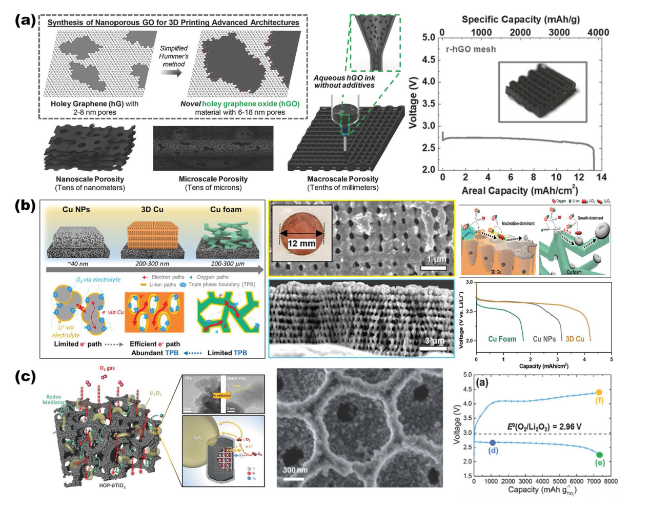
Highlights
1 Introduction
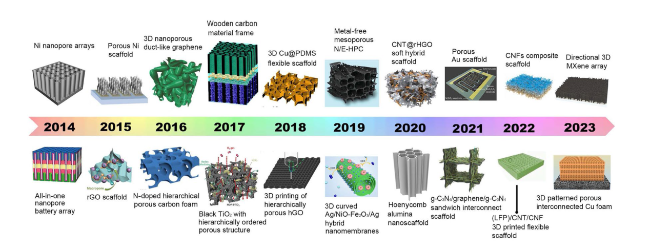
Fig. 1 Brief development timeline featuring representative 3D NMS scaffolds for SCs, AIBs, metal batteries, and MABs spanning the past decade. Inset images: Ni nanopore arrays. Reproduced with permission from Ref. [27]. Copyright 2014, Wiley-VCH; All-in-one nanopore battery array. Reproduced with permission from Ref. [28]. Copyright 2014, Springer Nature; Porous Ni scaffold. Reproduced with permission [29]. Copyright 2015, National Academy of Sciences; rGO scaffold. Reproduced with permission from Ref. [30]. Copyright 2015, Wiley-VCH; 3D nanoporous duct-like graphene. Reproduced with permission from Ref. [31]. Copyright 2016, Wiley-VCH; N-doped hierarchically porous carbon foam. Reproduced with permission from Ref. [32]. Copyright 2016, Elsevier; Wooden carbon material frame. Reproduced with permission from Ref. [33]. Copyright 2017, Royal Society of Chemistry; Black TiO2 with hierarchically ordered porous structure (Reproduced with permission from Ref. [34]. Copyright 2017, Wiley-VCH; 3D Cu@PDMS flexible scaffold. Reproduced with permission from Ref. [35]. Copyright 2018, Springer Nature; 3D printing of hierarchically porous hGO. Reproduced with permission from Ref. [21]. Copyright 2018, Wiley-VCH; Metal-free mesoporous N/E-HPC. Reproduced with permission from Ref. [36]. Copyright 2019, Wiley-VCH; 3D curved Ag/NiO-Fe2O3/Ag hybrid nanomembranes. Reproduced with permission from Ref. [37]. Copyright 2019, Elsevier; CNT@rHGO soft hybrid scaffold. Reproduced with permission from Ref. [38]. Copyright 2019, Wiley-VCH; Honeycomb alumina nanoscaffold. Reproduced with permission from Ref. [18]. Copyright 2020, Wiley-VCH; Porous Au scaffold. Reproduced with permission from Ref. [39]. Copyright 2021, American Chemical Society; g-C3N4/graphene/g-C3N4 sandwich interconnect scaffold. Reproduced with permission from Ref. [40]. Copyright 2021, Wiley-VCH; CNFs composite scaffold. Reproduced with permission from Ref. [41]. Copyright 2022, Elsevier; (LFP)/CNT/CNF 3D printed flexible scaffold. Reproduced with permission from Ref. [42]. Copyright 2022, Wiley-VCH; Directional 3D MXene array. Reproduced with permission from Ref. [43]. Copyright 2023, Wiley-VCH; 3D patterned porous interconnected Cu foam. Reproduced with permission from Ref. [44]. Copyright 2023, Springer Nature |
2 Fundamentals and Superiorities of NMS Scaffolds in EES
2.1 Fundamentals of NMS Scaffolds
Fig. 2 Schematic illustration of updated 3D interconnection NMS scaffolds with superiorities for high-performance EES devices over the past decades |
2.2 Superiorities of NMS Scaffolds in EES
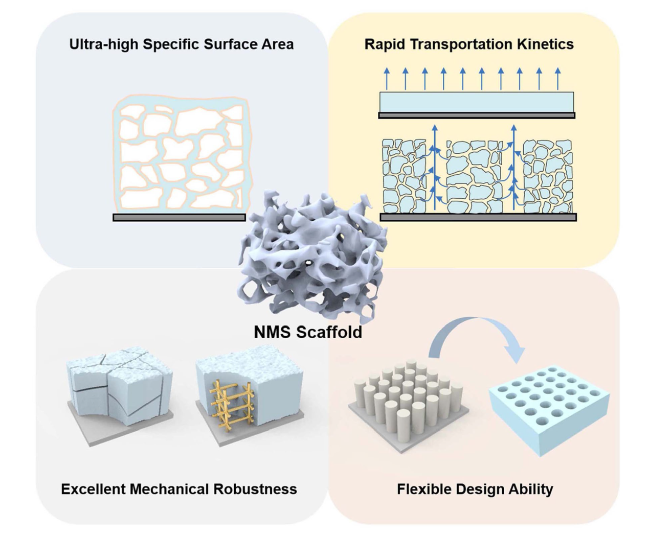
Fig. 3 Schematic illustration of the advantages of 3D NMS scaffolds in terms of chemical reaction, ion transport, mechanical robustness, and design ability |
2.2.1 Ultra-high Specific Surface Area
2.2.2 Rapid Transportation Kinetics
2.2.3 Excellent Mechanical Robustness
2.2.4 Flexible Design Ability
3 Classification and Preparation of NMS Scaffolds
