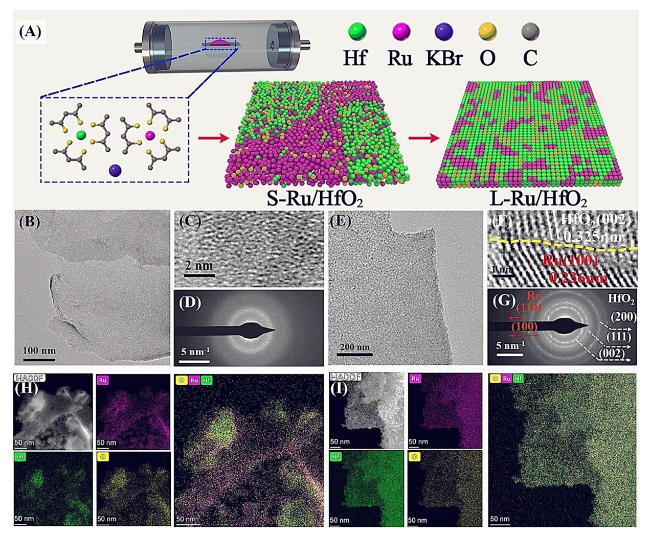
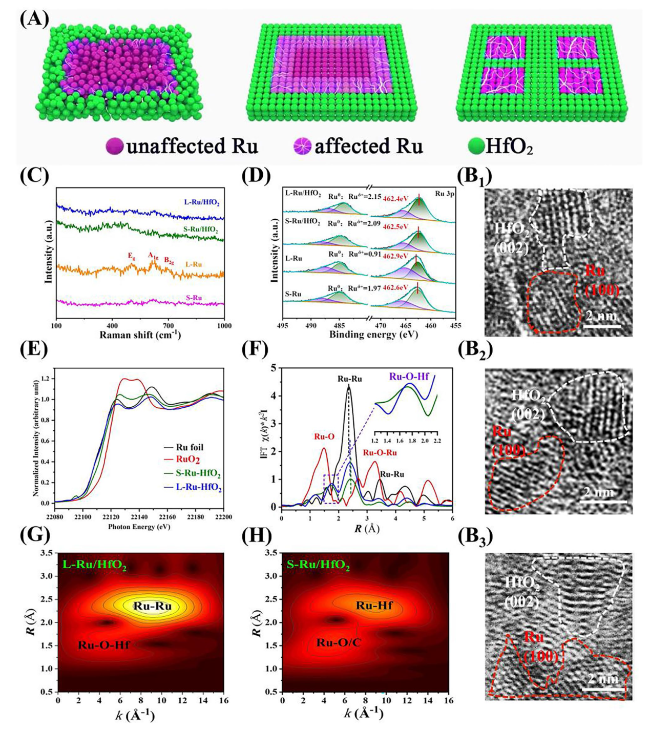
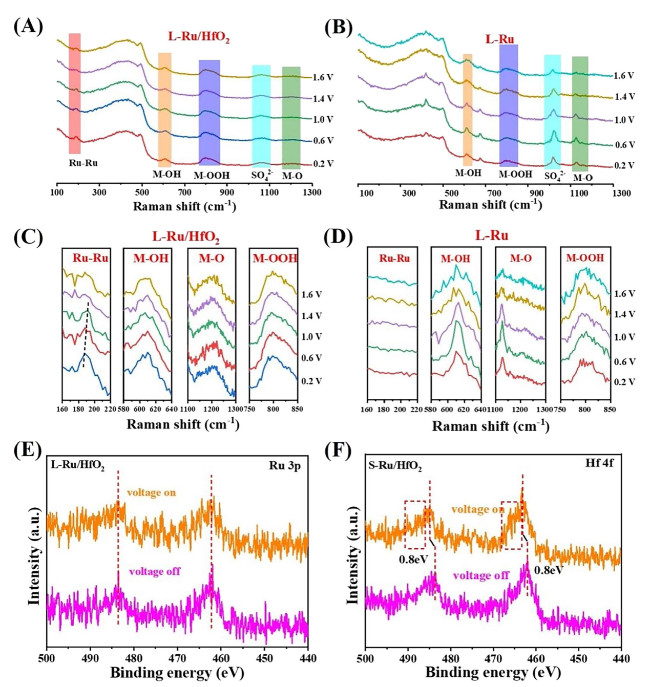
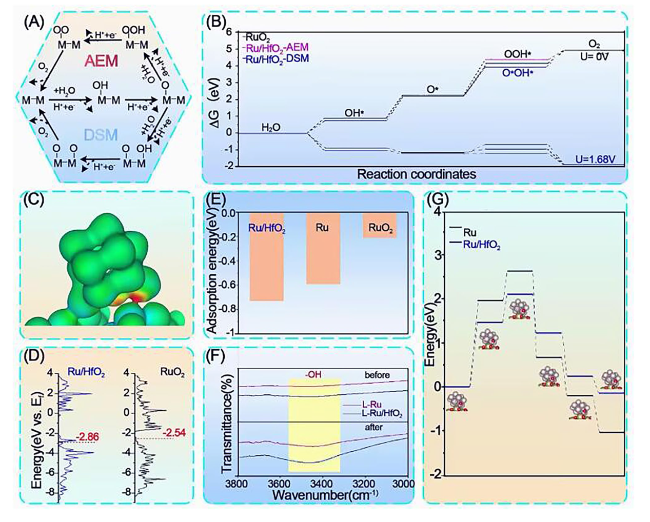
Afterward, X-ray absorption spectroscopy (XAS) was conducted to investigate the electronic configuration and local chemical environment of L-Ru/HfO
2. As shown in
Fig. 2E, X-ray absorption near-edge structure (XANES) analysis demonstrated that the Ru K-edge of S-Ru/HfO
2 suited between commercial Ru foil and RuO
2, which suggested the metaphase of Ru with partial positively charged state between Ru
0 and Ru
4+ [
50]. Notably, L-Ru/HfO
2 exhibited a much more negative absorption edge than S-Ru/HfO
2, which almost coincided with that of Ru foil. Beyond, the white line intensity of Ru foil was smaller than S-Ru/HfO
2 but larger than L-Ru/HfO
2, revealing the majority of Ru atoms in L-Ru/HfO
2 possessed Ru
0 state with negligible oxidation. Furthermore, the peak of S-Ru/HfO
2 in the pre-edge region disappeared on L-Ru/HfO
2, which indicated the strengthened interaction between Ru and HfO
2, with more empty orbitals of Ru being filled by the electron injection during the post-annealing procedure. The two scattering peaks observed at ~2.4 and 1.7 Å in EXAFS were indexed to Ru-Ru and Ru-O-Hf coordination, respectively (
Fig. 2F and Table S1). Apparently, the Ru-Ru peak of L-Ru/HfO
2 was significantly stronger than Ru-O, verifying the majority of metallic Ru state in the crystalline heterostructure. In contrast, S-Ru/HfO
