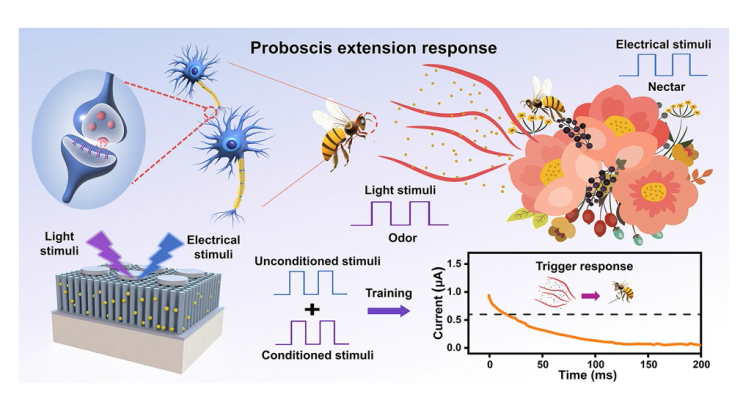
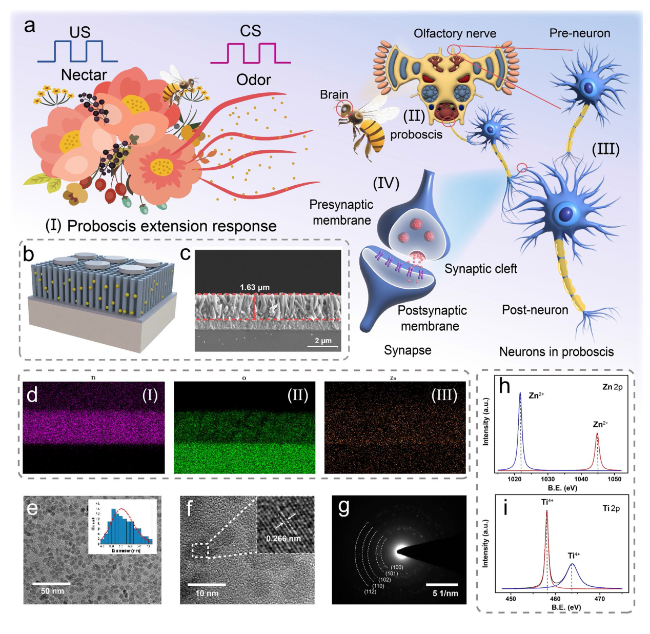
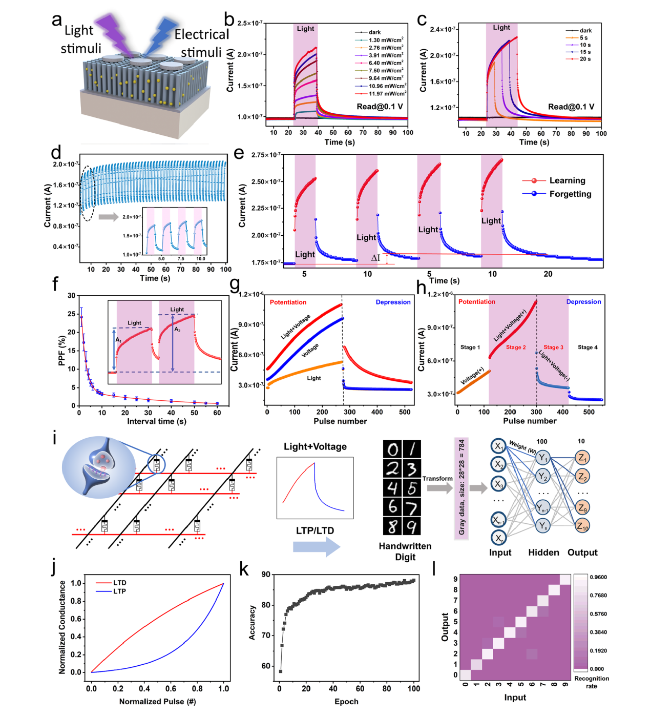
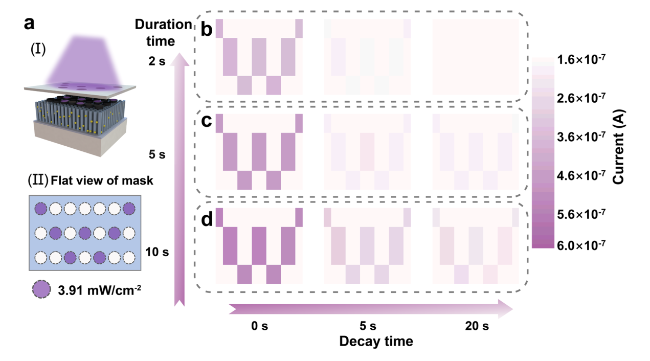
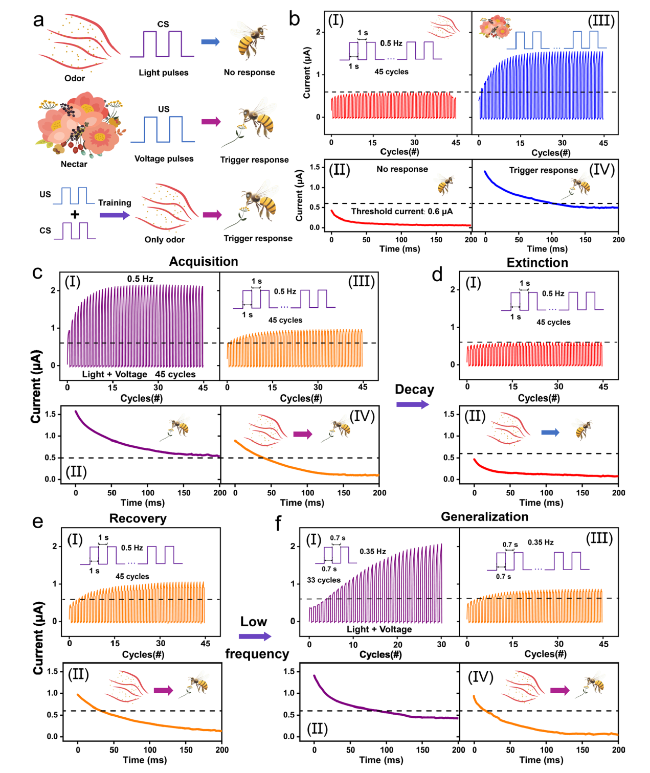
Classical conditional reflex behavior, which dominates the associative learning behavior in the biological brain, has been extensively analyzed in bionic electronics research. In this study, the PER behavior of honeybees is employed as a paradigm to exhibit the associative learning behavior in the device. During the implementation, the light pulses are denoted as CS (odor), and the voltage pulses are denoted as US (nectar), as shown in
Fig. 5a. The US naturally triggers proboscis extension (unconditioned response, UR), whereas the CS can induce proboscis extension only after repeated training using associative pairs of US (conditioned response, CR). During the measurement, a series of reading voltage pulses (0.1 V, 2 ms) is provided to read out the conductance response, and the threshold current is set at 0.6 μA to distinguish the occurrence of PER. The CR increases to ≈ 0.58 μA and cannot exceed the threshold even though the device is excited by consecutive CS inputs (45 cycles of purely light pulses, 0.5 Hz), thus indicating that the reflex is not triggered, as shown in
Fig. 5b-(I). Subsequently, the light was turned off, and the decaying current over time was observed by applying reading voltage pulses, as shown in
Fig. 5b-(II). The decaying current remained constant below the threshold (initial: 0.45 μA). Conversely, under the excitation of consecutive US (45 cycles of purely voltage pulses, 0.5 Hz), the UR gradually exceeds the threshold (reaches ≈ 1.55 μA), thus indicating that the PER is triggered (
Fig. 5b-(III)).