Fig. 4 Classification and typical structure of NMS scaffolds. Inset images: single-wall carbon nanotubes (SWCNTs).Reproduced with permission from Ref. [59]. Copyright 2023, Wiley-VCH; MoS2-rGO. Reproduced with permission from Ref. [60]. Copyright 2017, Wiley-VCH; Carbon cloth. Reproduced with permission from Ref. [61]. Copyright 2017, Springer Nature; Porous Cu-Zn. Reproduced with permission from Ref. [62]. Copyright 2015, Wiley‐VCH; Porous mixed ion- and electron-conducting garnet (MIEC). Reproduced with permission from Ref. [63]. Copyright 2023, Springer Nature; Mesoporous SnO2. Reproduced with permission from Ref. [64]. Copyright 2017, Royal Society of Chemistry; Wood derived carbon. Reproduced with permission from Ref. [36]. Copyright 2019, Wiley-VCH; 3D carbon lattices. Reproduced with permission from Ref. [65]. Copyright 2022, Wiley-VCH; Alumina. Reproduced with permission from Ref. [18]. Copyright 2020, Springer Nature |
3.1 Flexible NMS Scaffolds
3.2 Hard Stochastic NMS Scaffolds
3.3 Hard Periodic NMS Scaffolds
4 Updated NMS Scaffolds for Advanced EES
4.1 Updated NMS Scaffolds for Supercapacitors
Table 1 Electrochemical performance of SCs & MSCs based on NMS scaffolds |
| Electrode materials | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| 3D scaffolds | Active materials | Main techniques | Electrode capacitance (mF cm−2) | Peak energy (μWh cm−2) | Peak power (mW cm−2) | Electrolyte/cell voltage | Lifespan | Refs | |
| SCs based on NMS scaffolds | |||||||||
| CNTs/rHGO | FeOOH//MnO2 | Vacuum filtration | 6000 | 1050 | 17,200 | PVA LiCl/1.6 V | 90.9% (3000) | [15] | |
| rGO | Ni-Co-O//MnO2 | 3D print, Freeze-drying | 327(Ni-Co-O)//69(MnO2) | 90 | - | PVA KOH/1.3 V | 89.9% (10,000) | [95] | |
| Si microtubes | MnO2 | PL, Bosch dry etching, ALD, ED | 1500 | 100 | 7.1 | 5 M LiNO3/1 V | 82% (10,000) | [46] | |
| Si microtubes | MnO2 | PL, Bosch dry etching, ALD, ED | 1500 | 60 | 10 | 5 M LiNO3/1 V | 84% (10,000) | [46] | |
| Porous GA/COF | - | Freeze-drying, Polycondensation-termination | 289 (F g−1) | 25.3 (Wh kg−1) | 40,000 (W kg−1) | 1 M Na2SO4/0.8 V | 92% (5000) | [100] | |
| Natural wood | Ti3C2-Ni | Slicing, Quickly frozen, Freeze-drying | 930 | 23 | - | PVA H2SO4/0.6 V | 87% (5000) | [13] | |
| Wood carbon | MnO2 | Carbonization, ED | 4155 | 1600 | 24 (W cm−2) | PVA LiCl/1.8 V | 93% (10,000) | [33] | |
| wood carbon | - | Enzymolysis, Carbonization | 8410 | 210 | 500 | 6.0 M KOH/1.0 V | 86.58% (15,000) | [83] | |
| rGO framework | MnCO3 | Immerse, LbL, Hydrothermal | 320 (F g−1) | - | - | - | 77% (5000) | [101] | |
| LSG | MnO2 | Laser scribing, ED | - | 42 (Wh L−1) | - | 1 M Na2SO4/2.0 V | 96% (10,000) | [102] | |
| GF-CNT | Fe2O3//CoMoO4 | CVD, ALD | 370.2(Fe2O3) | ~ 74.7 (Wh kg−1) | ~ 11.2 (kW kg−1) | 2 M KOH/1.2 V | 95.4% (50,000) | [86] | |
| Graphene aerogel | PPY | 3D print, Freeze-drying | 2000 | 110 | - | 2 M KOH/0.8 V | 75% (5000) | [96] | |
| - | Graphene/MWCNT | 3D print, Freeze-drying | - | 53 | 1.54 | 6 M KOH/0.8 V | 90% (10,000) | [97] | |
| Graphite foam | NiCo2O4//NC | 3D print, CVD | ~ 3.2 (F cm3) | 36.9 (Wh kg−1) | 20 (kW kg−1) | PVA KOH/1.6 V | 87.8% (10,000) | [103] | |
| 3D graphene networks | PANI | Solvothermal self-assembly | 188 (F g−1) | - | - | PVA H2SO4 HQ/0.8 V | 82% (100,000) | [104] | |
| Polymer | rGO | 3D print, Freeze-drying, ED | 293.4 | 8 | 12.56 | PVA KOH/1 V | 96% (5000) | [16] | |
| MSCs based on NMS scaffolds | |||||||||
