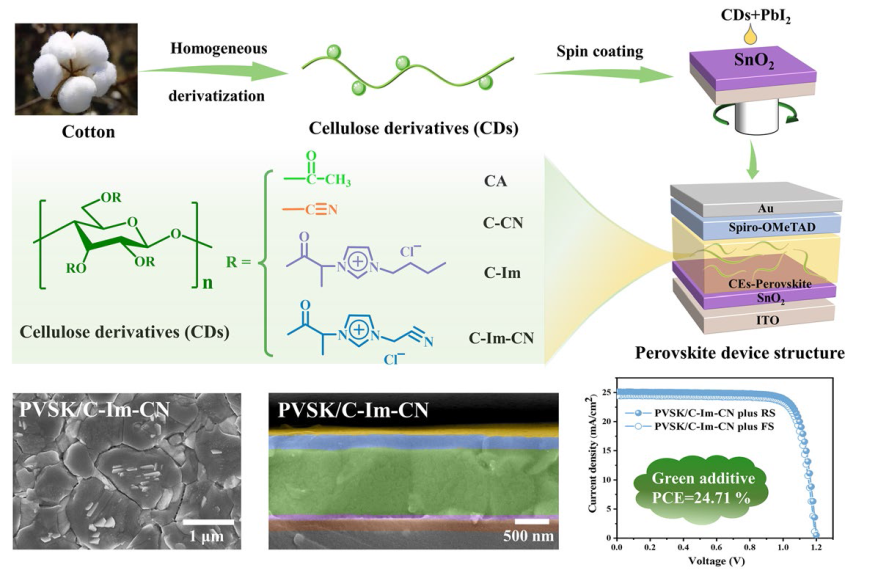
C-Im-CN has several functional groups, including Cl
− anion, cyano group, carbonyl group, hydroxyl group, and imidazolium cation (
Fig. 2a). They strongly interact with Pb
2+ cation, FA
+ cation, MA
+ cation, I
− anion, and Br
− anion in perovskite, via electrostatic interaction, coordination interaction, and hydrogen-bonding interaction. Compared with those in the control perovskite, the Pb 4
f peak of PVSK/C-Im-CN shifts 0.39 eV, and the I 3
d peak shifts 0.31 eV toward the low binding energy region (
Fig. 2b, c). Meanwhile, the uncoordinated Pb
0 in the control perovskite totally disappears after using C-Im-CN, suggesting that the C-Im-CN inhibits the defects in perovskite [
56]. In Raman spectra, the peaks at 82, 108, and 138 cm
−1 in perovskite shift to lower frequency region after the addition of C-Im-CN (
Fig. 2d). These three peaks represent the second phonon mode of [PbI
6]
4−, the asymmetric stretching vibration of I-Pb-I, and the oscillation mode of [PbI
6]
4− octahedron, respectively [
45]. These above phenomena indicate that there are strong interactions between C-Im-CN and perovskite [
11,
57,
58]. After the addition of C-Im-CN solution into the ammonium salt (FAI, MACl, and MABr) solutions, the ζ-potential changes from negative to positive (
Fig. 2e), indicating that C-Im-CN forms electrostatic interactions with the anions and cations of perovskite. After mixing PbI
2 solution with C-Im-CN solution, [PbI
2Cl]
−, [PbICl
2]
−, and [PbCl
3]
− complexes form, further demonstrating a strong coordination interaction between the PbI
2 and the Cl
− anion in C-Im-CN (
Fig. 2f). Moreover, the stretching vibration peak of C=O shifts from 1742 to 1755 cm
−1, the C≡N peak shifts from 2163 to 2251 cm
−1, and the C=N peak shifts from 1564 to 1620 cm
−1 (
Fig. 2g). FTIR results confirm that the C=O and C≡N in C-Im-CN form strong coordination interactions with Pb
2+, and the imidazolium cation in C-Im-CN forms electrostatic interactions with I
− anion. XPS results show the similar conclusion (Fig. S15). In addition, the hydrogen on the imidazolium cation shifts toward the higher field after mixing the PbI
2 solution with C-Im-CN solution (
Fig. 2h), demonstrating that the formation of complexes between the Cl
− anion and Pb
2+ ions weakens the hydrogen-bonding interaction between the Cl
− anion and the imidazolium cation. Besides, compared with the control perovskite, the N 1
s peaks of PVSK/C-Im and PVSK/C-Im-CN shift toward lower binding energies, while the N 1
s peaks of PVSK/CA and PVSK/C-CN show no shift, indicating that the Cl
− anions form electrostatic and/or hydrogen-bonding interactions with FA
+ cations in perovskites (
Fig. 2i). After mixing FAI solution with C-Im-CN solution, the O-H stretching vibration peak shifts from 3355 to 3280 cm
−1, and the N-H stretching vibration peak shifts from 3180 to 3071 cm
−1, demonstrating that FAI forms multiple hydrogen-bonding interactions with C-Im-CN (
Fig. 2j). Furthermore, the amine hydrogen of FAI at 8.99 ppm shifts to 9.07 ppm which demonstrates that the Cl
− anion forms a hydrogen-bonding interaction with the FA
+ ion (
Fig. 2k). Therefore, the Cl
− anion, cyano group, and carbonyl group in C-Im-CN form coordination interactions with Pb
2+ ion; the Cl
− anion, hydroxyl group, cyano group, and carbonyl group in C-Im-CN form hydrogen-bonding interactions with FA
+ ion and halogen anion in perovskite; and the imidazolium cation and Cl
− anion in C-Im-CN form electrostatic interactions with Pb
2+ ion, FA
+ ion and halogen anion in perovskite (
Fig. 2l). As a result, C-Im-CN markedly promotes the grain growth and directional orientation of perovskite, suppresses the ion migration, vacancies and boundary defects, as well as conducts the transfer of PbI
2.