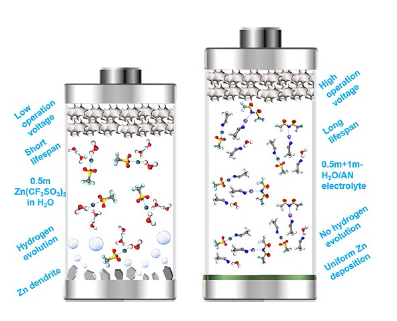
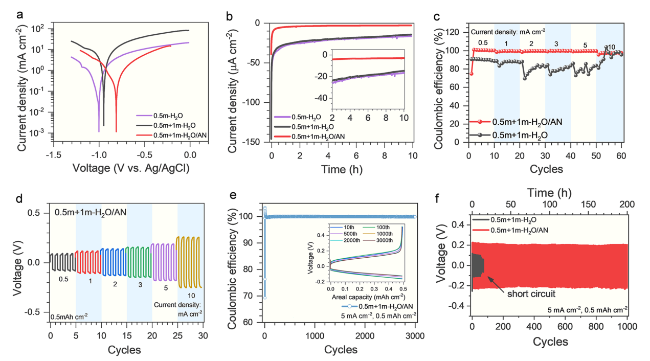
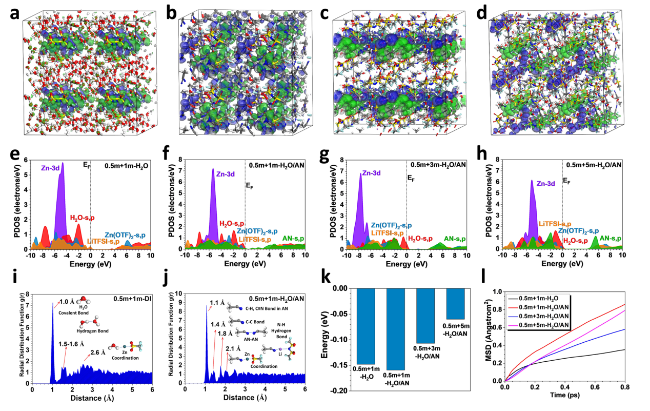
The Coulombic efficiency of Zn plating/stripping was further tested based on a Zn-Cu coin cell with the H
2O/AN electrolytes. As shown in Fig. S6, the cells with H
2O/AN electrolytes show stable and significantly higher Coulombic efficiency (average > 99%) after 100 cycles as expected, while the cells with aqueous electrolyte (0.5 m + 1 m-H
2O) demonstrate gradually decreasing and fluctuating Coulombic efficiency (from 90 to 70% after 50 cycles) under the same condition. Moreover, for the H
2O/AN electrolytes with different concentrations of LiTFSI (1-5 m), the initial Coulombic efficiency decreases with the increase of LiTFSI concentration, and the average Coulombic efficiency of Zn plating/stripping is the highest in 0.5 m + 1 m-H
2O/AN electrolyte. From the SEM images of Zn foils in the Zn-Cu cells after specific cycles (Fig. S7), a great number of macro-pores are observed on the Zn foils after the 10th, 20th, and 50th cycles in the aqueous electrolytes (0.5 m-H
2O and 0.5 m + 1 m-H
2O), which indicates an uneven Zn plating/stripping behavior and severe Zn corrosion during cycling. Nevertheless, a compact and uniform surface is revealed on the Zn foils cycled in 0.5 m + 1 m-H
2O/AN electrolyte, confirming an excellent Zn plating/stripping reversibility in 0.5 m + 1 m-H
2O/AN electrolyte. At enlarged current densities up to 10 mA cm
−2, a high Coulombic efficiency (97.3% on average) is still maintained in 0.5 m + 1 m-H
2O/AN electrolyte, in contrast to its rapid decrease and fluctuation in the aqueous electrolyte (0.5 m + 1 m-H
2O), as shown in
Figs. 1c and S8. However, for the H
2O/AN electrolytes with further increased LiTFSI concentrations (0.5 m + 3 m-H
2O/AN and 0.5 m + 5 m-H
2O/AN), a decreased Coulombic efficiency and increased voltage hysteresis are observed at higher current densities (e.g., > 5 mA cm
−2, Fig. S9), indicating that an excessive concentration of LiTFSI is unfavorable to the reversibility of Zn plating/stripping at high current densities. Correspondingly, the symmetric Zn-Zn cells with 0.5 m + 1 m-H
2O/AN electrolyte only show slightly increased voltage hysteresis with enlarged current densities (
Fig. 1d), while a dramatically increased voltage hysteresis followed by short-circuit is observed for 0.5 m + 3 m-H
2O/AN and 0.5 m + 5 m-H
2O/AN electrolytes (Fig. S10). After cycling at enlarged current densities (e.g., 5 mA cm
−2), the Zn foil maintains uniform and compact morphology in 0.5 m + 1 m-H
2O/AN electrolyte, in accordance with a low voltage hysteresis even at high current densities. In contrast, the Zn foils cycled in 0.5 m + 3 m-H
2O/AN and 0.5 m + 5 m-H
2O/AN electrolytes show a rough surface, revealing an uneven plating behavior at increased current densities (Fig. S11). These observations demonstrate that excessive LiTFSI in electrolytes is unfavorable to the transportation of Zn
2+ ions and deteriorates the rate performance of Zn metal anode, which is in agreement with the larger charge transfer resistances in the Zn-Zn symmetric cells with increased LiTFSI concentrations (Fig. S12). The long-term cyclic stability of the Zn metal anode in the 0.5 m + 1 m-H
2O/AN electrolyte was further verified by the Zn-Cu and Zn-Zn cells. A high average Coulombic efficiency of 97.3% is achieved in the 0.5 m + 1 m-H
2O/AN electrolyte for more than 3,000 cycles (
Fig. 1e), and no obvious changes are observed in the charge-discharge curves of specific cycles (the inset of
Fig. 1e). Correspondingly, the Zn-Zn cells with the 0.5 m + 1 m-H
2O/AN electrolyte also show stable voltage hysteresis with prolonged cycles, while a short-circuit quickly occurs in the Zn-Zn cells with the reference electrolyte (0.5 m + 1 m-H
2O), as shown in
Fig. 1f. These results further verify the superior stability of the Zn metal anode in the 0.5 m + 1 m-H
2O/AN electrolyte.