Introduction
Materials and methods
Study design
Human brain samples
Mice
Cell lines
Collection of conditioned medium (CM)
Quiescent primary astrocytes
Primary microglia
Cell treatments
RNA extraction, RNA sequencing and Quantitative PCR
Immunofluorescence labelling
Immunoblotting
Live calcium imaging
Confocal microscopy
Fluorescence intensity measurements
Single-cell fluorescence
Total cell fluorescence
3D imaging and morphometric analysis of astrocytes
Protein extraction
Cytokine measurement
Secretomics
Sample preparation
Liquid chromatography-mass spectrometry (LC-MS)/mass spectrometry (MS) analysis
MS data processing, quantification and statistical analysis
Statistical analysis
Results
α-Synuclein oligomers are correlated with persistent neuroinflammation in mouse brains
Fig. 1 Pathological α-synuclein oligomers are associated with inflammatory responses in A53T mouse striatum. a TNFα (**P = 0.0029), IL-1β (***P = 0.0003), IL-10 (***P = 0.0001) and IFNγ (#P < 0.0001) levels in striatal homogenates determined using cytokine-specific ELISA assays (N ≥ 4 per genotype). b, c Representative western blots of CHAPS-homogenized striatum using antibodies against total (anti-Syn1) and phosphorylated α-synuclein. d Immunoblots of mouse IgG heavy chain and densitometric quantification in WT (n = 16) and A53T Tg (n = 14) mice, #P < 0.0001. e-g Correlation analysis of mouse IgG light chain in A53T Tg mice with α-synuclein oligomers (e), α-synuclein monomers (f), and animal age (g) after immunoblotting and densitometric quantitation (n = 10). h-j Western blotting of homogenized striatal tissues from 1.5-month-old WT and A53T Tg mice (n = 4 mice per genotype) using antibodies against total α-synuclein (anti-Syn1) and densitometric quantification of monomeric α-synuclein levels (i, **P = 0.0019) and IgG levels (j, P = 0.7219). A six-month-old A53T Tg homogenate was used as reference for the presence of α-synuclein oligomers. k Representative immunoblots and quantification of C3d (n = 4 mice per genotype, **P = 0.0074). In b-d, h and k, GAPDH was used as a loading control. Statistics by Mann-Whitney test in (d) and unpaired Student’s t test in (a, i, j, k) |
Cell-produced α-synuclein oligomers, but not monomers, can induce microglial activation
Fig. 2 Cell-produced α-synuclein oligomers, but not monomers, can activate primary microglia. a, f Schematic representations of the experiments with primary microglia treated with CM from SH-SY5Y α-synuclein expressing cells. b Immunoblot showing the presence of cell-secreted (CM) and intracellular (Lys) α-synuclein conformers in the absence of doxycycline (− Dox). c, g Representative images of primary microglia immunostained with a specific antibody against NF-κB. DAPI was used as a marker for nuclei. Magnification of the boxed area is shown for each merged image. Scale bar: 20 μm (50 μm for the magnified images). d, h Quantification of mean fluorescence intensity of nuclear NF-κB (N ≥ 9 for all treatments). e, i Measurement of secreted TNFα from primary microglia upon indicated treatments (n = 3 biological replicates). In d, e, h, and i, statistics was performed by one-way ANOVA followed by Tukey’s multiple comparisons test. In d, #P < 0.0001 for untreated vs LPS, ***P = 0.0001 for + Dox versus − Dox. In e, #P < 0.0001 for untreated versus LPS and + Dox versus − Dox. In h and i, #P < 0.0001 |
