HIGHLIGHTS
1 Introduction
2 Methods
2.1 Preparation of DLC and PLA/DLC Membrane
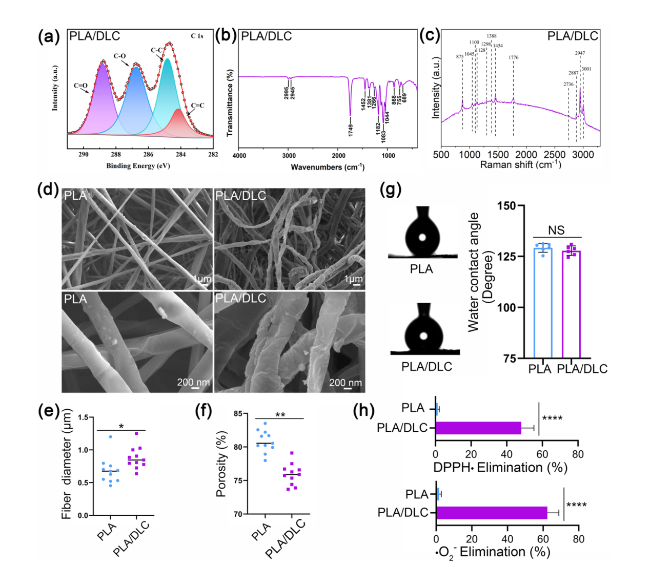
Fig. 1 Characterization of the PLA/DLC membranes. a XPS spectra of PLA/DLC. b FTIR spectrum. c Raman spectroscopy. d SEM micrographs, e fiber diameter and f porosity. g Water contact angle image and quantifying. h DPPH· and ·O2− scavenging. Data represent independent experiments, and all data are presented as mean ± SD; NS non-significant, P > 0.05; *P < 0.05; **P < 0.01; ****P < 0.0001 |
2.2 Characterizations
2.3 In vitro Investigation
2.3.1 PLA Degradation and Lactic Acid Release
2.3.2 Cell Culture and Treatment
2.3.3 Cell Proliferation, Live/Dead Cell Staining, and Cell Adhesion Assay
2.3.4 Cell Oxidative Stress Assessment
2.3.5 Macrophage Polarization
2.4 In Vivo Investigation
2.4.1 Preparation of Rat Achilles Tendon Adhesion Model
2.4.2 Gross Evaluation
2.4.3 Histological Assessment
2.4.4 Western Blotting
2.4.5 Detection of Tissue Oxidation Products
2.4.6 Inflammatory Cytokine Level in Peritendinous Tissue
2.5 Statistical Analysis
3 Results
3.1 PLA/DLC Suppressed ROS Production and Alleviated Oxidative Stress without Affecting the Fibroblast Proliferation and Adhesion
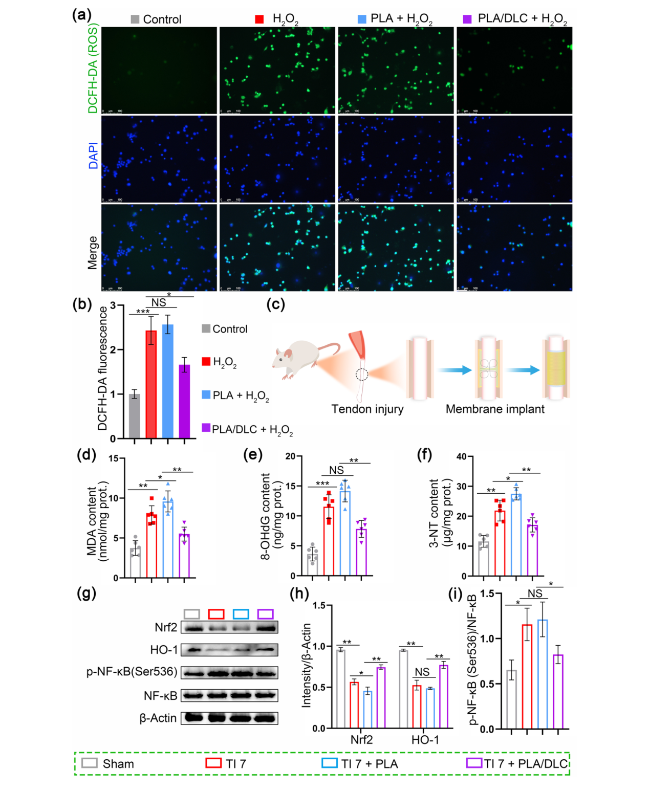
Fig. 2 DLC suppress ROS production and oxidative stress both in vitro and in vivo. a Detection of cell ROS level on different membranes using ROS probes and b statistical analysis. c Diagram of animal experiment. d, f Content of MDA, 8-OHdG, and 3-NT in peritendinous tissues after 7 days of injury was