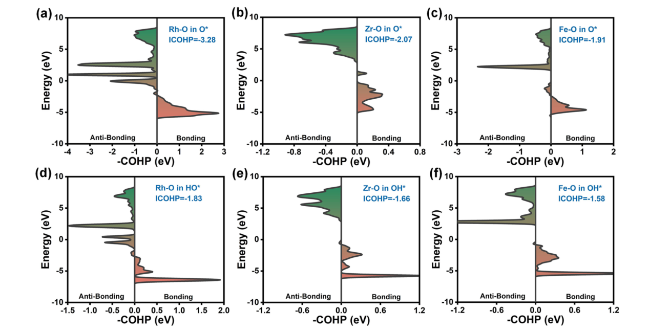
The
GO*-
GHO* is generally regarded as a crucial descriptor for evaluating OER activity since it quantifies the capacity of surface oxygen atoms to make and break bonds with hydrogen and oxygen atoms, which is prone to be the most thermodynamically challenging reaction steps for OER [
2,
43,
51]. To achieve more accurate predictions, we employed the microkinetic modeling as the function of
GO*-
GHO* and potential at various currents [
43,
52]. The current curves were obtained by numerically solving the rate of HOO formation considering the O
→ HOO transition state.
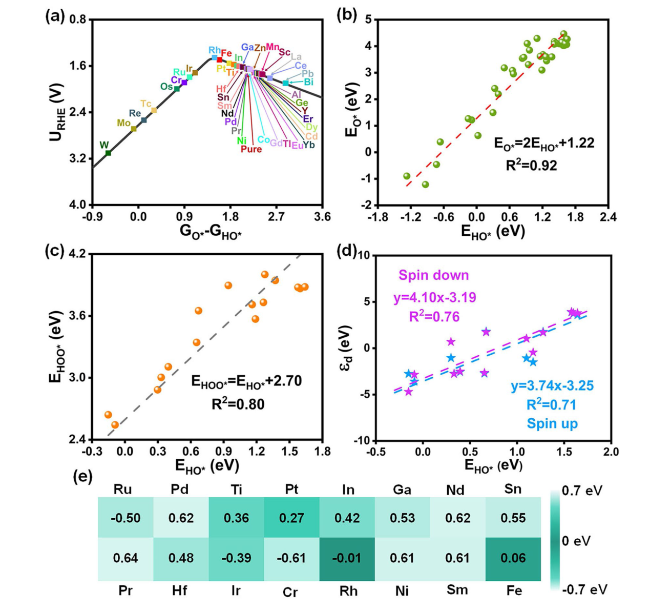
Figure 3a illustrates the kinetic OER activity relating potential to
GO*-
GHO* at a current density of 5
μ A cm
−2, featuring the characteristic volcano-shape. On the left side of the peak, decreased
GO*-
GHO* values correspond to lower OER kinetic activity. Similarly, catalysts exhibit the decaying OER performance along the right arm of the volcano curve. Catalysts close to volcanic peaks, such as Co, Mn, Rh, Fe, and Pt, demonstrate good catalytic activity. Especially, Rh sitting at the volcano peak, with an optimal
GO*-
GHO* value, requires relatively low overpotentials to achieve high reaction rates in acidic solutions, demonstrating the highest OER activity. In addition, we plot potential as a function of
GO*-
GHO* at 1 mA cm
−2 (Fig. S4) and 1 A cm
−2 (Fig. S5), and the results display that the activity order may change as the current increases, but the qualitative order remains constant; that is, a good catalyst is always good at different currents.
Figure 3b displays a linear scaling relation between the adsorption energy of O* (
EO*) and HO* (E
HO*), with the identified slope and intercept close to those of higher-index (where
h2 +
k2 +
l2 >
1) metal oxide surfaces [
52].
