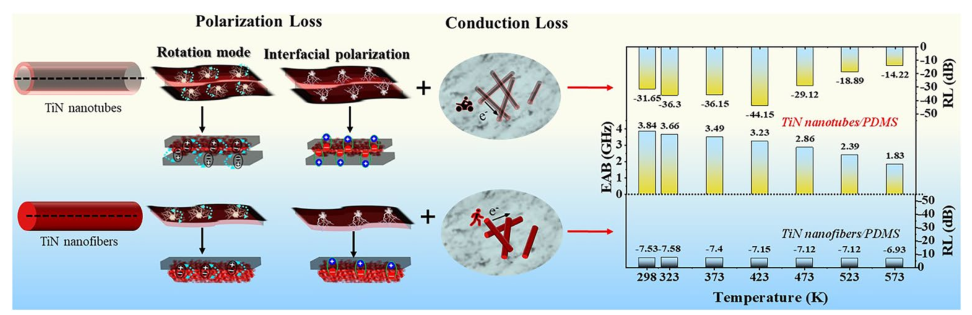
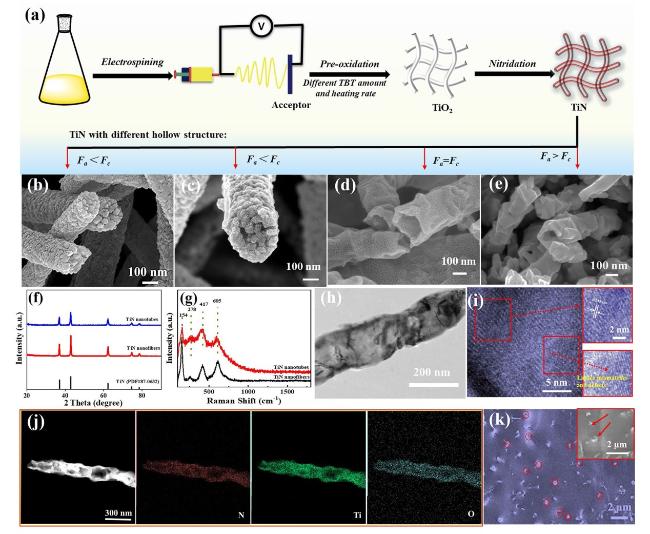
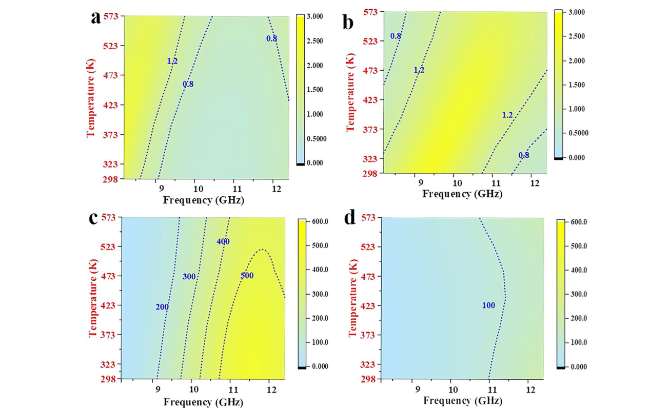
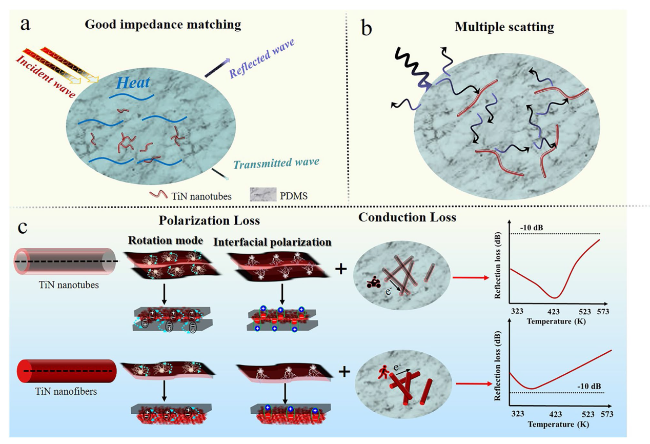
To obtain the controllable manipulation of interface engineering, TiN nanotubes are designed, as shown in
Fig. 1a. Beginning with the thought of creating and triggering more heterogeneous interfaces, TiN nanotubes are dexterously devised via changing the TBT amount and heating rate of pre-oxidation temperature. The TiN nanotubes are ultimately harvested by facile electrospinning and calcination methods with an aim of exchange reaction. Firstly, the uniform precursor nanofibers are prepared by an electrospinning method. Secondly, TiN nanotubes are created by the pre-oxidation and nitriding process, which are closely related with the decomposition of PVP, formation of TiN layer and diffusion of metal cations. Using 5.05 mL TBT and 2 °C min
−1 heating rate of pre-oxidation, the produced nanofibers are comprised of TiN nanoparticles, as shown in
Fig. 1b. With the decrease in TBT (3.8 mL) and invariability of heating rate of pre-oxidation (2 °C min
−1), the corresponding TiN nanofibers emerge a partial void (
Fig. 1c). When the heating rate is 1 °C min
−1, the product becomes nanotube structure with the ~ 25 nm thin wall (
Fig. 1d). Continue to reduce the heating rate (0.5 °C min
−1), TiN nanofibers become partial void (
Fig. 1e). The result reveals that the formation of nanotubes is predominantly influenced by the TBT amount and heating rate of pre-oxidation, which is closely related with Ostwald ripening procedure and kinetic diffusion, leading to the in situ formation of TiN layer and diffusion of metal cations [
31]. Reasonably, two opposing forces are acting simultaneously on the wall: contraction force (
Fc) and adhesive force (
Fa), which derives from the thermal degradation of organic species to facilitate the shrinkage of wall, and the rigid surface that restrains the inward shrinkage, respectively [
31,
32]. Spontaneously, through applying the suitable TBT amount and heating rate of pre-oxidation, scilicet
Fa =
Fc, the wall will be created due to the interaction and dynamic equilibrium between
Fc and
Fa [
31]. However, if TBT amount and heating rate of pre-oxidation is so much and fast, more TiN nanocrystals continuously diffuse outward and prefer to aggregate into larger particles, as shown in
Fig. 1b, c, the
Fc increases and the dynamic equilibrium is broken, thus the 1D nanofiber or/with partial void is generated [
33]. Contrariwise, when the heating rate of pre-oxidation is slow, the decomposition of PVP is low, leading to the less
Fc, which results in the nanofiber with partial void and small crystalline grain (
Fig. 1e). Therefore, by rational controlling the Ostwald ripening and kinetic diffusion procedure, both the growth of TiN nanocrystalline and the diffusion kinetics of Ti element could be regulated, which contribute to the formation of nanotube structure [
31].