examined by assay kit respectively. g Representative bands of Nrf2, HO-1, p-NF-κB were measured by Western blotting. h Nrf2 and HO-1 expression normalized to β-actin expression. i p-NF-κB expression normalized to NF-κB expression. Data represent independent experiments, and all data are presented as mean ± SD; NS non-significant, P > 0.05; *P < 0.05; **P < 0.01; ***P < 0.001, ****P < 0.0001 |
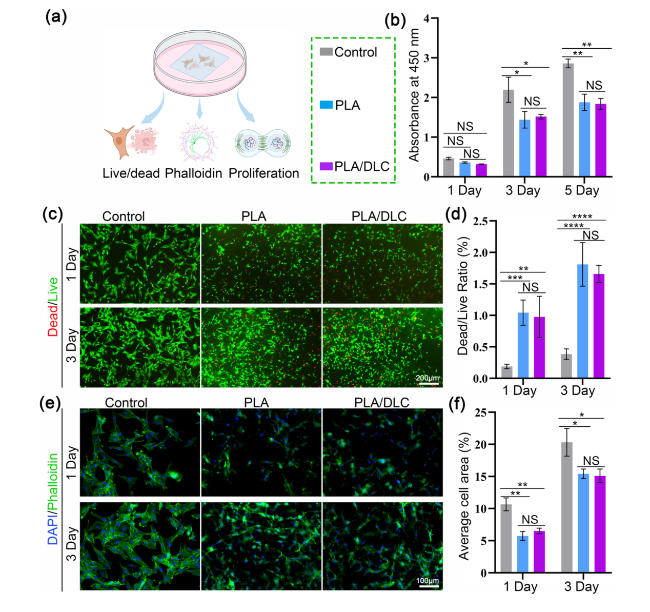
Fig. 3 DLC do not alter the proliferation and adhesion of fibroblasts in vitro. a Fibroblasts live/dead, proliferation, and adhesion analysis in vitro. b Proliferation of fibroblasts cultured on different membranes at days 1, 3, and 5 were measured using CCK8 kits. c Live/dead staining of fibroblasts cultured on different membranes for 1 and 3 days, and d Dead/live cell rate. Red: dead cells; green: live cells. e Adhesion analysis of fibroblasts cultured on different membranes at days 1 and 3, and f cellular arrangement area. Data represent independent experiments, and all data are given as mean ± SD; NS non-significant, P > 0.05; *P < 0.05; **P < 0.01; ***P < 0.001, ****P < 0.0001 |
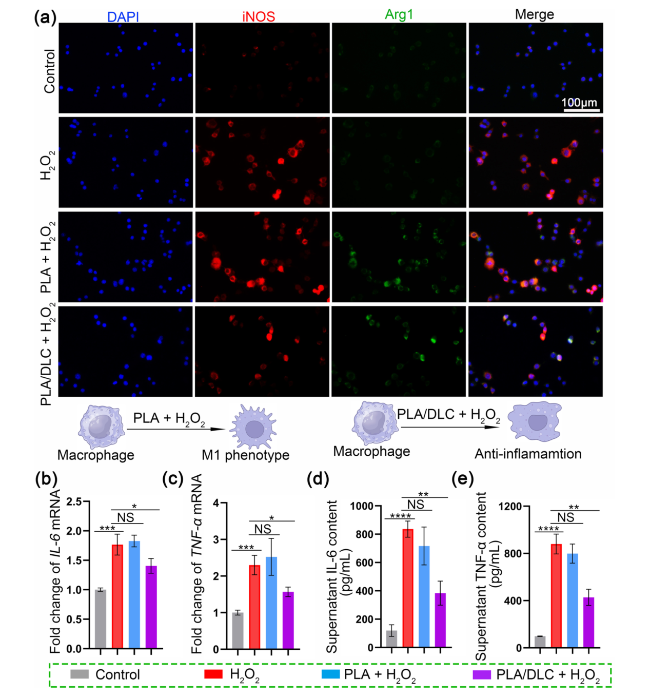
3.2 PLA/DLC Showed Anti-Inflammatory Properties in Vitro
Fig. 4 DLC showed anti-inflammatory properties in vitro. The H2O2 group was established in vivo. a iNOS/Arg-1 fluorescence double-staining was used to analyze the anti-inflammatory properties of PLA/DLC. b, c The mRNA expression levels of IL-6 and TNF-α were detected by real-time PCR. d, e The concentrations of TNF-α and IL-6 in cell supernatant were measured by ELISA. Data represent independent experiments, and all data are given as mean ± SD; NS non-significant, P > 0.05; *P < 0.05; **P < 0.01; ***P < 0.001, ****P < 0.0001 |
