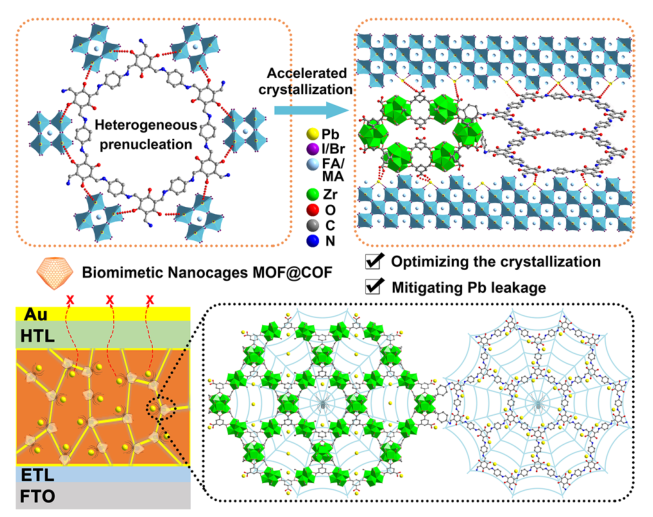
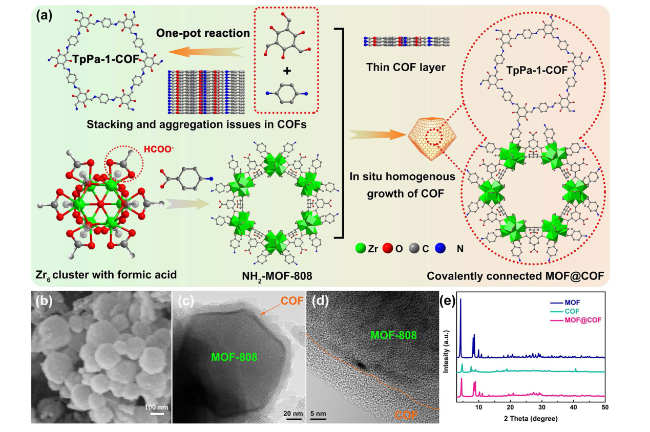
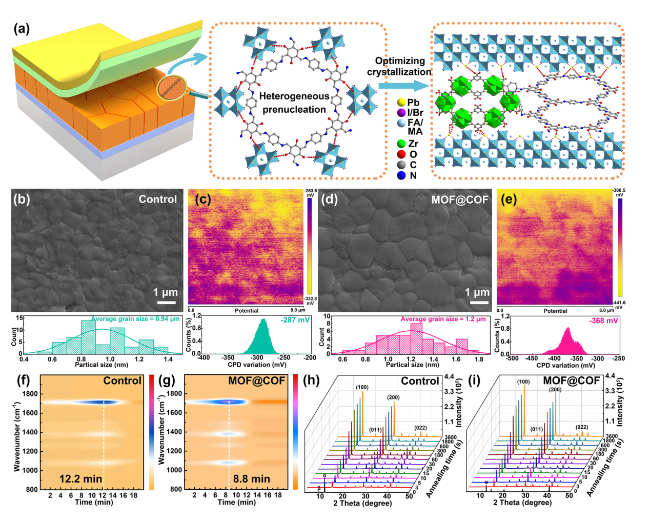
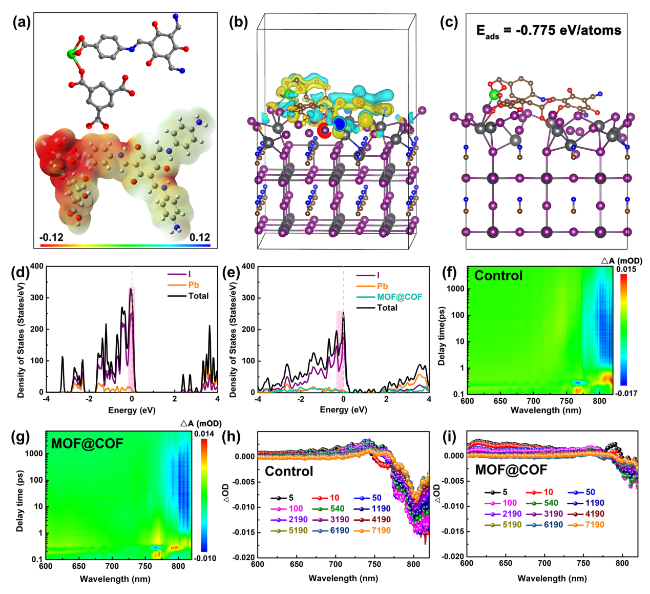
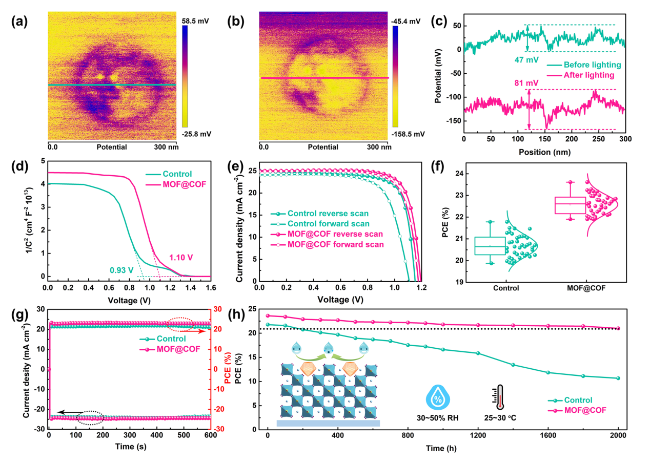
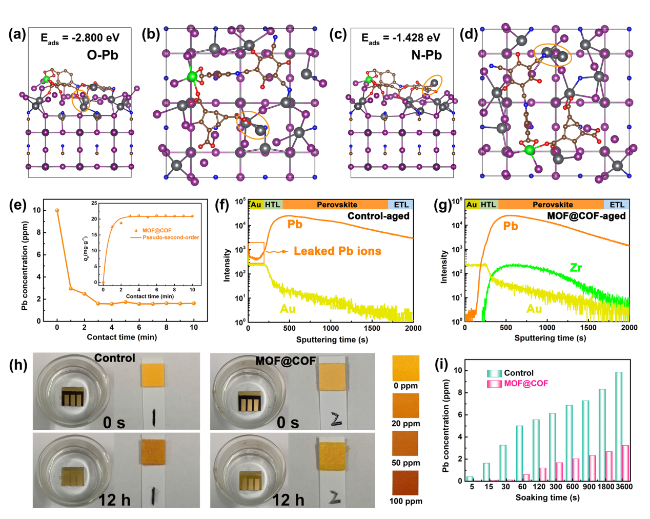
The inherently large surface area (Fig. S19) and porous structure endow MOFs and COFs with an excellent adsorption capacity for heavy metal ions, which prompted us to explore the in situ chemical fixation and adsorption of a MOF@COF in mitigating Pb leakage in PSCs [
53,
54,
55,
56,
57,
58]. DFT calculations were conducted to compare the interaction between the perovskite and N and O atoms in the MOF@COF, as shown in
Fig. 5a-d. The adsorption energy
Eads was calculated according to the following equation:
Eads =
EPero-MOF@COF-Pb-
EPero-MOF@COF-μPb, where
EPero-MOF@COF-Pb,
EPero-MOF@COF, and
μPb are the total energy of the adsorption configuration, the total energy of Pero-MOF@COF, and chemical potential of Pb, respectively. Accordingly, the
Eads values of N and O for leaking Pb
2+ ions were − 1.428 and − 2.800 eV, demonstrating that the active groups of C-N, -COO
−, and C=O exhibited a strong affinity for Pb
2+ ions. The results of the adsorption kinetics and isotherms showed that the maximum Pb adsorption capacity obtained from the pseudo-second-order model was 427.9 mg g
−1 (
Figs. 5e and S20) [
53]. These combined results indicate that the MOF@COF can effectively capture Pb
2+ to reduce the risk of Pb leakage. To further monitor the migration path of the leaked Pb
2+ ions, the depth profiles of Pb
