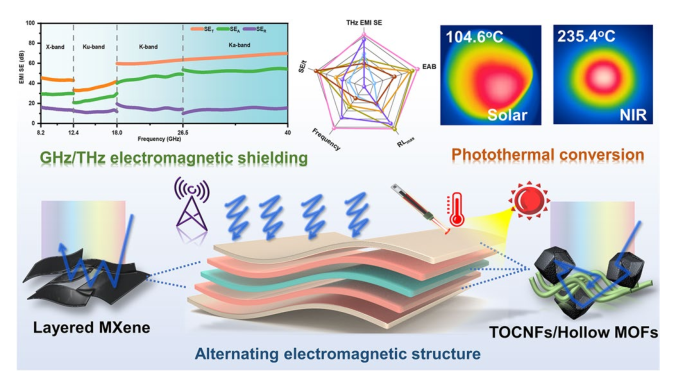
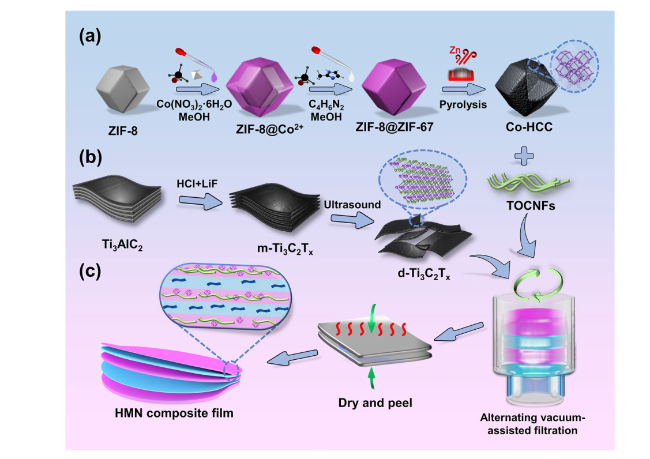
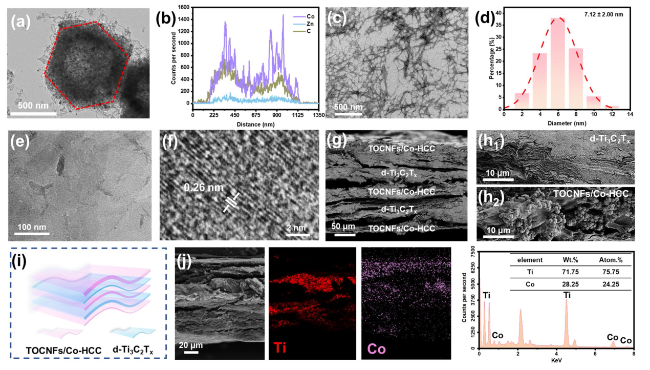
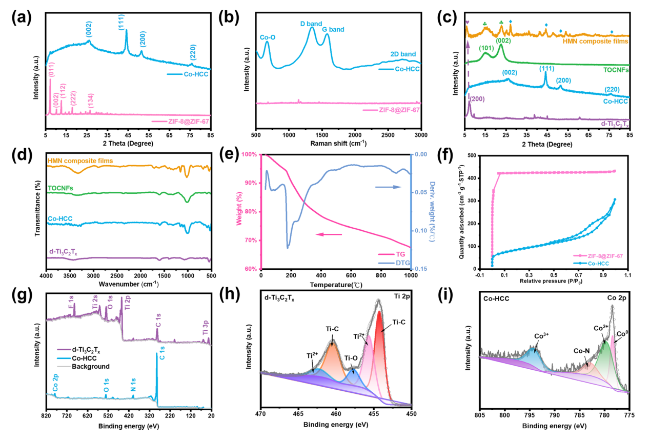
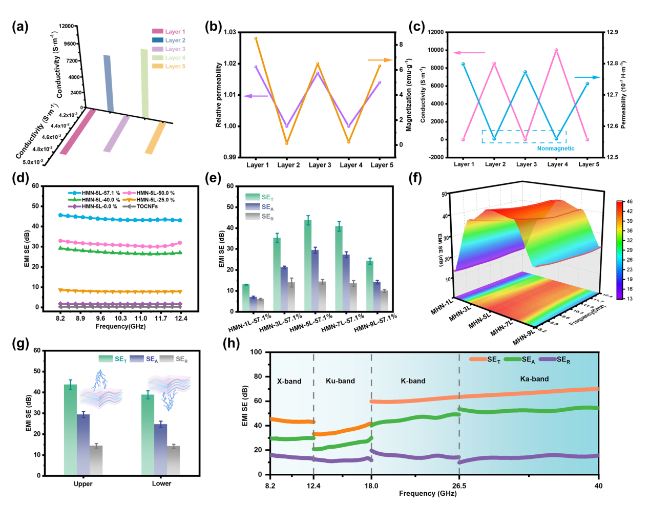
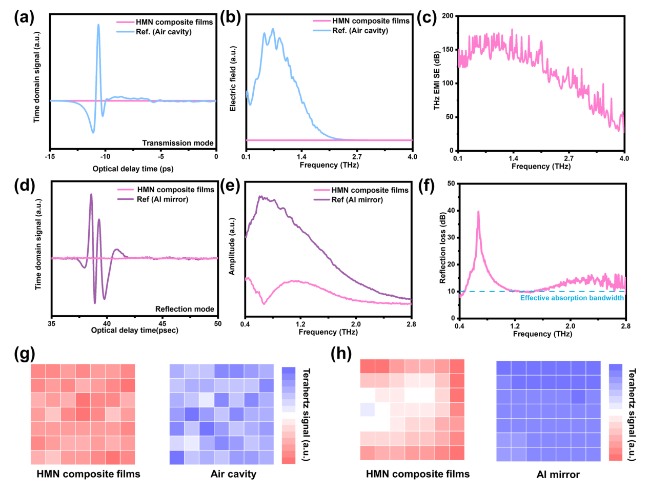
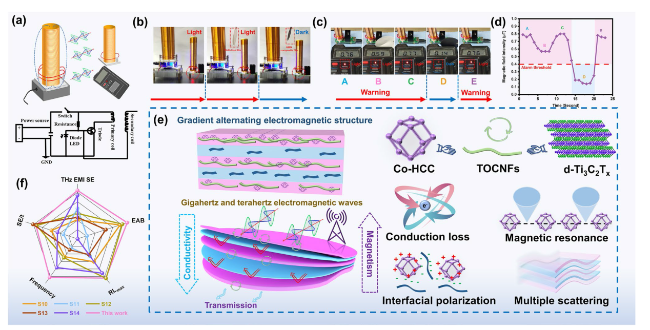
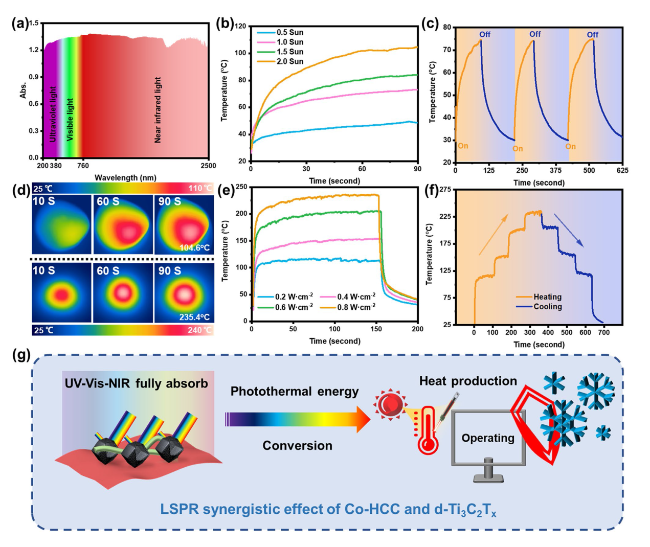
Besides, the above characteristic peaks are apparent in the HMN composite films, which prove the successful preparation of the composite materials. To further explore the components of HMN composite films, the FTIR spectrum is displayed in
Fig. 3d. The characteristic peaks of TOCNFs were observed at 1050 and 1600 cm
−1, attributing to the stretching vibration of the C-O-C group of the glucopyranose ring and the existence of the carbonyl [
37]. The Ti
3AlC
2 where no particular peak can be detected in the FTIR spectrum, depicts the purity of MAX. The d-Ti
3C
2T
x shows peaks at 580 and 1380 cm
−1 that correspond to -OH and C-F groups. Besides, all ZIF-67, ZIF-8, and ZIF-8@ZIF-67 present several characteristic peaks in the range of 500-2000 cm
−1 (Fig. S8). After carbonization at high temperature, the two new peaks remain at 560 and 650 cm
−1, corresponding to Co [
38]. These characteristic peaks were also observed in the FTIR spectrum of HMN composite films. TGA and DTG were used to analyze the composition of HMN composite films (