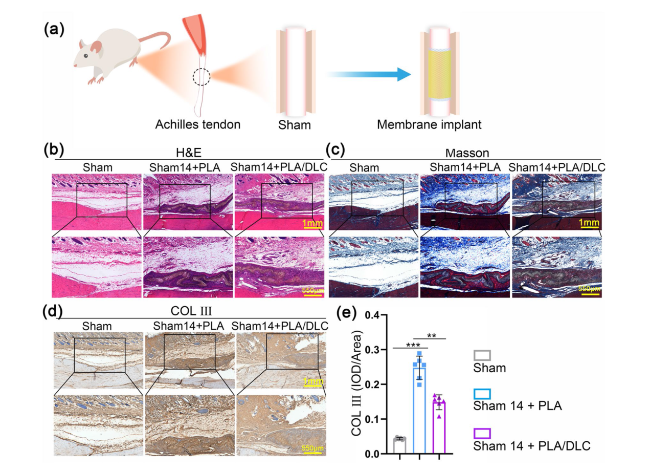
3.3 PLA/DLC Mitigated Inflammation Response and Abnormal Hyperplasia Caused by PLA Membrane Implantation
Fig. 5 DLC inhibited inflammatory infiltration and abnormal hyperplasia induced by PLA membrane implantation. a Diagram of animal experiment. b H&E staining and c Masson trichrome staining of normal tendon wrapped by PLA and PLA/DLC for 14 d. d Immunohistochemical staining for COL III of Sham group, Sham + PLA group, and Sham + PLA/DLC group at 14 d. e Optical density of COL III was measured in the peritendinous tissues. Data represent independent experiments, and all data are presented as mean ± SD; NS non-significant, P > 0.05; *P < 0.05; **P < 0.01; ***P < 0.001, ****P < 0.0001 |
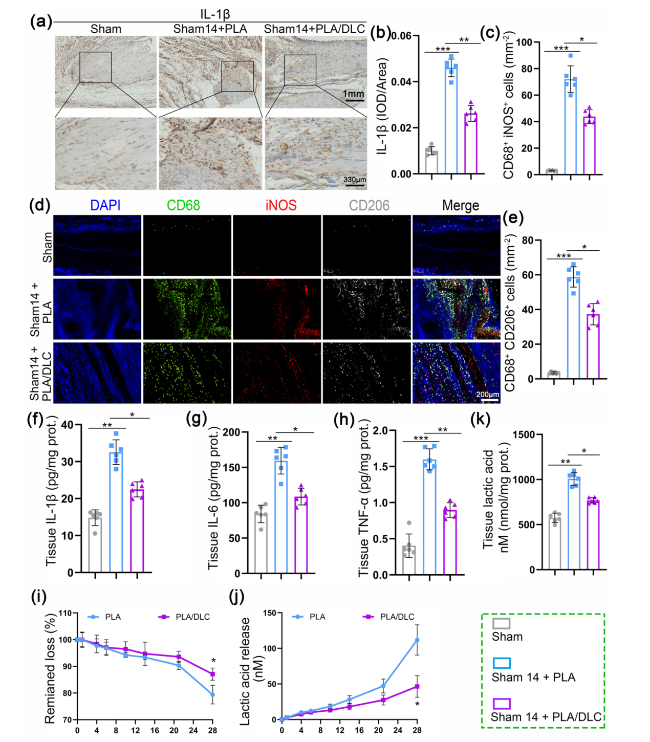
Fig. 6 DLC inhibited PLA implantation-induced foreign body inflammation response and delayed PLA degradation. a Immunohistochemical staining for IL-1β in Sham group, Sham + PLA group, and Sham + PLA/DLC group at 14 d. b The optical density of IL-1β in the peritendinous tissues was measured. c The mean cell area of iNOS-positive cells in macrophages. d CD68/iNOS/CD206 fluorescence trichrome staining was used to measure the macrophage polarization around normal tendons. e The mean cell area of CD206-positive cells in macrophages. f-h The expression of IL-1β, IL-6, and