| Si | GNW/RuOx | DRIE, MPECVD | - | 15.1 | 2.49 (mW cm−2) | PVA H3PO4/1 V | 90.2% (3000) | [105] | |
| AuNPs | δ-MnO2 | Chemical dealloyed, ED | 922 (F cm−3) | 24.3 (mWh cm−3) | 295 (W cm−3) | 1 M Na2SO4/0.8 V | 88% (20,000) | [106] | |
| SnO2NT | PPY//MnO2 | AAO, ALD, ED | 260 (F g−1)//910 (F g−1) | 27.2 (Wh kg−1) | 24.8 (kW kg−1) | 1 M Na2SO4/1.7 V | 80% (2000) | [107] | |
| Porous Au | RuO2.xH2O | HBDT, ED | 3250 | 126 | 7.9 (mW cm−2) | PVA H3PO4-H4SiW12O40/0.9 V | 95% (2000) | [62] | |
| Porous Au | RuOxNySz | HBDT, ED | 14,300 | 432 (mJ cm−2) | 421 (mW cm−2) | PVA H3PO4-H4SiW12O40/0.9 V | 100% (5000) | [39] | |
| PEDOT:PSS | Mxene | 3D print, LbL | - | 80 | 3.8 | PVA-H2SO4/0.8 V | 83% (6000) | [108] | |
| Ni nanopore arrays | MnO2 | AAO, ED | 570 (F g−1) | - | - | 1 M Na2SO4/0.8 V | 83% (3000) | [27] | |
| FTO/AAO | MnO2 | AAO, USP, ED | 87.4 | 1.56 (mWh cm−3) | - | 1 M Na2SO4/0.8 V | 81.6% (3000) | [98] | |
| FTO/AAO | MnO2 | AAO, USP, ED | 540 | 2.36 (mWh cm−3) | 4.45 (Wh cm−3) | 1 M Na2SO4/0.8 V | 85.5% (5000) | [99] | |
| SnO2/AAO | PPY//MnO2 | AAO, ALD, ED | - | 160 | 40 (mW cm−2) | EMIM-TFSI/3 V | 82.5% (10,000) | [18] | |
| Exfoliated graphite | PPY/MoOx//Na0.5MnO2 | Electrochemical exfoliation, ED, LbL | 398 (F g−1) | 72.7 (Wh kg−1) | 343 (W kg−1) | 5 M LiCl/2.2 V | 92.3% (10,000) | [109] | |
| CuSe | Ni(OH)2 | ED | 5.4 (mWh cm−3) | 833.2 (mW cm−3) | PVA LiCl/1 V | 100% (10,000) | [110] | ||
| CNW | Hydrous-RuO2 | PECVD, ED | 1094 | 49 | 31.3 | PVA H3PO4-H4SiW12O40/0.9 V | ~ 90% (2000) | [111] | |
| GNW | Ni(OH)2 | MPECVD, EB, Electrochemical oxidation | 33.6 | 2.1 (mWh cm−3) | 5.91 (W cm−3) | - | 96% (2000) | [112] | |
| Porous carbon | - | MIL, ALD | 3.61 | 1.3 | PVA/H3PO4 /1 V | 97% (30,000) | [113] | ||
| GF | PPY | Laser induction | 2412.2 | 134.4 | 6.5 | PVA H2SO4/1.3 V | 95.6% (10,000) | [114] | |
| Au pillars | GMP | Inkjet print, LbL | 10 | ~ 1 | - | PVA H2SO4/0.8 V | 80% (2000) | [115] | |
| Graphene aerogel | MnO2 | 3D print, ED | 44,130 | 1560 | - | 3 M LiCl/2.2 V | 92.9% (20,000) | [116] | |
| Mxene-AgNW-MnONW-C60 | - | 3D print, LbL, Unidirectional freezing, Freeze-drying | - | 19.2 | 58.3 (mW cm−2) | PVA KOH | - | [14] | |
PEDOT:PSS, poly(3,4-ethylenedioxythiophene):polystyrene sulfonate; DRIE, deep reactive ion etching; PL, photolithography; MIL, multibeam interference lithography; ALD, atomic layer deposition; ED, electrodeposition; GNW, graphene nanowalls; HBDT, dynamic hydrogen bubble template; USP, ultrasonic spray pyrolysis; FTO, fluorine-doped tin oxide; EMIM-TFSI, 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide; PANI, polymer polyaniline; GMP, graphene/MnO2/PANI; CNW, carbon nanowalls; EB, electron beam; LSG, Laser-scribed graphene; PPY, polypyrrole; GF, graphene framework; SLM, selective laser melting; CVD, chemical vapor deposition; NC, N-doped carbon nanosheet arrays |
4.1.1 NMS Scaffold Designs for SCs
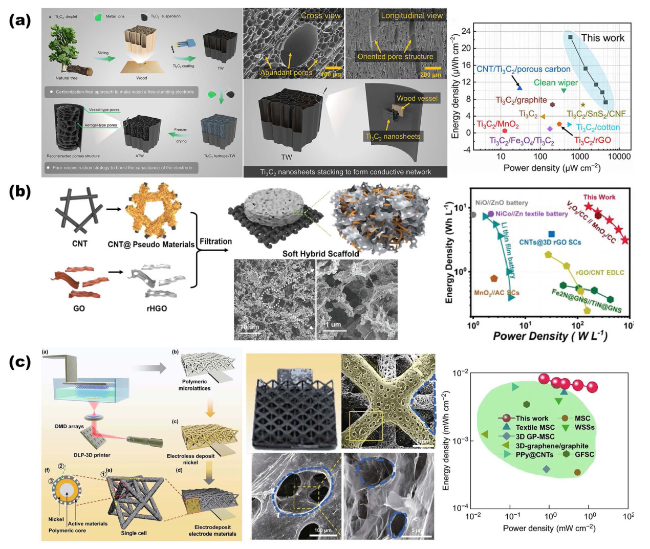
Fig. 5 Overview of representative electrodes for SCs based on NMS scaffolds. a The ultimate 3D continuous network structure is based on porous wood NMS scaffold. Reproduced with permission from Ref. [13]. Copyright 2023, Wiley‐VCH. b Soft hybrid scaffolds consist of CNTs and rHGOs for wearable SCs. Reproduced with permission from Ref. [38]. Copyright 2020, Wiley‐VCH. c An octet-truss lattice polymer scaffold was synthesized via acrylate-based UV photosensitive resin. Reproduced with permission from Ref. [16]. Copyright 2019, Springer Nature |
