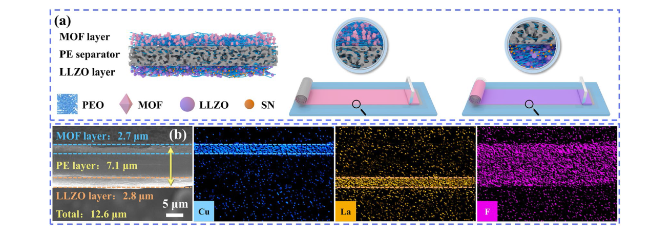
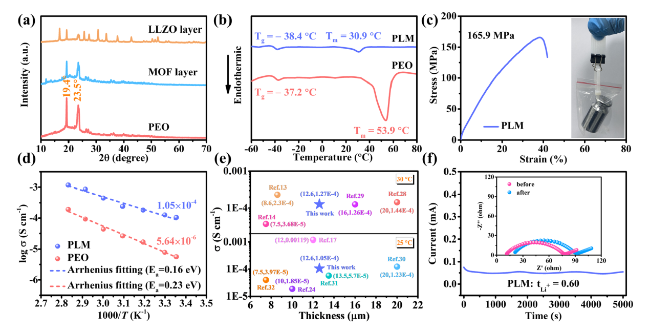
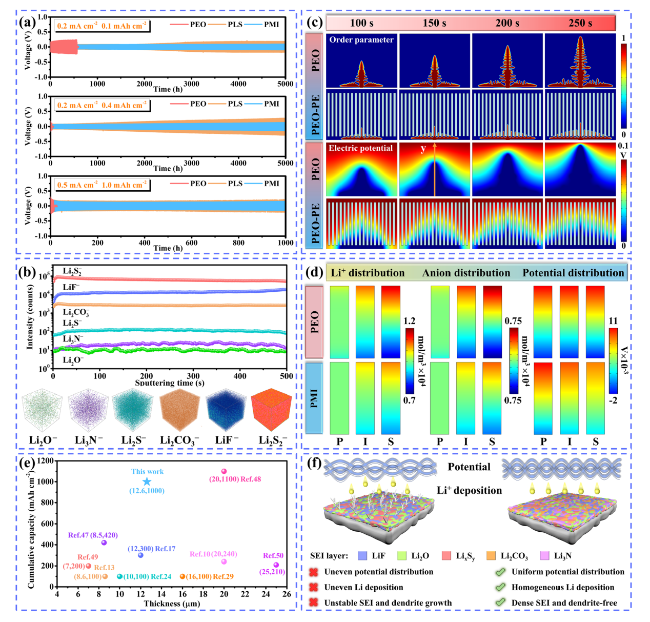
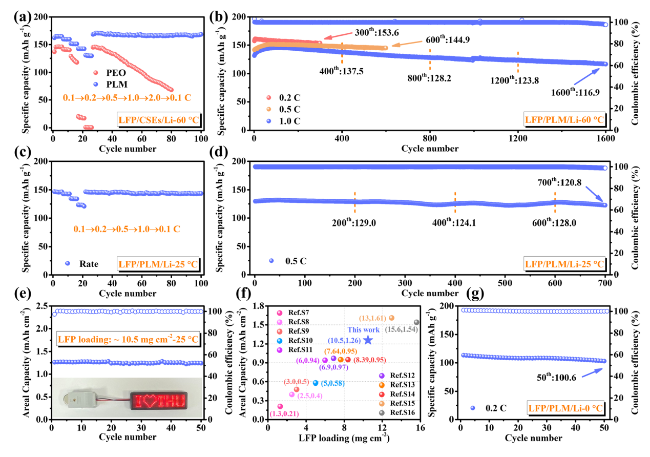
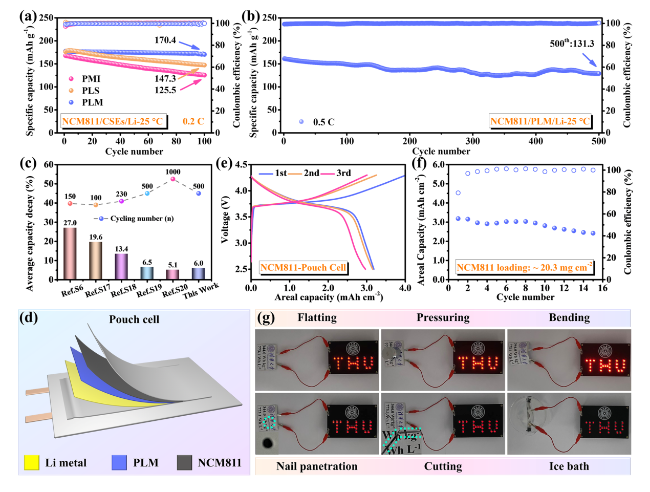
To demonstrate the potential applications of PLM electrolytes for SSLMB, the electrochemical performance of LFP batteries was first tested at high temperature (60 °C). LFP/PLM/Li battery exhibited superior rate performance (
Fig. 4a), providing a specific capacity of 129.8 mAh g
−1 even at 2.0 C. When the rate returns to 0.1 C, the specific capacity can be fully recovered (167.3 mAh g
−1). In contrast, LFP/PEO/Li battery cannot provide sufficient specific capacity at high rates. Since the PLM electrolyte has higher ionic conductivity and Li
+ transference number, LFP/PLM/Li exhibits lower polarization potentials in different rate tests [
51] (Fig. S28). In addition, the ultrathin characteristics and the low interfacial impedance (Fig. S29) of the PLM electrolyte ensures fast Li
+ transport at high rate of SSLMBs [
15]. A further comparison of the long-term cyclic stability of the two SSLMBs is performed. The specific capacity of LFP/PEO/Li battery gradually decreases with the extension of the cycle number (Fig. S30), exhibiting poor cycling stability, excessive polarization potential (Fig. S31) and interfacial impedance (Fig. S32). This instability is due to poor interfacial contact and Li dendrite growth in PEO electrolyte, leading to capacity fading [
16,
52]. In contrast, with the help of the MOF/LLZO layer and PE separator, the PLM electrolyte achieves low interfacial impedance and dendrite suppression, allowing the stable operation of SSLMBs at different rate (0.2 C/153.6 mAh g
−1-300th, 0.5 C/144.9 mAh g
−1-600th). Even after 1600 cycles at 1.0 C (
Fig. 4b), it can still provide a specific capacity of 116.9 mAh g
−1 with an average specific capacity decay as low as 1.78% (per cycle), which is better than the recently reported SSLMBs [
14,
15,
16,
24,
36,
47,
53,
54,
55] (Fig. S33). In addition, the design of the asymmetric PLM electrolyte significantly improved the cycling performance of the PE separator reinforced polymer matrix electrolyte at 1.0 C (Fig. S34). The potential distributions of SSLMBs configured with different electrolytes (PEO and PLM) were simulated by COMSOL Multiphysics (Fig. S35) to further reveal the essence of the excellent electrochemical performance of asymmetric electrolytes. Larger potential gradient changes are observed in the 2D cross-sectional view of SSLMBs with PEO electrolytes, especially higher potentials exist at the LFP/PEO interface. These factors lead to the formation of a space charge layer, resulting in severe polarization of the battery [
10]. Benefiting from the designed PLM electrolyte with asymmetric structure, a more uniform potential is generated, which will alleviate the ion concentration polarization and enhance the cycle stability.