Fig. 3e). The decline curve of TG accords with cellulose. The high residual weight percentage is attributed to d-Ti
3C
2T
x derivative titanium dioxide at high temperature and Co-HCC [
39,
40]. The specific surface areas and N
2 absorption-desorption of ZIF-8@ZIF-67 and Co-HCC were researched (
Figs. 3f and S9a). N
2 absorption-desorption results show that saturation is achieved at low relative pressure for the ZIF-8@ZIF-67, demonstrating the microporous structure of the materials with the typical type I adsorption-desorption isotherms. The specific surface area of ZIF-8@ZIF-67 reached 1565.6 and 1862.9 m
2 g
−1 in the BET model and Langmuir model, respectively. By contrast, the isotherms of Co-HCC show a relatively low N
2 absorption-desorption capacity and specific surface area (297.7 and 920.7 m
2 g
−1 in the BET model and Langmuir model, respectively), probably caused by partial collapse of the frame. The typical type IV and the H3 hysteresis loop of Co-HCC exhibited the generation of hollow structure of materials [
41]. Moreover, the average pore size (1.71 nm) of ZIF-8@ZIF-67 was less than the average pore size (6.38 nm) of Co-HCC. The pore size and pore volume of Co-HCC increased and gradually deviated from the microporous structure, which proved the hollow structure of Co-HCC (Figs. S9b and S10). The XPS analysis was used to further investigate the chemical compositions of d-Ti