Fig. 6 Overview of representative electrodes for MSCs based on NMS scaffolds. a Schematic illustration of porous Cu-Zn NMS scaffold fabricated via DHBT method. Reproduced with permission from Ref. [94]. Copyright 2018, Wiley‐VCH. b SEM images of the porous Au NMS scaffold. Reproduced with permission from Ref. [62]. Copyright 2015, Wiley‐VCH. c Electrode based on porous Au NMS scaffold with RuOxNySz as active material for MSCs. Reproduced with permission from Ref. [39]. Copyright 2021, American Chemical Society. d Honeycomb-like porous scaffold with fine gel (MXene‐AgNW‐MnONW‐C60) endowed by 3D print and freeze-drying techniques. Reproduced with permission from Ref. [14]. Copyright 2020, Wiley‐VCH. e Honeycomb alumina as stiff NMS scaffold with only 16 ± 2 nm cell‐wall for MSCs. Reproduced with permission from Ref. [18]. Copyright 2020, Springer Nature |
4.1.2 NMS Scaffold Designs for MSCs
4.2 Updated NMS Scaffolds for AIBs
Table 2 Electrochemical performance of AIBs & AIMBs based on NMS scaffolds |
| Electrode materials | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| 3D scaffolds | Active materials | Main techniques | Capacity (mAh cm−2) | Peak energy (mWh cm−2) | Peak power (mW cm−2) | Electrolyte | Lifespan | Refs | |
| Bs based on NMS scaffolds | |||||||||
| Porous carbon | ZnFe2O4 | Carbonization | 711 (mAh g−1) | - | - | LiPF6/EC/DEC | ≈100% (400) | [122] | |
| Porous Cu-Zn | - | HBDT, ED | ≈300 (mAh g−1) | - | - | LiPF6/EC/DEC | 83.3% (500) | [94] | |
| Carbon fibers | LiNi0.5Mn0.3Co0.2O2 | CVD | 1500 | ≈380 (Wh kg−1) | - | LiPF6/EC/EMC | 90.4% (500) | [87] | |
| CNTs/CNFs | LFP//LTO | 3D print, Freeze-drying, | 8.4 | 15.2 | 75.9 | LiPF6/EC/DEC | 84% (10,000) | [42] | |
| Porous carbon | LFP//Ge-N | PS template | 942 (mAh g−1) | - | - | LiPF6/EC/DEC/VC | 90% (200) | [123] | |
| CB/CNF network | LFP | Self-assembly, Freeze-drying | 8.8 | 538 (Wh L−1) | - | LiPF6/EC/DEC | 91% (150) | [120] | |
| NPC | - | Self-template | - | - | - | KPF6/EC/DEC | - | [15] | |
| 3D graphene | Fe2O3 | Self-assembly, Freeze-drying, Ostwald ripening | - | - | - | LiPF6/EC/DMC | - | [124] | |
| 3D Ti3C2Tx | LFP//Ga-In | Self-assembly | 132 (mAh g−1) | - | - | LiPF6/EC/DEC/FEC | 90.2% (150) | [125] | |
| 3D porous Bi | Na3V2(PO4)3//3D porous Bi | Liquid phase reduction | 306 (mAh g−1) | 116 (Wh kg−1) | - | Na2O | - | [126] | |
| 3D MXene | V2O5 | Ice template | - | - | - | LiPF6/EC/DEC | - | [127] | |
| 3D honeycomb-like carbon grafted | PB// 3D honeycomb-like carbon grafted | Freeze-drying, Carbonization | - | 113 (Wh kg−1) | - | KFSI/DME | - | [128] | |
| Na3V2(PO4)3 | Na3V2(PO4)3//Hard carbon | Phase separation | 90 (mAh g−1) | - | - | NaClO4/EC/DEC/FEC | 99% (100) | [129] | |
| Vertically aligned graphene | LiNi0.6Co0.2Mn0.2O2//Red-phosphorus | Silica template, Coassembly | 5.6 | 405 (Wh kg−1) | 1224 (W kg−1) | LiPF6/EC/DMC/FEC | 76.6% (300) | [130] | |
| AIMBs based on NMS scaffolds | |||||||||
| Nanoporous Au | KxMnO2·nH2O//KxV2O5·nH2O | Chemically dealloying, ED | ~ 64 (mAh cm−3) | ~ 103 (mWh cm−3) | ~ 600 (W cm−3) | 0.5 M K2SO4 | 80% (10,000) | [121] | |
