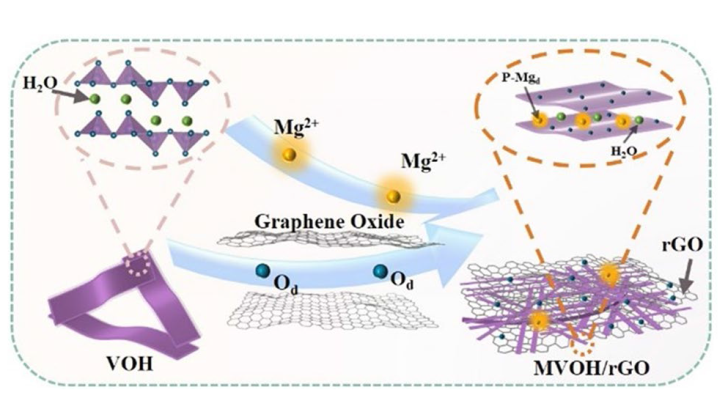
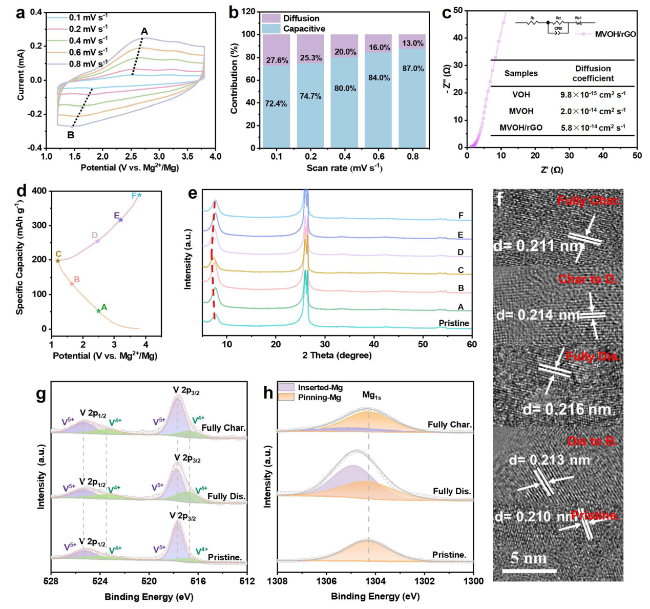
We employed a facile one-step hydrothermal method to synthesize rGO-substrated Mg
0.07V
2O
5·1.4H
2O nanobelts (MVOH/rGO) with dual defects, namely, interlayer P-Mg
d and surface O
d in V-O layers (Table S1). The XRD pattern shown in
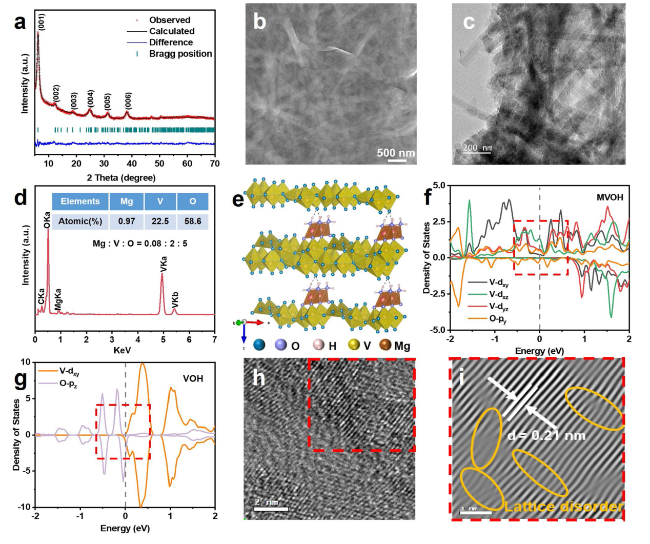
Fig. 1a displays a predominant diffraction peak at 6.1°, corresponding to the (001) plane, indicating a typical layered structure with an impressively large interlayer spacing of 14.5 Å [
17,
38]. The Rietveld refinement method was employed to analyze the further detailed crystal structure of MVOH/rGO (Table S2), which reveals a monoclinic structure with space group C2/m, and the refined parameters are calculated to be
a = 14.19 Å,
b = 3.77 Å, and
c = 14.34 Å;
α =
γ = 90°, and
β = 83.9°. Notably, MVOH/rGO comprises anisotropic nanobelts several micrometers in length, and the finite width (50-70 nm) of the nanobelts effectively shortens the migration pathway of Mg
2+ (
Fig. 1b and
c). The corresponding elemental mapping results (
Figs. 1d and S1) confirm the presence of Mg, V, O, and C, aligning with the results of ICP-OES (Table S1). The P-Mg
d generates MgO
5 pyramids between the V-O layers (
Figs. 1e and S2). These MgO
5 pyramids function as pins, fixing the lamellar structure of V
2O
5 in MVOH/rGO and thereby steadily restraining volume changes caused by Mg
2+ insertion/extraction during discharging/charging. In addition, the P-Mg
d induces the polarization of V-O bonds [
39]. According to ab initio calculations, this polarization leads to 3
d orbital spitting, altering orbital hybridization in V-O bonds (
Figs. 1f and S3a, b). Consequently, the valence band of V
2O
5 with P-Mg
d overlaps with its conduction band, resulting in substantially higher electronic conductivity of V
2O
5 in MVOH/rGO than that of V
2O
5·nH
2O (