A53T microglia and astrocytes exhibit distinct biochemical and morphological alterations
Fig. 3 A53T microglia and astrocytes exhibit biochemical and morphological alterations. a Representative confocal images of WT and A53T Tg striatal sections stained with antibodies against Iba1, CD11b and CD68. DAPI (blue) was used as a nuclei dye. Scale bar: 50 μm (10 μm for the magnified images). b Quantification of the number of Iba1+ microglia cells (n = 21 and n = 19 images from WT and A53T Tg mice, respectively, #P = 0.0239) upon immunostaining of coronal brain sections in the striatum of WT and A53T Tg mice with an Iba1-specific antibody. c Quantification of the total mean fluorescence intensity of Iba1+ cells (n = 7 and n = 13 images from WT and A53T Tg mice, respectively, #P < 0.0001) stained as above. d Immunoblotting analysis of striatum homogenates from WT and A53T Tg mice using a specific antibody to Iba1 and densitometric quantification (n = 10 and n = 11 for WT and A53T Tg mice, respectively, *P = 0.0438). GAPDH was used as a loading control. e Quantification of the mean fluorescence intensity of CD68 in individual Iba1+ cells in three independent pairs (1, 2, 3) of WT and A53T Tg mice; **P = 0.0018 for pair 1, #P < 0.0001 for pairs 2 and 3, n ≥ 74 astrocytes for each genotype. f Representative confocal images of WT and A53T Tg striatal sections stained with a specific antibody for GFAP. Scale bar, 200 μm. g Quantification of the number of GFAP+ astrocytes in the striatum following immunostaining and confocal imaging (n = 9 and n = 13 images for WT and A53T Tg mice, respectively, ***P = 0.0001). h Western blot and GFAP quantification in striatal homogenates (n = 7 mice per genotype, *P = 0.219). i Representative confocal images of WT and A53T Tg striatal sections stained with specific antibodies for TNFα and GFAP. Scale bar: 50 μm (10 μm for the magnified images). j Representative images of 3D reconstructed GFAP-immunoreactive astrocytes from WT and A53T Tg striatum, using the IMARIS software. DAPI was used for nuclei staining. Scale bar, 20 μm. k, l Quantification of the number of branches; k from 16 astrocytes per genotype, *P = 0.0174, and (l) pre-averaged per animal (n = 5 and n = 4 for WT and A53T Tg mice, respectively, **P = 0.0024). m, n Quantification of filament length; (m) length distribution from 16 astrocytes per genotype and (n) pre-averaged per animal (n = 5 and n = 4 for WT and A53T Tg mice, respectively, P = 0.1058). For all comparisons statistics were performed by unpaired Student’s t test |
High levels of α-synuclein motivate neuropeptide-, mitogen-activated protein (MAP) kinase (MAPK)- and Ca2+-dependent signaling pathways
α-Synuclein conformers are associated with activation of the p38MAPK signaling pathway and advancement of the ATF2/7-dependent transcription in microglia
Fig. 4 The p38MAPK and the NF-κB pathways are activated in A53T Tg striatum. a Immunoblotting analysis and quantification of phospho-p38MAPK levels relative to total p38MAPK expression (n = 5 mice per genotype, #P < 0.0001). b Immunoblotting analysis and quantification of phospho-ATF2/7 levels relative to total ATF2 expression (n = 4 mice per genotype, *P = 0.0378). c, e, h Representative magnified confocal images of striatal sections co-stained with antibodies against phospho-p38 (c), phospho-ATF2/7 (e) and phospho-NF-κB (h) and Iba1 or