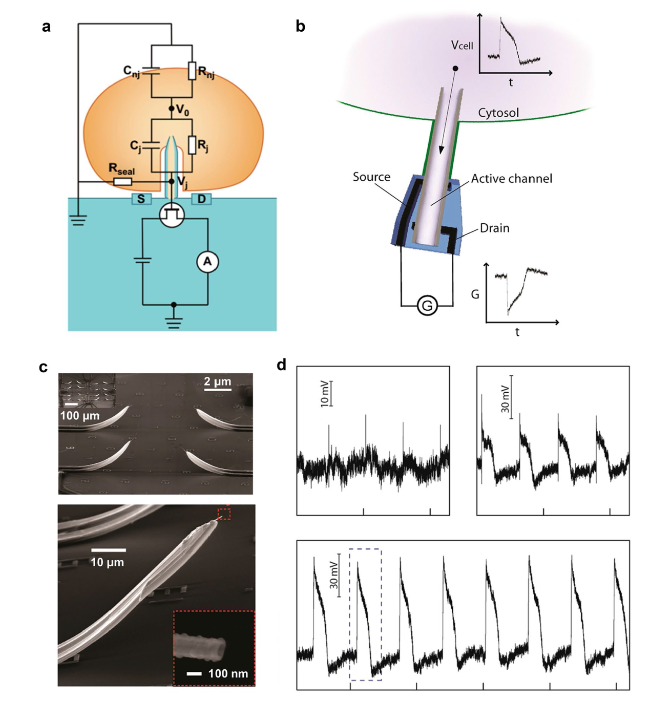
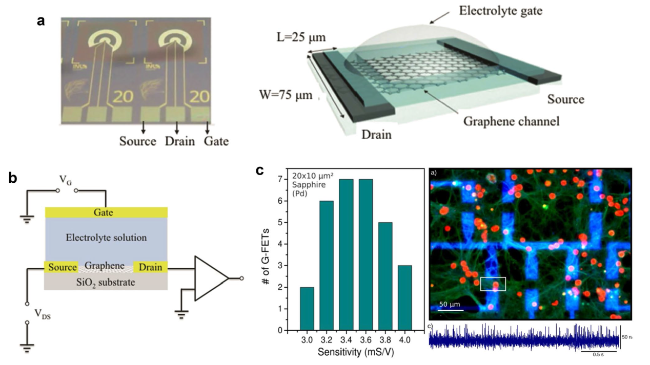
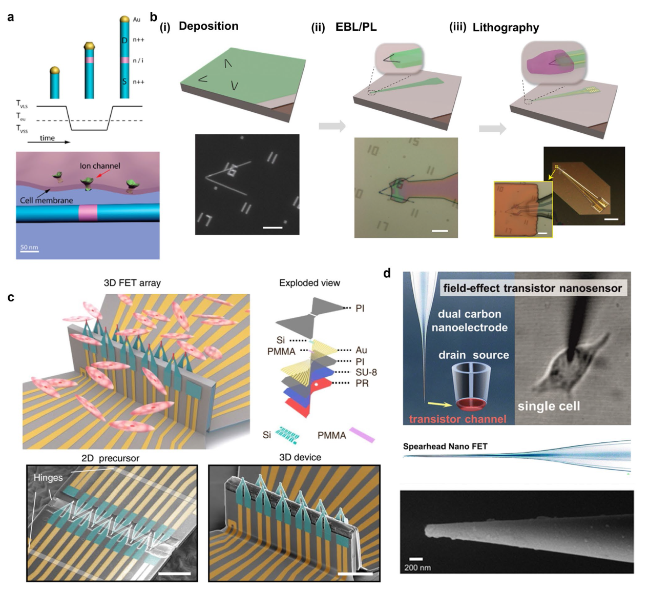
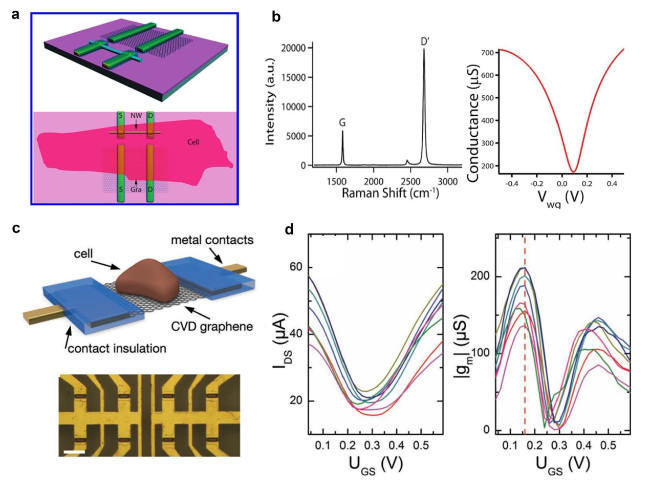
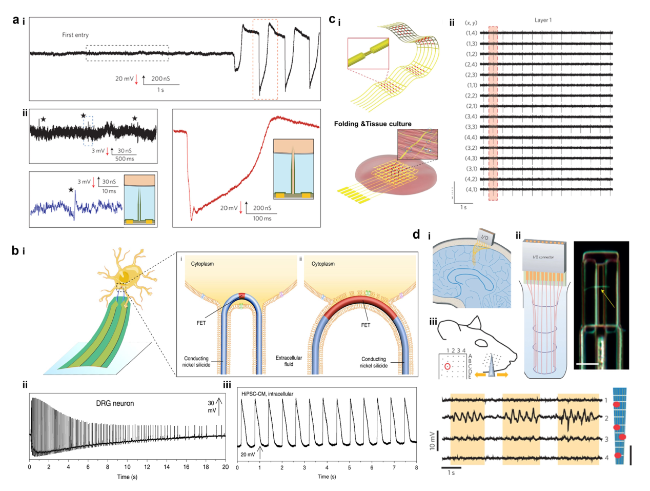
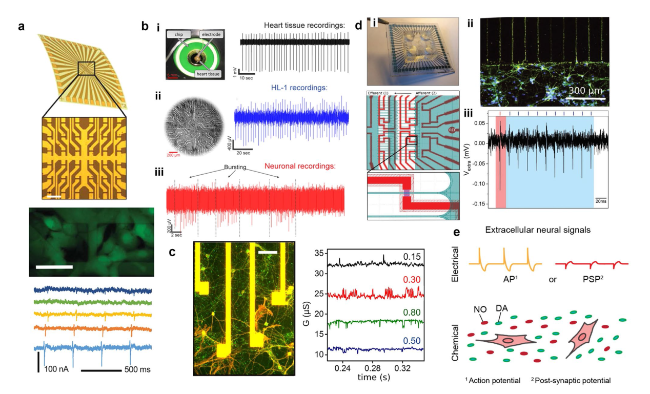
Cardiomyocytes serve as the principal functional constituent of the heart, featuring a distinctive membrane structure comprising a phospholipid bilayer and membrane proteins that regulate ion transport, sustain intracellular homeostasis, and generate basal action potentials through specific ion channels [
126,
127]. The transmembrane potential of cardiac myocytes can be categorized into resting and action potentials based on their respective properties. During the resting state, cell membrane is in polarization and its potential across the membrane is ~ −90 mV [
128,
129,
130]. The autonomous cardiomyocytes generate the action potential spontaneously, which is subsequently transmitted to the working cardiomyocytes. The process of generating an action potential in cardiomyocytes can be segmented into five distinct phases, which include phase 0 (characterized by rapid depolarization due to inward flow of Na
+), phase 1 (marked by early rapid repolarization due to outward flow of K
+), phase 2 (a plateau phase resulting from the combined effects of outward flow of K
+ and inward flow of Ca
2+), phase 3 (late rapid repolarization due to outward flow of K
+), and phase 4 (involving the restoration of intra- and extracellular concentrations of Na
+, K
+, and Ca
2+ primarily through the action of ion pumps) [
131,
132,
133]. The action potentials obtained by using patch-clamp measurements are typically ~ 100 mV and the duration of the action potential is ~ 200-400 ms [
