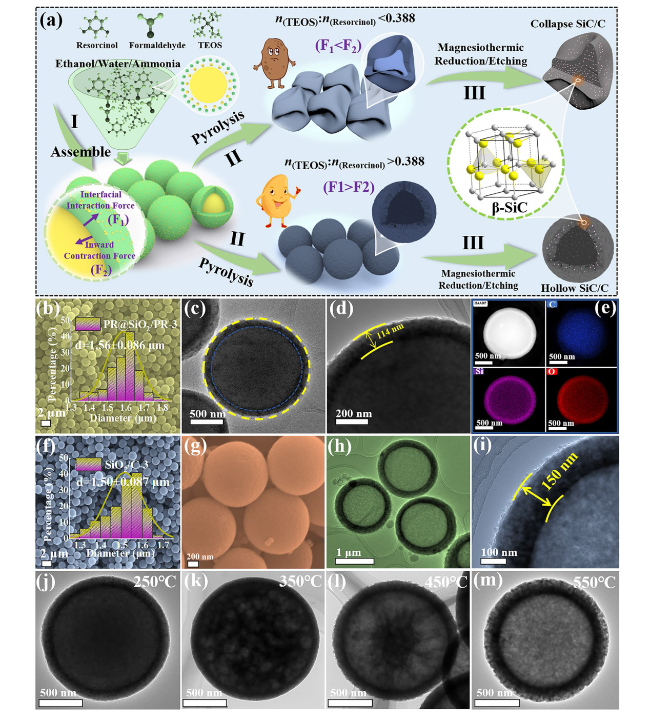
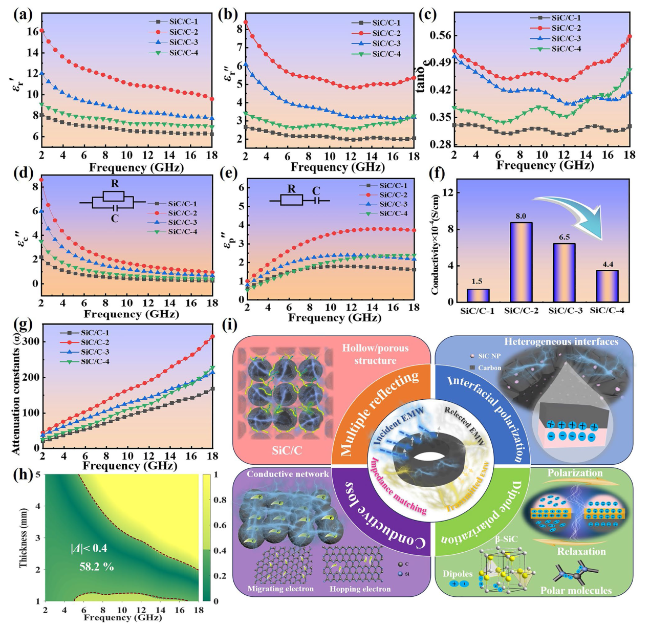
Figure 1a illustrates the step-by-step preparation procedures of SiC/C composites with a heterogeneous interface anti-contraction strategy. First, resorcinol and formaldehyde are sequentially dispersed in alkaline aqueous ethanol solution to generate phenolic resin (PR) microspheres. After the introduction of TEOS, silica oligomers from the hydrolysis will be co-assembled with the residual PR oligomers to produce core-shell PR@SiO
2/PR microspheres. Scanning electron microscopy (SEM) images show that all the resultant PR@SiO
2/PR microspheres have regular spherical morphology, good dispersion, and smooth surface, and their average diameters range from 1.27 ± 0.079 to 1.61 ± 0.086 μm with increasing the dosage of TEOS (
Figs. 1b and S1). Transmission electron microscopy (TEM) images identify the core-shell configuration in PR@SiO
2/PR-3, as well as the thickness of the external shells at about 114 nm (
Fig. 1c, d). EDS mapping results further firmly support that a desirable core-shell configuration has been successfully created in these microspheres, because O and Si elements are mostly distributed in an outer ring, whose size is obviously larger than the distribution region of C element (
Fig. 1e), and meanwhile, a fraction of C atoms can be also observed in the region of O and Si elements, implying the presence of PR in theshells. It is found that the dosage of TEOS plays an important role in maintaining the spherical morphology of these composites during high-temperature pyrolysis. For example, SiO
2/C-1 derived from PR@SiO
2/PR-1 (the TEOS/resorcinol molar ratio of 0.388) is composed of numerous wizened particles and almost completely loses its original morphology (Fig. S2a, b), and in contrast, the other intermediate composites with higher TEOS dosages (the molar ratio of TEOS/resorcinol is more than 0.582), i.e., SiO
2/C-2, SiO
2/C-3, and SiO
2/C-4, all inherit the spherical morphology very well except a slight shrinkage in the average diameter (
Figs. 1f, g and S2c, d). Of note is that some impurities are detected in SiO
2/C-4, suggesting that the dosage of TEOS in this case may be a little excessive. TEM characterization is further carried out by taking SiO
2/C-3 as a representative sample. Compared with its precursor (PR@SiO
2/PR-3), SiO
2/C-3 not only displays an unexpected hollow structure, but also gives a thicker shell at about 150 nm (
Fig. 1h, i). According to previous studies, when some pure organic precursors, e.g., polymers and MOFs, are pyrolyzed under high-temperature inert atmosphere, there will be a dramatic inward shrinkage behavior, resulting in the formation of carbon-based products with much smaller size [
25,
26]. However, if a stable external shell is pre-constructed on the surface of organic precursors, the interaction between the shell and the precursor will induce preferential carbonization at the interface and produce an interfacial interaction force (F1) to resist the inward contraction force (F2), and more importantly, such an interfacial interaction will also promote the inside-out diffusion of organic precursors and finally create an interior cavity [
27]. The microstructure evolution from PR@SiO
2/PR-3 to SiO
2/C-3 at different pyrolysis temperature indeed records the process of gradual internal cavitation (
Figs. 1j-m and S3). When the pyrolysis temperature is 250 °C, the intrinsic microstructure remains consistent with the precursors, indicating that the contraction process of the heterogeneous interface between the core and the shell has not been triggered yet. As the temperature continues to increase to 350 °C, it is observed that the heterogeneous interfaces boundaries between the core and the shell are not as dense as they are at first, and their interior regions have many apparent holes with different size. With a further increase of the temperature to 450 °C, the PR core undergoes continuous decomposition, resulting in the gradual aggregation of small pores into some larger ones. Upon reaching a temperature of 550 °C, the internal hollow structure is completely formed, and the spherical morphology can still be maintained without obvious collapse and fragmentation phenomenon. These results indicate that the formation of hollow SiO
2/C composites follows the mechanism of heterogeneous interface anti-contraction and also explains why the thickness of the external shells increases. The collapse of SiO
2/C-1 can be attributed to the fact that the relatively thin SiO
2 shells fail to provide enough interfacial interaction force (
Fig. 1a).