Figure 5b-(IV) depicts the decaying current over time after excitation by the US. The decaying current was initially above the threshold current corresponding to the response status; it then gradually decreased below the threshold within 100 ms, thus indicating synaptic action potential firing until the end of the reflex. Classical conditioning is a temporary neural connection established based on unconditioned reflexes and manifests four features: acquisition, extinction, recovery, and generalization. These features correspond to the storage of information, elimination of old information, rememorization, and storage of new information in the biological brain. Therefore, the acquisition feature is the basic condition required to establish a neural connection between CS and US. In this case, associative pairs comprising light stimuli (CS, 0.5 Hz) and voltage stimuli (US, 0.5 Hz) were concurrently applied to the device for acquisition. The conductance response rapidly increases above the threshold, triggering the PER, then rises to ≈ 2.19 μA after 45 training cycles, as shown in
Fig. 5c-(I). The sum of the individual response (≈ 2.13 μA) is slightly lower than the case induced by the associative pairs (≈ 2.19 μA), which can be attributed to the saturated excited states in the active layer.
Figure 5c-(II) depicts the decaying current after removing the associative pairs; evidently, the PER can be maintained until 200 ms without relaxing below the threshold. Immediately after training with the associative pairs, the triggering behavior of the PER was investigated using only the CS. The CR reaches ≈ 0.92 μA after 45 cycles of light stimuli, which is sufficiently large to trigger PER, thus indicating the establishment of a connection between the US and CS (acquisition), as shown in
Fig. 5c-(III).
Figure 5c-(IV) shows that the decaying current is initially above the threshold (≈ 0.89 μA) and then gradually decreases below the threshold at ≈ 45 ms, representing the relaxation of the PER. The training results of the associative pairs demonstrate that only the CS could trigger the response; however, this neural connection is not permanent. Extinction can dominate the disappearance of the conditioning response. After 10 min, 45 cycles of the light stimulus (CS, 0.5 Hz) were applied to each device. The CR below the threshold in
Fig. 5d-(I) indicates that the CS alone failed to trigger the PER after 10 s of decay time, and the initial decaying current (≈ 0.48 μA) after excitation also remained constant below the threshold (extinction). However, the initial decaying current after training is slightly larger than that of the untrained case (
Fig. 5b-(II), ≈ 0.45 μA) and below the threshold, thus demonstrating the weak neural connection between the US and CS. Additionally, synaptic plasticity enables previously extinguished responses to be activated rapidly and with large magnitudes when the same associative pairs arrive, which is the feature of recovery.
Figure 5e-(I) depicts the conductance response after training using the same associative pairs. The CR is enhanced to approximately 0.97 μA, and the initial decaying current is excited to 0.95 μA (
Fig. 5e-(II)), both of which are larger than the previous acquisition (conductance response: 0.92 μA, initial decaying current: 0.89 μA), thus indicating the re-strengthening of the neural connection between the US and CS (recovery). Under generalization, similar stimuli can also trigger the same CR after forming a neural connection under specific associative pairs. Low-frequency associative pairs comprising light stimuli (CS, 0.35 Hz) and voltage stimuli (US, 0.35 Hz) were considered as similar stimuli and applied simultaneously to the device after a recovery period. The conductance response gradually increased above the threshold, and the PER was effectively triggered even if the triggering process was slow, as shown in
Fig. 5f-(I). In
Fig. 5f-(II), the decaying current initially decreases from approximately 1.41 μA and continues to decay below the threshold at 88 ms. Similar stimuli elicit a weaker response and demonstrate a faster decay tendency compared with the high-frequency training. The CR was tested after training with similar stimuli, and it gradually increased to approximately 0.81 μA, as shown in
Fig. 5f-(III); this proves that the CS can trigger PER. Subsequent measurements of decaying currents (
Fig. 5f-(II)) also verified that similar stimuli can trigger the PER and establish neural connections. However, for generalization, the differentiated stimulus produced a lower response than the previous stimulus, as can be observed from the low CR and fast decay trend. In summary, the PER of the honeybee was emulated in the ATZ-based device, and its four key features, i.e., acquisition, extinction, recovery, and generalization, were successfully implemented.