2 exhibited a relatively high intensity for Ru-O peak but the intensity of Ru-Ru bonding was much lower as compared. The enlarged view in
Fig. 2F unraveled the Ru-O-Hf peak of L-Ru/HfO
2 shifted to a smaller distance when compared to that of S-Ru/HfO
2, presumably originating from the stronger interaction imposed between Ru and HfO
2. The Ru-Ru coordination numbers increased from 4.7 to 6.6 as S-Ru/HfO
2 transformed to L-Ru/HfO
2 (Table S1), further confirming the higher crystallinity induced more Ru atoms to be coordinated and protected by HfO
2. On basis of these discussions, it was concluded that the Ru sites were more effectively protected by the HfO
2 matrix in L-Ru/HfO
2 and thereby generated a higher oxidation resistance for the higher crystalline catalyst during operation. Besides, the wavelet transforms of Ru-edge EXAFS oscillations demonstrated the maximum-intensity values of L-Ru/HfO
2 were located at
k ≈ 4.9 and 9.1 Å
−1, which were assigned to Ru-O-Hf and Ru-Ru scattering paths, respectively (
Fig. 2G). It also reflected that L-Ru/HfO
2 possessed more metallic Ru states and fewer Ru-O bonds than S-Ru/HfO
2 (
Fig. 2G, H), exhibiting a metallic feature that quite close to Ru foil. Since ICP measurements disclosed a similar atomic ratio of Ru to Hf for S-Ru/HfO
2 (0.88) and L-Ru/HfO
2 (0.80), we attributed the more stable Ru
0 state of L-Ru/HfO
2 to the enhanced interfacial interaction constructed by increasing the crystallinity of Ru and HfO
2.