| Porous Ni | LiMnO2//Sn | 3D HL, PL | - | 6.5 (μWh cm−2 μm−1) | 3600 (μW cm−2 μm−1) | LiClO4/EC/DEC | 80% (100) | [29] | |
| Porous Ni | V2O5//Li | ILP, Self-assembly, ED | - | 1.242 (J cm−2) | 75.5 | PEO/LiTFSI/DOL/DME | 75% (200) | ||
| Porous Ni | PIN | PL, ED, Hydrothermal | - | - | - | LiPF6/EC/DEC | - | [131] | |
| Porous Ni | LiMnO2//Sn | 3D HL, PL | - | 15 (mWh cm−2 μm−1) | 7.4 (mW cm−3 μm−1) | LiClO4/EC/DEC | 64% (15) | [132] | |
| Porous Ni | Ni(OH)2//Zn | Colloidal templating, ED, facile anodizing | 150.1 (μAh cm−2) | 0.26 | 33.8 | PVA-KOH/ZnO | 74.6% (1800) | [133] | |
| 3D Ni5P4 nanofoams | Li | Virus-template | 677 (mAh cm−3) | - | - | LiPF6/EC/DEC | 80% (100) | [134] | |
| Porous Ni | MnO2 | PL, PS template, ED | - | 45.5 (mWh cm−2 μm−1) | 5300 (μW cm−2 μm−1) | LiClO4/EC/DMC | - | [135] | |
| AAO | V2O5 | AAO, ALD | - | 0.6 (μWh cm−2 μm−1) | 0.49 (μW cm−2 μm−1) | LiPF6/EC/DEC | 81% (1000) | [28] | |
| Porous Ni | NH4CuHCF-PEDOT//Zn | ED, Coating | - | 0.31 | 80.83 | (HN4)2SO4/Zn(CF3SO3)2 | - | [136] | |
| Porous Ni | NH4CuHCF-PEDOT//Zn | ED, Coating | - | 0.33 | 83.12 | (HN4)2SO4/CuSO4/guar gum | 94% (1000) | [136] | |
DEC, diethyl carbonate; EC, ethylene carbonate; EMC, ethyl methyl carbonate; PEO, poly(ethylene oxide); VC, vinylene carbonate; DMC, dimethyl carbonate; FEC, ethylene fluorocarbonate; HBDT, dynamic hydrogen bubble template; ED, electrodeposition; LFP, lithium iron phosphate; LTO, lithium titanate; PS, polystyrene colloidal nanospheres; CB, carbon black; NPC, nitrogen-doped porous carbon; PL, photolithography; HL, holographic lithography; ILP, imprint lithography; RIE, reactive ion etching; PIN, Polyimide-based nanoparticles; EMIM-TFSI, 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide; PB, Prussian blue |
4.2.1 NMS Scaffold Designs for AIBs
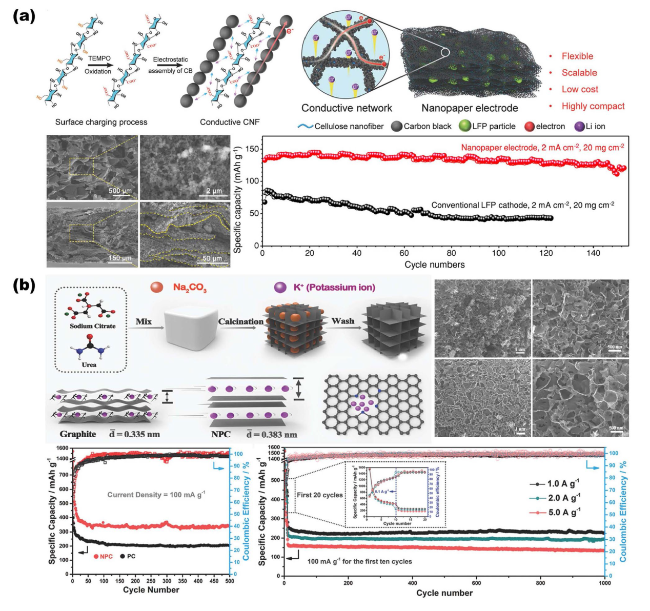
Fig. 7 Overview of representative NMS scaffolds for AIBs. a Interconnected 3D CNF NMS scaffold with tightly wrapped LFP as close-packed nanopaper electrode for Li-ion batteries. Reproduced with permission from Ref. [120]. Copyright 2018, Wiley‐VCH. b Porous carbon synthesized via a self-template technique as both an electrode and a scaffold for K-ion batteries. Reproduced with permission from Ref. [15]. Copyright 2018, Wiley‐VCH |
4.2.2 NMS Scaffold Designs for AIMBs
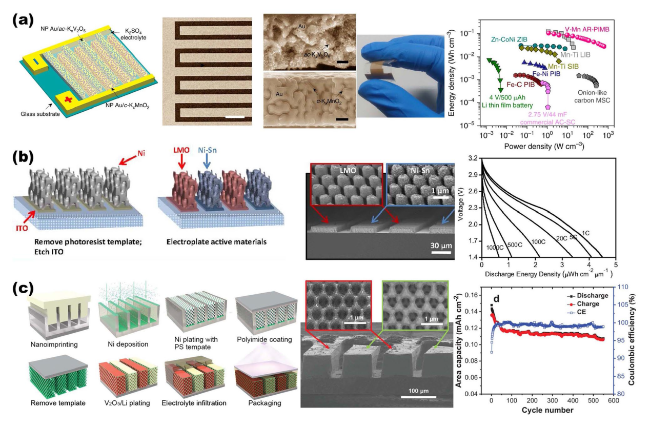
Fig. 8 Overview of representative NMS scaffolds for AIMBs. a Porous Au simultaneously acts both as micro-current collectors and NMS scaffolds, integrating the c-KxMnO2 cathode and ac-KxV2O5 anode for Li-ion micro-batteries. Reproduced with permission from Ref. [121]. Copyright 2019, Springer Nature. b 3D Ni holographic scaffold, integrating the NiSn anode and LiMnO2 cathode for Li-ion micro-batteries. Reproduced with permission [29]. Copyright 2015, National Academy of Sciences. c Interconnected Ni porous NMS scaffold, integrating the Li metal anode and V2O5 cathode for Li-ion micro-batteries. Reproduced with permission [71]. Copyright 2021, Wiley‐VCH |
4.3 Updated NMS Scaffolds for Anode of Metal Batteries
4.3.1 Advanced NMS Scaffolds as Current Collector of Metal Anodes
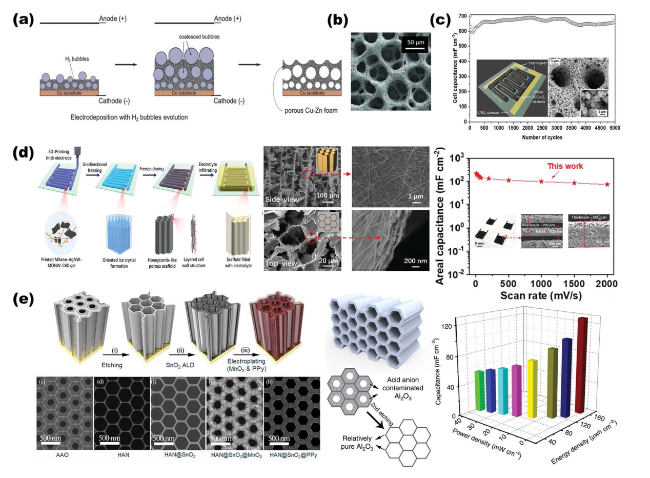