Figs. 1g and S3c, d). Furthermore, the P-Mg
d causes the reduction of trace V
5+ to V
4+, as verified by the average vanadium valence of + 4.93 in MVOH/rGO according to the chemical formula of Mg
0.07V
2O
5·1.4H
2O (Fig. S5). The 3
d electrons of V
4+ can easily hop to the vacant 3
d orbitals of V
5+, further increasing the electronic conductivity of V
2O
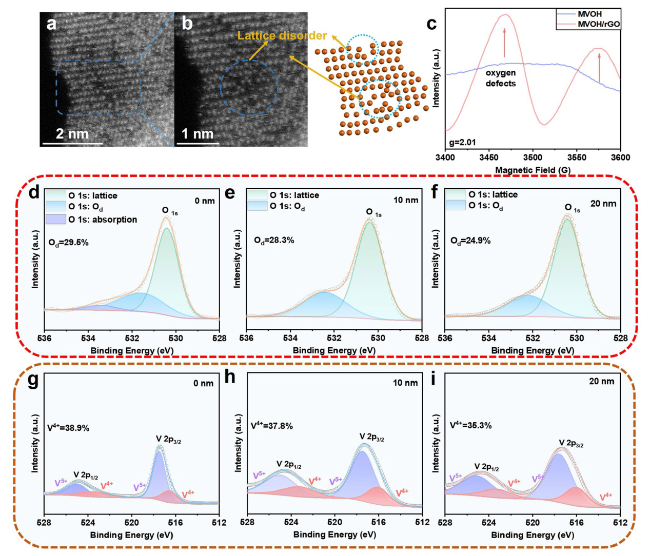
5 nanobelts in MVOH/rGO. The O
d in the V-O layers is generated by rGO and reduces the negative charge density of the layers. This leads to looser bonding between the V-O layers and inserted Mg
2+, resulting in a considerable interlayer spacing of 14.5 Å between the V-O layers of V
2O
5 in MVOH/rGO. In comparison, V
2O
5·1.5H
2O (VOH) synthesized under similar conditions exhibits an interlayer spacing of 14.5 Å, while Mg
0.05V
2O
5·1.0H
2O (MVOH) synthesized under the same conditions with only P-Mg
d has an interlayer spacing as small as 13.5 Å (Fig. S6); this result shows the interlayer spacing enlargement effect of the O
d endowed by rGO [
40]. The O
d also causes the reduction of superficial V
5+ to V
4+ at the interface between rGO and V
2O
5, further increasing the electronic conductivity of V
2O
5 in MVOH/rGO. According to the Raman analysis (Fig. S7), the signal intensity ratio of
sp2 graphite carbon (D band) and
sp3 disordered carbon (G band) in MVOH/rGO (
ID/
IG = 1.14) is significantly higher than that in GO (
ID/
IG = 0.8), implying that the deoxidization of GO is realized by the redox reaction between carboxyl groups in GO and V
5+ [
41,
44]. This is verified by the gas bubbles being observed after the hydrothermal synthesis of MVOH/rGO, which are attributed to the CO
2 gas produced by the oxidation of carboxyl groups in GO. Due to the excellent electronic conductivity of rGO, the V
2O
5 nanobelts are connected by the conductive rGO network, ensuring rapid electrochemical insertion/extraction of Mg
2+.