GFAP. DAPI (blue) was used for nuclei staining. Scale bar: 20 μm. d Measurement of phospho-p38 mean fluorescence intensity in Iba1+ cell surfaces (n ≥ 35 cells per mouse for three independent Wt-A53T Tg animal pairs, #P < 0.0001, ***P = 0.0004, *P = 0.0239). f Representative immunoblot and quantification of phospho-NF-κB relative to total NF-κB in striatal homogenates (n = 8 mice per genotype, #P < 0.0001). g qPCR measurement of Egr1 expression (n ≥ 13 per genotype, ***P = 0.0004). (i) Immunoblotting detection and quantification of A20/TNFAIP3 (n ≥ 8 per genotype, **P = 0.0011). GAPDH was used as a loading control. All statistics by unpaired Student’s t test |
A53T astrocytes exhibit an unconstrained activation of the NF-κB pathway
L- and T-type Ca2+ channels are differentially expressed in A53T reactive astrocytes
Fig. 5 Cav1.2 and Cav3.2 are differentially expressed in A53T striatal astrocytes. a-d Representative confocal images of striatal sections co-stained with antibodies against Cav1.2 (a), Cav3.1 (b), Cav3.2 (c), and Cav3.3 (d) and the astrocyte marker, GFAP. DAPI (blue) was used for nuclei staining. Images on the right represent magnification of the boxed area. Scale bar: 50 μm (10 μm for the magnification images). e-h Quantification of mean fluorescence intensity of Cav VGCCs in individual GFAP+ astrocytes following 3D reconstruction in three independent pairs (1, 2, 3) of WT and A53T Tg mice; (e) for Cav1.2, #P < 0.0001, n ≥ 68 astrocytes for each genotype, f for Cav3.1, P = 0.3298, 0.1878, 0.0950 for pairs 1, 2, 3 respectively and n ≥ 44 astrocytes for each genotype, g for Cav3.2, #P < 0.0001, n ≥ 129 astrocytes, and h for Cav3.3, P = 0.2434, 0.2479, 0.0955, n ≥ 16 for each genotype. i Pre-averaged values of Cav mean fluorescence intensity relative to WT for 3 WT and A53T animal pairs. Data are presented as means ± SEM. All statistics by unpaired Student’s t test |
T-type Cav3.2 Ca2+ channels are up-regulated by cytokines in a NF-κB-dependent manner
Fig. 6 T-type Cav3.2 Ca2+ channels are up-regulated by cytokines in a NF-κB-dependent manner. a Representative confocal images of primary astrocytes co-stained with specific antibodies for Cav3.2 and GFAP. b qPCR measurement of CACNA1H mRNA levels following treatment with TNFα, ***P = 0.0003, IL-1β, **P = 0.0042 and IFNγ, ***P = 0.0005 (n ≥ 8 per condition). c CACNA1C mRNA levels assessed as above (TNFα; P = 0.2536, IL-1β; P = 0.8093, IFNγ; P = 0.0537) (n ≥ 8 per condition). Statistics in (b, c) by one-way ANOVA followed by Dunnett’s multiple comparisons test. d-f Representative western blots showing Cav3.2 and p-NF-κB levels following treatment with TNFα, IL-1β or IFNγ for the indicated time points. g, h Fold induction of pNFκB (g, n ≥ 3 independent replicates per treatment) and Cav3.2 (h, n = 4 independent replicates per treatment) expression levels relative to untreated controls following cytokine treatment for 5 min. i Experimental timeline explaining the treatment of primary astrocytes with IL-1β in the absence or presence of the NF-κB pathway inhibitor, BAY 11-7083. j Representative western blot showing the levels of Cav3.2, p-NF-κB and p-IκB in the absence or presence of IL-1β and BAY 11-7083. k Fold induction of Cav3.2 expression levels following treatment with IL-1β in the absence or presence of BAY 11-7083 for 5 min (*P = 0.0354) and 60 min (**P = 0.0060). Values