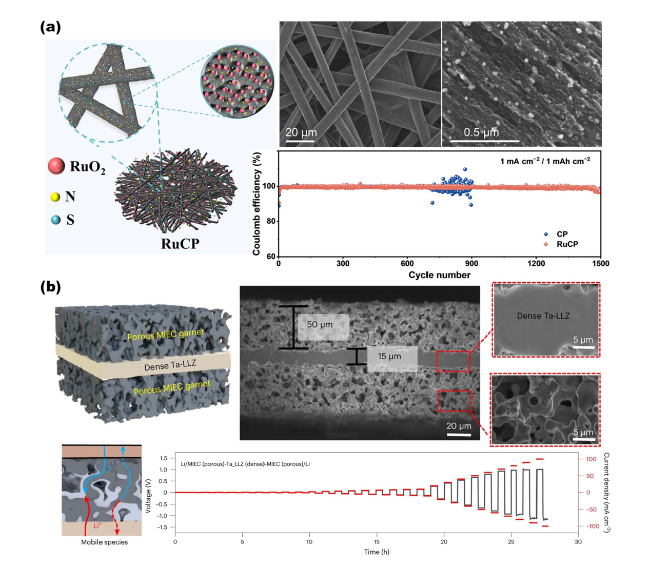
Fig. 9 Overview of representative NMS scaffolds as anode current collectors for metal batteries. a RuCP scaffolds composited from carbon paper and RuO2 nanoparticles for Na-CO2 battery anodes. Reproduced with permission from Ref. [150]. Copyright 2023, Wiley-VCH. b Single-phase MIEC as an anode collector for Li-metal batteries. Reproduced with permission from Ref. [63]. Copyright 2023, Springer Nature |
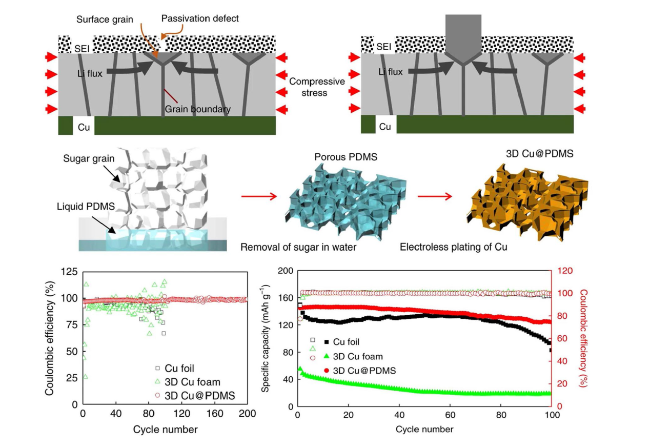
Fig. 10 Modeling of internal stress-driven dendrite generation and design of flexible 3D Cu@PDMS scaffolds for Li-metal anode collectors. Reproduced with permission from Ref. [35]. Copyright 2018, Springer Nature |
4.3.2 Advanced NMS Scaffolds as SEI Layer of Metal Anodes
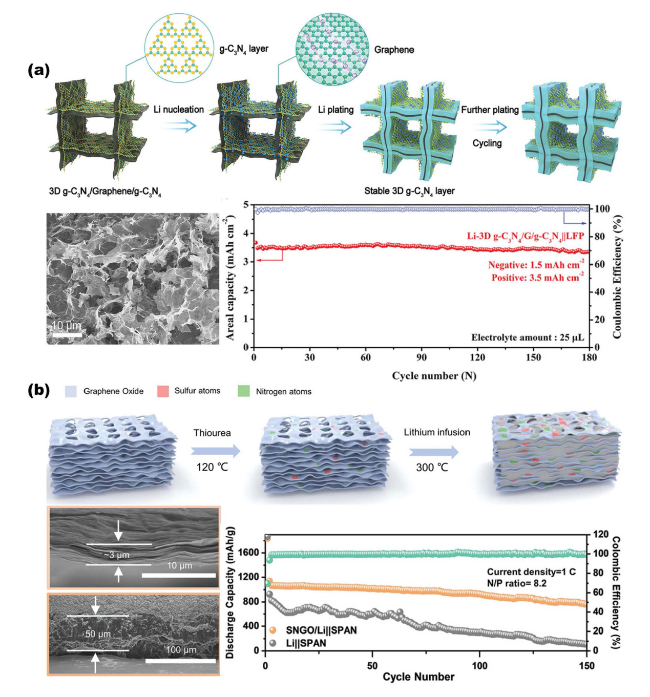
Fig. 11 Overview of the SEI layer for metal battery anodes based on NMS scaffolds. a 3D sandwich structure composite scaffold composed of g-C3N4/graphene/g-C3N4 for Li metal battery anode. Reproduced with permission from Ref. [40]. Copyright 2021, Wiley-VCH. b The SNGO framework possesses Li3N-Li2S SEI layers for improving Li metal anode performance. Reproduced with permission from Ref. [20]. Copyright 2022, Wiley-VCH |
Table 3 Electrochemical performance of metal batteries anode based on NMS scaffolds |
| Functionality | Anode metal | Scaffolds | Main techniques | Half cells (current density (mA cm−2), areal capacity (mAh cm−2), cycle umber) | Symmetric cell (current density (mA cm−2), areal capacity (mAh cm−2), life span (h)) | Full cells (cathode, rate performance, and life span) | Refs |
|---|---|---|---|---|---|---|---|
| Current collector | Li | MIEC garnet | Scalable tape-casting process | - | 1, 1, 50 & 5, 5, 50 | NMC622, 1C, 500 cycles | [63] |
| Li | Cu-coated carbon fabrics | Polymer-assisted metal deposition | 1, 3.5, 90 | 1, 2, 500 | NSHG/S8/NiCF, 1 (mA cm−2), 260 cycles | [151] | |
| Li | Cu/Ni core-shell network | Hydrothermal and sintering | 2, 1, 100 | 3, 1, 208.3 | LiCoO2, 1C, 200 cycles | [152] | |
| Li | 3D Cu&CuAux matrix | High energy heavy iontracking | - | 1, 1, 2160 | LiFePO4,10 C, 200 cycles | [153] | |
| Li | 3D porous CuZn | Dealloying | 0.5, 0.5, > 800 | 0.5,1, > 500 | NCM811, 1 C, 500 cycles | [154] | |
| Li | 3D printed porous Cu | 3D printing | 1, 5, > 450 | 1, 2.5, 450 | - | [155] | |
| Li | P-Cu@Cu6Sn5 | Electroless plating | 1, 1, 325 | 1, 1, > 1000 | LiFePO4, 1 C, 600 cycles | [156] | |
| Zn | 3D Ti3C2Tx nanosheets | Freeze-drying | 5, -, 150 | 1, 1, 1500 | VO2, 5 A g-1, 1000 cycles | [43] | |
| Zn | 3D Ti-TiO2 | Template-free electrodeposition and vapor dealloying | 5, 1, 120 | 1, 1, 2000 | S-MXene@MnO2, 5 (A g−1), 500 cycles | [157] | |
| Zn | 3DCEP-MXene | 3D cold-trap environment printing | 1, -, 450 | 0.25, 0.1, 1400 | 3DCEP-MXene/Co-MnHCF, 0.9 C, 1600 cycles | [158] | |
| Na | 3DHS with Mg clusters | Solution mixed and carbonization | - | 0.5, 1, 450 | FeS2, 0.5, 50 cycles | [159] | |
| Na | 3D Zn@Al | Magnetron sputtering | 0.5, 0.5, 1200 | 2, 1, 1500 | NVP, 5 C, 2000 cycles | [72] | |
| Na | SnO2-CNFs | Carbonizing PAN nanofibers and magnetron sputtering | 3, 3, 1500 | 1, 1, 3000 | NVP@C@CNTs, 1 C, 130 cycles | [41] | |
| K | Co/NOC/CNM | Solution mixed annealing | 0.5, 0.5, 110 | 1, 1, 1000 | PTCDA, 10 C, 100 cycles | [160] | |