TNF-α in normal peritendinous tissue was measured by ELISA after 14 d of implantation. i The remaining loss and j lactic acid release from enzymatic ester hydrolysis of PLA and PLA/DLC were measured at different time points. k The lactic acid level in the peritendinous tissue was measured in each group after 14 d of implanting PLA and PLA/DLC into the normal tendon. Data represent independent experiments, and all data are presented as mean ± SD; NS non-significant, P > 0.05; *P < 0.05; **P < 0.01; ***P < 0.001, ****P < 0.0001 |
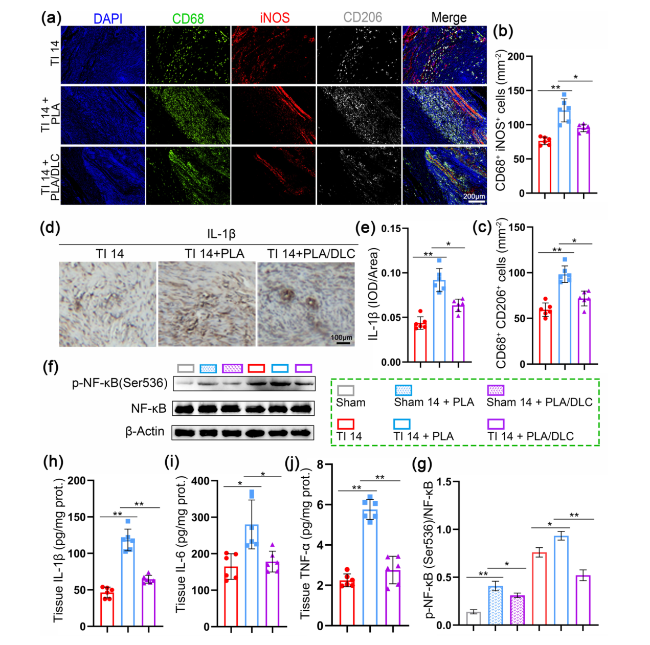
Fig. 7 DLC suppressed the inflammatory response during tendon repair. a CD68/iNOS/CD206 fluorescence trichrome staining was used to measure the macrophage polarization around injured tendons. b, c The mean cell area of iNOS-positive and CD206-positive cells. d, e Immunohistochemical staining for IL-1β in the TI group, TI + PLA group, and TI + PLA/DLC group at 14 d, and relative IL-1β-positive cell area. f Representative bands of p-NF-κB in peritendinous tissues were measured by Western blotting. h-j The expression of IL-1β, IL-6, and TNF-α in peritendinous adhesion tissue of 14 d was measured by ELISA. g p-NF-κB expression was normalized to NF-κB expression. Data represent independent experiments, and all data are presented as mean ± SD; NS non-significant, P > 0.05; *P < 0.05; **P < 0.01; ***P < 0.001, ****P < 0.0001 |
3.4 DLC Depositing Delayed the Degradation of the PLA Membrane
3.5 DLC Alleviated the Inflammatory Response during Tendon Repair