are estimated relative to untreated from three independent biological replicates. Statistics by unpaired Student’s t test. l-n Representative plots from live calcium imaging of primary astrocytes upon exposure to IL-1β with or without pre-treatment with the Cav3.2 inhibitor, NiCl2. o Quantification of F350/380 peak ratios from primary astrocytes after addition of IL-1β alone (*P = 0.0431 vs control) or IL-1β and NiCl2 (*P = 0.0364 vs IL-1β alone). Statistics were performed by one-way ANOVA followed by Tukey’s multiple comparisons test (n ≥ 12 images per condition in triplicates) |
Induction of Cav3.2 T-type channels induces alterations of astrocyte secretome, promoting the release of the neuroprotective protein IGFBPL1
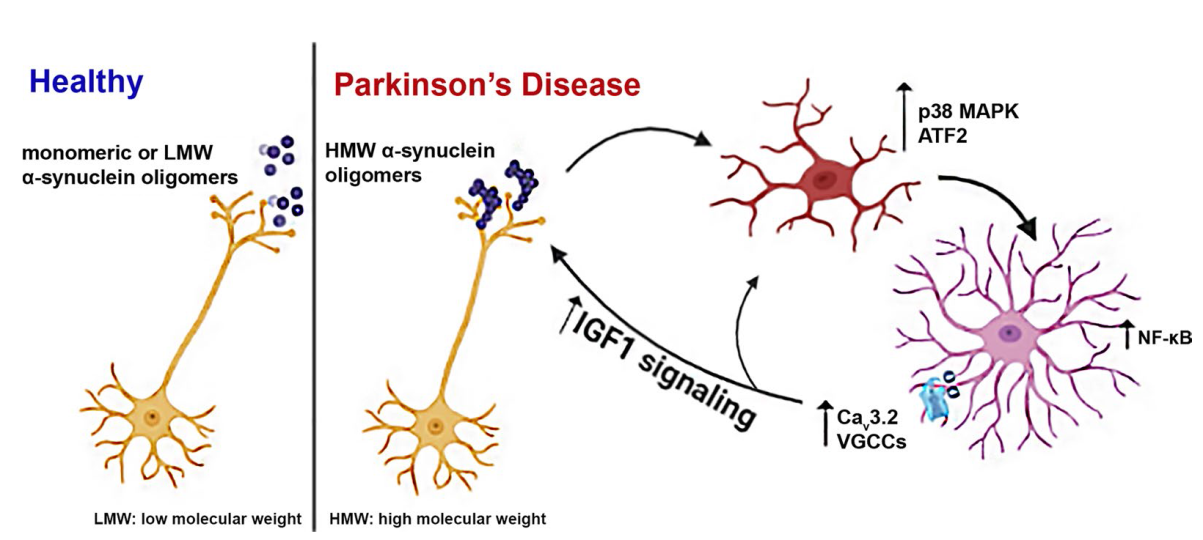
Fig. 7 The induction of Cav3.2 VGCCs alters astrocyte secretome promoting the release of the neuroprotective protein IGFBPL1. a Representative immunoblot of mock- and a1H-transfected primary quiescent astrocytes using an antibody against Cav3.2. b Volcano plot showing the up- and down-regulated proteins identified by SignalP as secreted proteins identified by the LC-MS/MS analysis from the CM of mock- and a1H-transfected astrocytes (n = 3 independent replicates). c List of the top ten pathways as classified by Enrich-r. d List of the top five secreted proteins identified in the CM of astrocytes after a1H overexpression. e Immunoblot from mock and a1H-transfected astrocytes using an antibody against IGFBPL1 and densitometric quantification (n = 5 per condition, *P = 0.0203). f Representative confocal images of striatal sections co-stained with antibodies against IGFBPL1 and GFAP. DAPI (blue) was used to stain nuclei. Scale bar: 50 μm. g Quantification of mouse CXCL-10 levels in striatal homogenates (n ≥ 5, P = 0.1864). h Proposed mechanism through which α-synuclein oligomers activate microglia and astrocytes and induce Cav3.2 channels that mediate IGFBPL1 secretion. GAPDH was used as a loading control. In e, g, statistics were performed by Unpaired Student’s t test |
Human PD brains are also characterized by α-synuclein oligomers, C3 complement activation and increased Cav3.2 mRNA levels
Fig. 8 Human PD brain is characterized by α-synuclein oligomers, C3 complement activation and altered Cav3.2 mRNA levels. a Comparison of the SNCA mRNA levels in the caudate nucleus (n = 8 per group, P = 0.6926) and putamen tissue (n = 7 Non-PD and n = 8 PD, P = 0.2917) of PD vs non-PD subjects. b Representative western blots depicting oligomeric and monomeric α-synuclein in the two brain areas. c Densitometric quantifications of monomeric α-synuclein in the caudate nucleus, P = 0.8613 and putamen, P = 0.4783 (n = 8 per group per brain area). d Representative western blots depicting phosphorylated oligomeric and monomeric α-synuclein in the two brain areas. e qPCR assessment of C3 mRNA in caudate nucleus, *P = 0.0373 and putamen, *P = 0.0140 (n = 8 per group, per brain area). f Representative western blots showing the levels of C3a in caudate nucleus (n = 7 non-PD and n = 8 PD, *P = 0.0168) and putamen (n = 8 per group, *P = 0.0651). g qPCR assessment of CACNA1H mRNA in caudate nucleus (n = 7 Non-PD and n = 8 PD, P = 0.2184) and putamen (n = 7 per group, **P = 0.0044). In all western blots, GAPDH was used as a loading control. All statistics were performed by unpaired Student’s t test |