| Current collector & SEI layer | Li | g-C3N4/graphene/g-C3N4 | 3D self-assembly and in-situ calcination | 1, 1, 500 | - | LiFePO4, 0.3 C, 180 cycles | [40] |
| Li | 3D Cu2S NWs-Cu foam | Anodizing and sulfuration | 1, 1, 500 | - | LiFePO4, 0.5 C, 300 cycles | [161] | |
| SEI layer | Li | SNGO | Hydrothermal and 3D printing | - | 1, 1, 450 | NCM811, 0.5 C, 100 cycles | [20] |
| Li | NiFx@NF | One-step fluorination | 1, 1, 450 | 1, 1, 1300 | LiFePO4, 2 C, 500 cycles | [162] | |
| Li | ZnO-PAN-ZnO skeletons | Electrostatic spinning | 0.5, 1, 120 | 3, 1, 200 | LiFePO4, 1 C, 200 cycles | [163] | |
| Li | LiF-rich 3DSF | Toroidal magnetic field | 1, 1, 100 | 1, 1, 1700 | LiFePO4, 1 C, 150 cycles | [164] |
MIEC, mixed ion- and electron-conducting; NMC622, LiNi0.6Mn0.2Co0.2O2; NSHG/S8/NiCF, graphene/sulfur mixture on Ni-coated carbon fabric; NCM811, LiNi0.8Co0.1Mn0.1O2; S-MXene, S-doped MXene; 3DCEP, 3D cold-trap environment printing; 3DHS, 3D hierarchical structure; NVP, Na3V2(PO4)3; NOC, N, O codoped carbon; CNM, carbonaceous nanofiber matrix; PTCDA, perylene-3, 4, 9, 10-tetracarboxylic dianhydride; SNGO, Porous graphene oxide films are doped with sulfur and nitrogen; NF, Ni foam; 3DSF, three-dimensional SEI framework |
4.4 Updated NMS Scaffolds for Cathode of Metal-Air Batteries
Table 4 Electrochemical performance of MABs cathode based on NMS scaffolds |
| Battery type | Scaffolds | Main techniques | Electrolyte | Voltage hysteresis, applied current | Peak power density | Discharge capacity | Cyclability | Refs |
|---|---|---|---|---|---|---|---|---|
| Li-O2 | Graphene oxide aerogel/NiCo2O4 | Electrostatic assembly and freeze-drying | 0.1 M LiTFSI in DOL/DME (1:1) with 1% LiNO3 | ≈1 V, 0.1 (mA cm−2) | 3398.4 (mAh g−1) | 2000 h (400 cycles) at 0.1 (mA cm−2) with a cut-off capacity of 0.25 (mAh cm−2) | [69] | |
| Li-O2 | Carbon frameworks/Co | Extrusion-based 3D printing | 0.5 M LiClO4-DMSO | - | - | 798 (Wh kg−1) | 165 h at a limited capacity of 1 (mA h) and 0.1 (mA cm−2) | [176] |
| Li-O2 | Porous black TiO2 | Hydrogen reduction | 1 M LiN(SO2CF3)2 in TFGDMF and 0.05 M LiI | 0.37 V at 500 (mAh g−1) | 7500 (mAh g−1) | ≈340 cycle at 500 (mA g−1) and 1000 (mAh g−1) | [34] | |
| Li-O2 | Tri-pathway carbon nanotube/Ru | Chemical delignification and coating process | 111 μL cm-2 of 1 m LiTFSI/TEGDME | 0.85 V at 100 (mA g−1) | 7300 (mAh g−1) | 220 cycles with 1000 (mAh g−1) at 200 (mA g−1) | [177] | |
| Li-O2 | Ag/NiO-Fe2O3/Ag | Thermal treatment | 1 M LiCF3SO3/TEGDME | 1.24 V, 100 (mA g−1) | 5138 (mAh g−1) | 180 cycles at 300 (mA g−1) with a cut-off specific capacity of 1000 (mAh g−1) | [37] | |
| Li-O2 | Au/Cu@FCu | Hydrothermal and reduction methods | 1 M LiTFSI in (DOL)/DME (1:1, w/w) with 0.1 M LiNO3 | 0.64 V, 100 (mA g−1) | 27,270 (mAh g−1) | 220 cycles with 100 mA g−1 at a fixed capacity of 500 (mAh g−1) | [178] | |
| Li-O2 | NiCo2O4/CNFs | Hydrothermal method | 1 M LiTFSI/TEGDME | ≈1.7 V, 100 (mA g−1) | 4179 (mAh g−1) | 350 cycles at 200 (mA g−1) with a cut-off specific capacity of 1000 (mAh g−1) | [179] | |
| Li-O2 | Porous hGO meshes | 3D printing | 0.1 LiTFSI in DMSO | < 1 V, 0.1 (mA cm−2) | - | 13.3 (mAh cm−2) or ≈3879 (mAh g−1) | 13 cycles at 0.1 (mA cm−2) | [21] |