3.6 DLC Depositing Improved the Anti-Adhesion Effect of the PLA Membrane
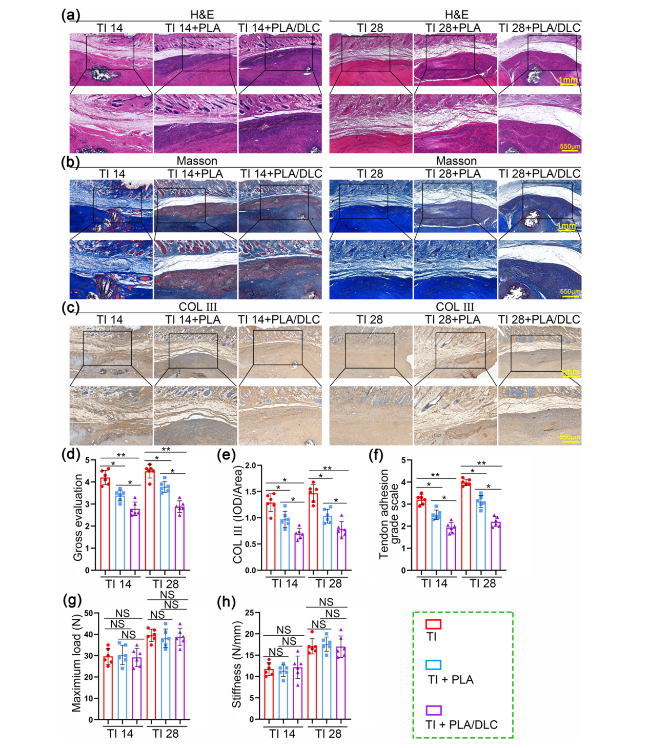
Fig. 8 DLC depositing improved the anti-adhesion efficiency of the PLA membrane. a H&E staining and b Masson trichrome staining of normal tendon wrapped by PLA and PLA/DLC for 14 and 28 days. c Immunohistochemical staining for COL III of TI group, TI + PLA group, and TI + PLA/DLC group at 14 and 28 days. d Gross histological view, e Average optical density of COL III in the peritendinous adhesion tissues were exhibited. f Adhesion score was assessed after 14 and 28 days. g Maximum load and h stiffness of the regenerated tendon. Data represent independent experiments, and all data are presented as mean ± SD; NS non-significant, P > 0.05; *P < 0.05; **P < 0.01; ***P < 0.001, ****P < 0.0001) |
4 Conclusion
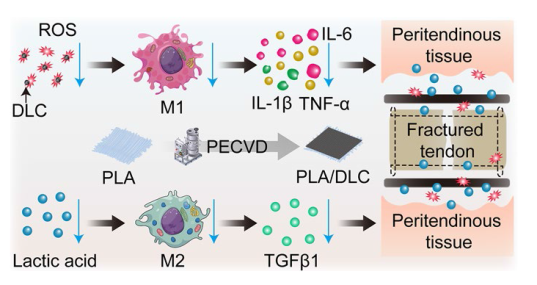
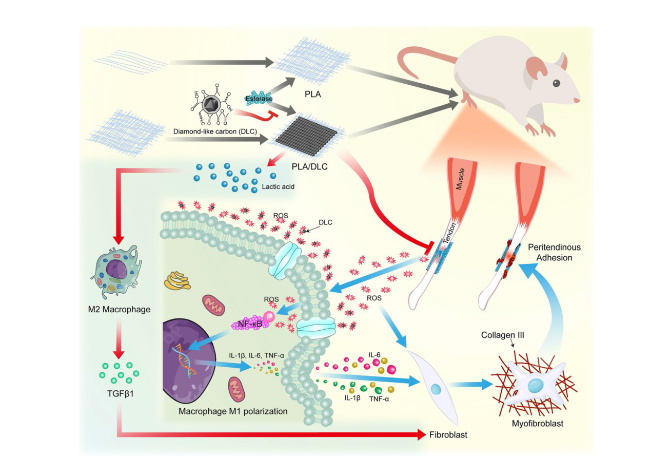
Fig. 9 Putative mechanisms by which DLC-deposited PLA membrane displays more effective anti-adhesion capacity than the PLA membrane. DLC reduces the production of ROS and alleviates oxidative stress because of the abundant functional groups on the DLC surface. Decreasing ROS levels suppresses NF-κB phosphorylation and then mitigates macrophage M1 polarization during FBR after PLA implantation. DLC blocks the ester bonds in PLA fibers from esterase spatially and delay lactic acid releasing, thereby inhibiting the lactic acid-induced M2 polarization of macrophages. Thus, DLC reduces the side effects of the implants and enhance their anti-adhesion efficiency |