3C
2T
x and Co-HCC (
Fig. 3g-i). The XPS spectra show that the d-Ti
3C
2T
x and Co-HCC contain C, O, Ti, F, and C, O, N, Co elements, respectively. In the Ti 2
p spectrum, a series of characteristic peaks for Ti-C, Ti-O, and Ti
2+ were detected, thereby corroborating the existence of surface partial oxidation on the d-Ti
3C
2T
x [
42]. In the Co 2
p spectrum, a series of characteristic peaks for Co
0, Co
2+, and Co
3+ suggest the presence of metallic Co
0, cobalt oxide, and cobalt tetroxide in the Co-HCC [
40]. Besides, as shown in Fig. S11, the XPS fine spectrum of the N 1
s peak was deconvolved into four peaks, including oxidized-N, graphitic N, Co-N, and pyridinic N, respectively. In addition, the rich N element is helpful in improving the electromagnetic shielding performance of the materials [
43]. The XPS fine spectra of Zn 2
p indicated a doublet peak fitted at 1022.0 and 1045.1 eV attributed to Zn 2
p3/2 and Zn 2
p1/2. The binding energy difference between these observed Zn 2
p3/2 and Zn 2
p1/2 peaks about 23.1 eV, which is the same as the standard reference value of the ZnO (Fig. S12) [
44].