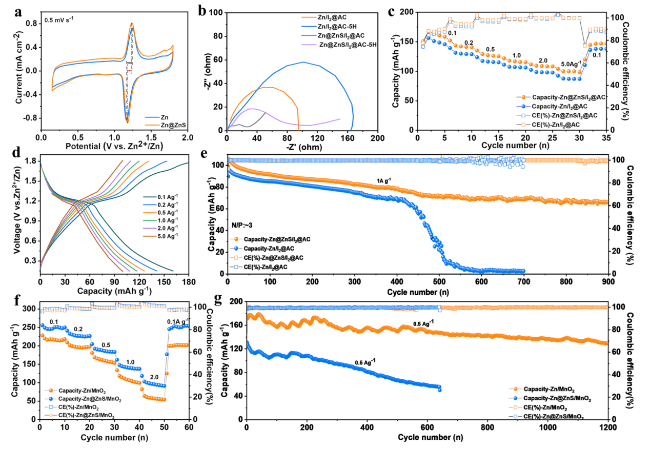
To test the practical application of the Zn@ZnS anode in ZIBs, the full cells with I
2/AC and MnO
2 as cathode materials were assembled. Figure S30a shows the XRD patterns of I
2 crystal (JCPDS#72-0012), active carbon (AC), and a composite of I
2/AC. The XRD peaks of the I
2/AC composite generally match with the XRD peaks of active carbon (AC), both exhibit two broad peaks at around 22° and 43°, corresponding to the (002) and (100) diffraction patterns of amorphous carbon materials. However, no evident I
2 peaks can be detected in the I
2/AC, indicating the formation of non-crystalline iodine [
54]. Figure S30b indicates the α-MnO
2 matches with the PDF card of JCPDS#72-1982.
Figure 5 shows the electrochemical performance of Zn||I
2@AC supercapattery and Zn||MnO
2 battery. The CV curves in
Fig. 5a demonstrate that the zinc-iodine supercapattery with Zn@ZnS anode reached a higher current density and smaller redox overpotential due to the faster desolvation process and Zn
2+ migration rate. This is consistent with the Nyquist plots of the cell at a different resting time shown in
Fig. 5b. With the ZnS protection layer, the cells all displayed lower charge transfer resistance at the beginning and after 5 h’ resting, further proving the suppressed side reactions and enhanced interaction of Zn@ZnS anode with Zn
2+ ions. The galvanostatic charging/discharging process was further conducted to elucidate the impact of the ZnS SEI layer in a full cell. The rate performance in
Fig. 5c shows that the full cell with Zn@ZnS anode presents better rate performance and higher specific capacity, and a higher capacity of 160 mAh g
−1 is reached at 0.1 A g
−1. The charge and discharge curves of Zn@ZnS||I
2@AC full cell in
Fig. 5d display a wide working window of 0.2-1.8 V and evident working plateaus at ~ 1.2 V, corresponding to the redox potential of I
2/I
− [
55]. Figure S31 shows the long-term cycling stability at 10 A g
−1 of the cells with and without the ZnS layer. As reported, the generation of soluble polyiodide intermediates during the cycling process tends to corrode the zinc anode due to the “shuttle effect” [
56]. Herein, to study the influence of the ZnS SEI layer on the inhibition of “shuttle effects”, the cell was subjected to 2,000 cycles followed by a day rest before restarting. Figure S31 shows that during the whole cycling process, the cell with Zn@ZnS anode exhibits higher capacity than the cell using a pure zinc anode. And a high capacity of 115 mAh g
−1 is obtained in the first 2,000 cycles. After 1 day of rest, the cell can still stably cycle for more than 8,000 cycles, with a high capacity retention of 82%. By contrast, the cell with pure zinc anode exhibits a faster capacity decay after one day’s resting, and the capacity drops to 60 mAh
−1 after 10,000 cycles. To meet harsh operating conditions, the long cycling performance of full cells with a low N/P ratio of 3 (ultrathin zinc foil of 20 μm) was further studied at a current density of 1A g
−1 (
Fig. 5e). With the ZnS protection layer, the full cell sustains stable cycling for more than 900 cycles, with a capacity retention of 63%. In comparison, the cell with pure zinc anode manifests lower specific capacity than the Zn@ZnS anode in the first 400 cycles and then suffers drastic capacity decay to zero. These results suggest that the side reactions and zinc dendrite are greatly suppressed with ZnS coating.