP and F-LSCFP fuel electrodes were tested in the fuel cell (FC) mode within the temperature range of 650-800 °C, where the H-LSCFP outperformed the F-LSCFP cell across all tested temperatures (Fig. S13). The MPD values are summarized in Table S2. This difference in performance can be attributed to the microstructure differences between H-LSCFP and F-LSCFP fuel electrodes, as the rest of the cell configuration was identical.
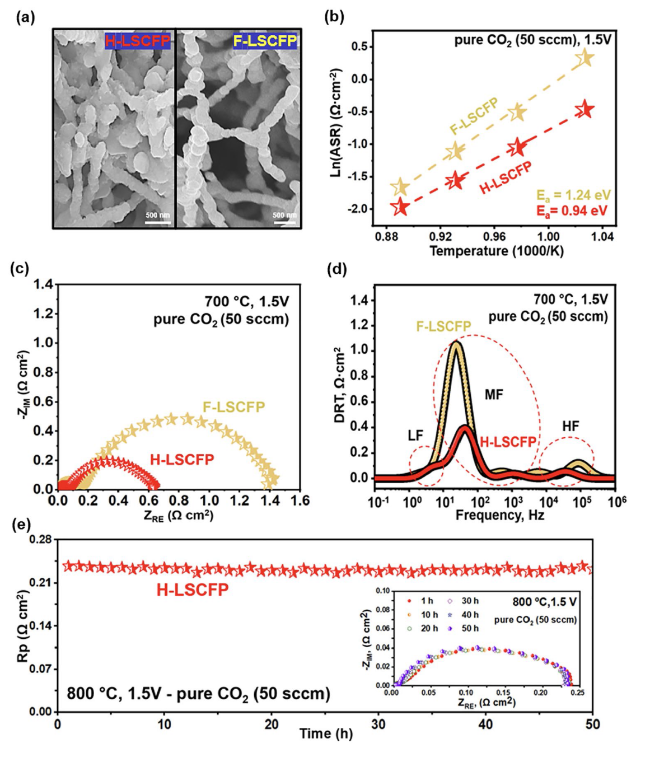
Figure 5b displays the corresponding Nyquist and Bode plots of EIS data of both cells at 1.5 V. In the Nyquist plots, the area specific ohmic (ASR
ohm) and electrode (ASR
elec) resistances were evaluated from the high-frequency intercept and the difference between the high and low frequency intercepts on the real axis, respectively [
33,
57]. The H-LSCFP cell showed roughly 52% lower ASR
elec (0.061 Ω cm
2) than that of F-LSCFP (0.127 Ω cm
2). Moreover, the ASR
ohm of the H-LSCFP cell (0.149 Ω cm
2) was approximately 2.4 times lower than that of the F-LSCFP (0.352 Ω cm
2). Considering the identical thickness of the LSGM electrolyte, the difference in the ASR
ohm can be attributed to disparities in the contact adhesion between the H-LSCFP and F-LSCFP fuel electrodes at the LSGM electrolyte interface, as confirmed by the cross-sectional image of both electrodes (Fig. S14). The impedance responses of both cells were further compared using Bode plots shown in the inset of
Fig. 5b. Both cells exhibited a dominant large semicircular arc in the low-frequency region related to the CO
2 surface adsorption/desorption processes and smaller semicircular arc in the high-frequency region associated with the oxygen ion transfer process at the electrode/electrolyte interface [
1]. The integral area of the SOEC with the H-LSCFP fuel electrode was significantly smaller than that of the SOEC with the F-LSCFP, indicating improved CO
2RR kinetics and better contact adhesion of the H-LSCFP electrode on the electrolyte surface.
Figure 5c shows the current density values obtained from a potentiostatic stability test of both SOECs in 100% CO
2 of 50 sccm at 800 °C and a voltage range of 1.0-1.5 V. The H-LSCFP cell yielded significantly higher current density values than the F-LSCFP cell at all tested potentials. Specifically, the current density of the H-LSCFP cell (1.18 A cm
−2) at 1.2 V was ~ 87% higher than that of the F-LSCFP cell (0.63 A cm
−2), with the remaining results summarized in Table S3. Additional short-term potentiostatic stability test was conducted at 750 °C in 100% CO
2 (50 sccm), within a potential range of 1.0-1.4 V, to verify the effect of the exsolution treatment on CO
2 electrolysis performance of the H-LSCFP cell. Following exsolution, the H-LSCFP cell consistently demonstrated significantly higher current density values across all tested potentials (Fig. S15) when compared to the H-LSCFP cell without exsolution, as summarized in Table S4.