132]. It is noteworthy that the morphology of action potentials can be influenced by distinct cell types, including pacemakers, ventricular myocytes, and atrial myocytes, which exhibit variations in duration and under-phase. Furthermore, the high-sealing resistance and low interface impedance enhance the fidelity of the recorded signals. Presently, intracellular recordings utilizing three-dimensional nano-transistors represent the most accurate approximation of authentic action potentials. BIT-FETs, which are intracellular nanotube FETs, demonstrated exceptional action potential recordings from cardiac myocytes, exhibiting amplitudes of 75-100 mV and durations of ~ 200 ms (
Fig. 5a) [
41,
81]. The scalability and minimally invasive properties of nanodevices are crucial for the concurrent, prolonged monitoring of cardiac myocytes. The deterministic shape-controlled nanowire, coupled with spatially defined semiconductor-to-metal conversion, facilitated the production of scalable three-dimensional nano-transistors with controlled tip geometry and sensor size, thereby enabling the recording of intracellular action potentials of up to 100 mV from primary neurons
(Fig. 5b(ii)) [
42,
105]. Empirical investigations of neurons (
Fig. 5b(ii)) and cardiomyocytes (
Fig. 5b(iii)) have demonstrated that the regulation of device curvature and sensor dimensions is crucial for attaining intracellular recordings with elevated amplitude. The U-NWFET configuration permits multifaceted recordings from individual cells and cellular networks, thereby facilitating prospective inquiries into the dynamics of the brain and other tissues. Intracellular signals provide insights into cell types and the correlation between ion channel densities and cellular pathology in disease states. Subthreshold signals have the potential to uncover intercellular synchronization processes and electrophysiological regulatory mechanisms, which have significant implications for comprehending cellular physiology, pathology, and intercellular interactions [
31,
134].