2+ ions in the aged PSCs were investigated using time-of-flight secondary ion mass spectrometry (ToF-SIMS) [
54]. As illustrated in
Fig. 5f, g, a Pb
2+ signal appeared on the Au electrode, indicating that Pb ions easily migrated into the environment and seriously threatened the ecosystem. Interestingly, only a small trace Pb
2+ signal was observed on the Au electrode for the MOF@COF-functionalized PSCs, which demonstrated that the MOF@COF could effectively trap Pb ions through the synergistic effect of in situ chemical fixation and adsorption. Finally, the PSCs were immersed in water for different durations to investigate the amount of Pb that leaked from the degraded PSCs. As shown in
Fig. 5h, the Pb concentration in the contaminated water was quickly monitored by the color of the Pb
2+ testing paper for the control PSCs, which was much darker than that of the MOF@COF-functionalized PSCs after 1 h. Inductively coupled plasma optical emission spectroscopy (ICP-OES) was used to precisely determine the Pb concentration. As shown in
Fig. 5i, the target PSC exhibits a much lower Pb concentration than the control PSC, thereby reducing the risk of Pb leakage. Based on the above analysis, the MOF@COF played a crucial role in mitigating Pb leakage. The C-N, -COO
−, and C=O active groups in the MOF@COF could in situ chemically fix uncoordinated Pb
2+ to strengthen the internal stability of the perovskite film. However, even if the perovskite degrades, the biomimetic nanoparticles of the MOF@COF could also act as an adsorbent (like a spider web) to “trap” Pb
2+ ions through an in situ chemical adsorption method.