Figure 3c further demonstrates that the adsorption energy of HOO* (
EHOO*) and E
HO* exists a good linear correlation and a constant difference between E
HOO* and E
HO* on effectively doped catalysts, which implies
GHOO* –
GHO* = (
GO* –
GHO*) + (
GHOO* –
GO*) = constant, i.e.,
GHOO* –
GHO* = (
GO* –
GHO*) + [constant(
GO* –
GHO*)]. Therefore,
GO* –
GHO* serves as a distinctive descriptor elucidating the OER activity, further reinforcing the rationale behind the selection of
Fig. 3a. Furthermore, E
HO* and
$\varepsilon_{d^{-}} \text {-up } / \varepsilon_{d} \text {-down }$ demonstrate a robust linear correlation, as depicted in
Fig. 3d. Such correlation offers valuable insights into finding optimal HO binding energies by manipulating the
$\varepsilon_{d}$ value. Generally, the optimal OER catalyst has surface hydroxide deprotonation free energy value of 1.5 to 1.7 eV [
43,
53,
54,
55]. In our work, taking 1.5 eV as the standard fits well with the dynamic trend (
Fig. 3a), we use (
GO* −
GHO* = 1.5 eV) as the descriptor to further embody catalytic behavior. A smaller absolute value of (
GO* −
GHO* = 1.5 eV), represented by a darker color, suggests better OER performance (
Fig. 3e).
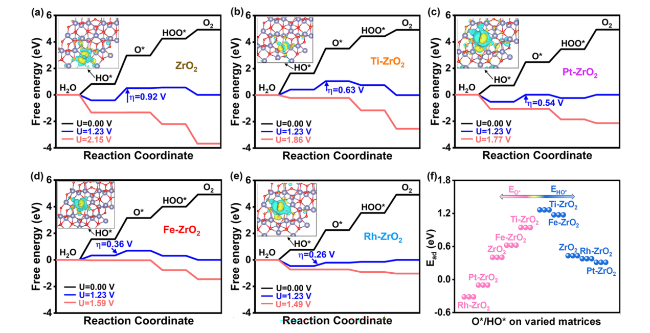
Figure 3e selects all metals closing to the volcanic peak exhibits improved OER performance compared to the pristine ZrO
2, in which Rh has the best catalytic activity with values closest to zero. Theoretical overpotential (
η), a fundamental thermodynamic parameter, has emerged as a crucial metric for evaluating and predicting the OER performance of catalysts. Thus, we plot
η as a function of
GO* −
GHO*, which has a volcano relationship (Fig. S6). Rh standing on the peak of the volcano map exhibits the highest OER activity with minimum
η, followed by Fe, Pt, etc. This thermodynamic volcano diagram trend is roughly consistent with the dynamic volcano diagram (
Fig. 3a), and the gap may be attributed to the omission of transition states in thermodynamics. To acquire a more profound comprehension of the impact of doping on the electronic properties of the catalyst, we calculate the density of states (DOS) of ZrO
2 (Fig. S7a). Near the Fermi level, the valence band is predominantly contributed by O orbitals, while the conduction band is primarily composed of Zr orbitals. A wide forbidden region exists between the top of the valence band and the bottom of the conduction band with a bandgap of 2.6 eV, further observed by the band structure diagram (Fig. S7c). Also, the precise HSE functional displayed that ZrO
2 is a semiconductor material with a wide bandgap of 4.45 eV (Fig. S7b). Figures S8 and S9 show the DOS of metal doped ZrO
2. The bandgap values exhibit a progressively decreasing trend in the following order: Ir (2.54 eV), Ru (2.49 eV), Sn (2.28 eV), Ti (1.82 eV), and Cr (1.71 eV). In, Sm, Pr, Fe, Nd, Ni, Pd, Ga, Co, Pt, Mn, and Rh metals, on the other hand, induce a direct transformation of the catalyst from a semiconductor to a semi-metallic property characterized by a band gap of 0 eV, effectively promoting the electron transfer and thereby improving the conductivity of the ZrO
2. In short, the microkinetic volcano diagrams screen out some promising catalysts, the linear relationship obtains the abnormal adsorption points and the correlation between different adsorbates, and DOS uncover enhanced conductivity of catalysts through doping.