| Zn-air | N-doped hierarchically porous carbon plates | Enzyme treatment and subsequent pyrolysis with NH4Cl | 6 M KOH and 0.2 m zinc acetate | ≈0.7 V, 10 (mA cm−2) | 192.7 (mW cm−2) | 801 (mAh g−1) | 110 h at 10 (mA cm−2) | [36] |
| Zn-air | Carbon nanotube network | Oxygen-induced electron density modulation | 6 M KOH and 0.2 M zinc acetate | - | 130.5 (mW cm−2) | - | 340 h GCD test at 5 (mA cm−2) | [180] |
| Zn-air | Fe8Co0.2-NC-800 | Pyrolysis of core-shell microspheres | 6 M K(OH) and 0.2 M Zn(CH3COO)2 | - | 124.9 (mW cm−2) | 704 (mAh gZn−1) | 311 h at 5 (mA cm−2) with discharge for 5 min and charge for 5 min | [181] |
| Zn-air | GH-BGQD | One-step hydrothermal route | 2.0 M KOH | - | 112 (mW cm−2) | 687 (mAh g−1) | 300 cycles at a current density of 5 (mA cm−2) | [182] |
| Zn-air | N-droped carbon nanosheets/Co | Salt-template method | 6 M KOH | 0.63 V | ≈194 (mW cm−2) | ≈690 (mAh gZn−1) | 20 h at 5 (mA cm−2) | [183] |
| Al-air | 3D carbon | Polyaniline-assisted template method | 0.1 M KOH | - | 130.5 (mW cm−2) | - | discharge under 1.27 V for 30 h | [184] |
| Al-air | Defect-rich MnO2 NWs networks | Ar plasma approach | 4 M NaOH | - | 159 (mW cm−2) | - | 6 h at a current density of 100 (mA cm−2) | [185] |
| Al-air | N-doped carbon nanosheets | Thermally converted | 4 M KOH | - | 130 (mW cm−2) | 552 (mAh g−1) | 11 h at a current density of 5 (mA cm−2) | [186] |
| Mg-air | Co/nitrogen-doped CNTs | Hydrothermal method and calcination | 6 M NaOH | - | 58.6 (mW cm−2) | - | 5000 s at current density of 10 (mA cm−2) | [187] |
| Mg-air | Amorphous-MnO2/CNTs-OH | Direct reaction method and heat-treated | 15 wt% NaCl | - | 79.2 (mW cm−2) | - | 15 h at a current density of 50 (mA cm−2) | [188] |
LiTFSI, Lithium bis(trifluoromethylsulfonyl)imide; DOL, 1,3-dioxolan; DME, 1,2-dimethoxyethan; DMSO, dimethyl sulfoxide; TEGDME, tetraethylene glycol dimethylether; GH-BGQD, B-doped graphene quantum dots anchored on a graphene hydrogel; NWs, nanowires; CNTs, carbon nanotubes |
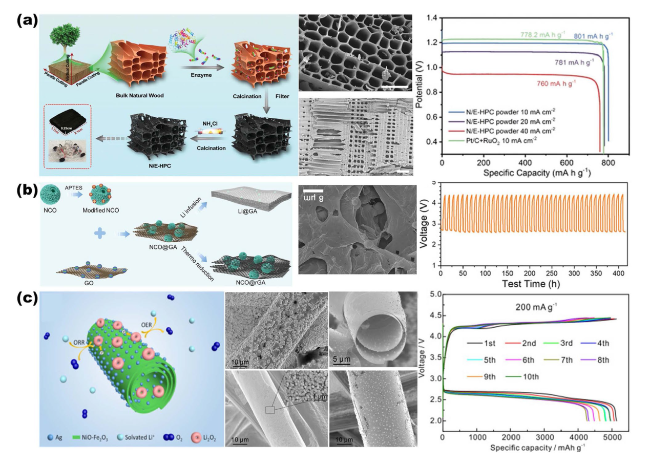
Fig. 12 a Preparation of N-doped carbon porous scaffolds from virgin wood for Zn-air battery cathodes. Reproduced with permission from Ref. [36]. Copyright 2019, Wiley-VCH. b NiCo2O4 microspheres embedded into 3D graphene oxide aerogel (NCO@rGA) for Li-O2 battery air electrode. Reproduced with permission from Ref. [69]. Copyright 2020, Wiley-VCH. c Ag/NiO-Fe2O3/Ag (Ag/NFO/Ag) hybridized micrometer tube porous structure for Li-O2 air cathode. Reproduced with permission from Ref. [37]. Copyright 2018, Elsevier |
Fig. 13 a 3D printed graphene oxide hierarchical porous structure scaffolds for air cathodes. Reproduced with permission from Ref. [21]. Copyright 2018, Wiley-VCH. b Photolithographically prepared 3D ordered porous Cu electrodes for Li-O2 battery cathodes. Reproduced with permission from Ref. [44]. Copyright 2023, Wiley-VCH. c Ordered macro/mesoporous structure of anoxic black TiO2 scaffolds and its use as air cathode for growth kinetic modulation of Li2O2. Reproduced with permission from Ref. [34]. Copyright 2017, Wiley-VCH |
5 Summary and Perspectives
Fig. 14 Development overview of NMS scaffolds through integration of advanced design, manufacturing, characterization, and simulation technologies |