Highlights
1 Introduction
2 Fabrication Strategies and Properties of MXenes/Polymer Nanocomposites (MPCs)
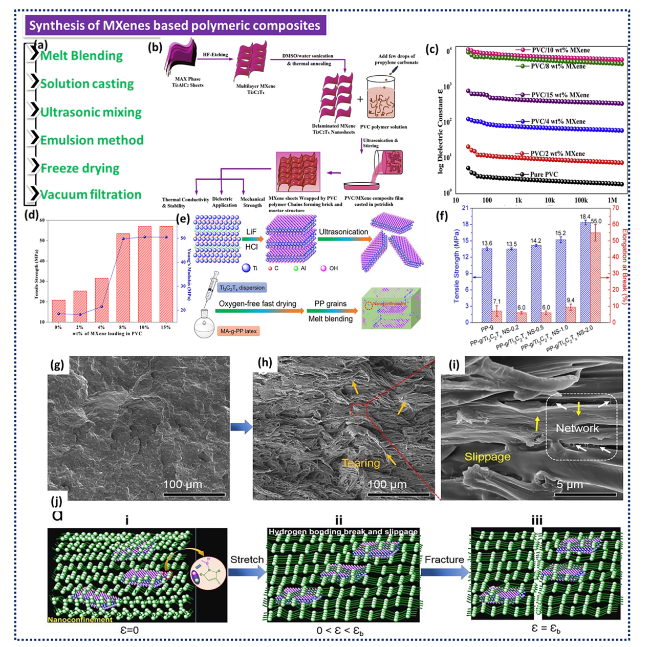
Fig. 1 a Different techniques for synthesising MPCs. b Schematic illustration showing the preparation of d-Ti3C2Tx (HF-etching prior to delamination by DMSO and water and followed by high-temperature annealing) and PVC/Ti3C2Tx nanocomposite films. c Dielectric constant and (d) Young’s modulus as well as tensile strength of PVC/MXene nanocomposites. Reproduced with the permission from Ref. [90], Copyright Elsevier 2020. e Schematic representation of melt blending for the fabrication of Ti3C2Tx/PP nanocomposites. f Tensile strength and elongation at break of the Ti3C2Tx/PP nanocomposites as a function of the Ti3C2Tx content. SEM images of 2.0 wt% Ti3C2Tx/PP nanocomposite as well as g their fracture surface after freeze-brittle fracture. h and i Stretch-fracture surface; j Schematic models of mechanical failure (including H bonding break and slippage) of PP-g/Ti3C2Tx NS-2.0. Reproduced with the permission from Ref. [91], Copyright Elsevier 2019 |
Table 1 Overview of typical MXenes-based polymer composites with the respective application area and key features |
| MXenes type | Polymer matrix | Application area | Features | References |
|---|---|---|---|---|
| AgNWs/Ti3C2Tx | PU | Strain sensing | 1000 cycling stability, response/recovery time of 344/344 ms | [77] |
| Ti3C2Tx | Chitosan and PU | Pressure sensing | High compressive strength | [78] |
| Mo2C | PVA | Humidity sensing | 1/1.8 s recovery time | [79] |
| TiO2/Ti3C2Tx | Nafion | Peroxide sensing | Linear range of 0.1-380 μM, sensitivity of 447.3 μA (mM cm2)−1, detection limit of 14 nM, 94.6% left after 60-day storage | [80] |
| GOx/Au/Ti3C2Tx | Nafion | Glucose sensng | Sensitivity of 4.2 μA (mM cm2)−1, detection limit of 5.9 μM (S/N = 3) | [81] |
| Ti3C2Tx | PVA | Capacitor | Electrical conductivities of 2.2 × 104 S m−1, volumetric capacitance of 530 F cm−3 at 2 mV s−1 | [82] |
| Ti3C2-S/d-Ti3C2 | PP | Battery | Reversible capacitance | [83] |
| Ti3C2Tx | PDT | Supercapacitor | Capacitance of 52.4 mF cm−2 (3.52 F cm−3) at 0.1 mA cm−2, 10,000 charge-discharge cycles (100%) | [84] |
| Ti3C2Tx | PVDF | Antibacterial | Antibacterial rate of 73 and 67% against Bacillus subtilis Escherichia coli, respectively | [85] |
| Ti3C2Tx | PAN | Air purify | 2.5 removal efficiency of 99.7%, low pressure drop of 42 Pa | [86] |
| Ti3C2Tx | PANI/PP | EMI | ~ 40 dB | [87] |
2.1 MXene-based Thermoplastic Polymer Composites and Their Properties
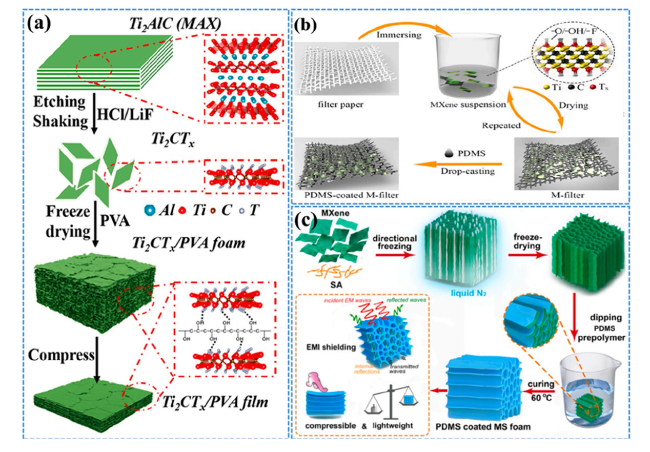
Fig. 2 a Graphical representation of the fabrication of composite foams and films made from f-Ti2CTx/PVA. Reproduced with the permission from Ref. [93], Copyright American Chemical Society 2019. b Schematic representation of the synthesis procedure for freestanding nanocomposites. Reproduced with the permission from Ref. [94], Copyright Elsevier 2020. c Diagram depicting the generation of hybrid aerogels and PDMS-coated MS foams. Reproduced with the permission from Ref. [95], Copyright Elsevier 2020 |
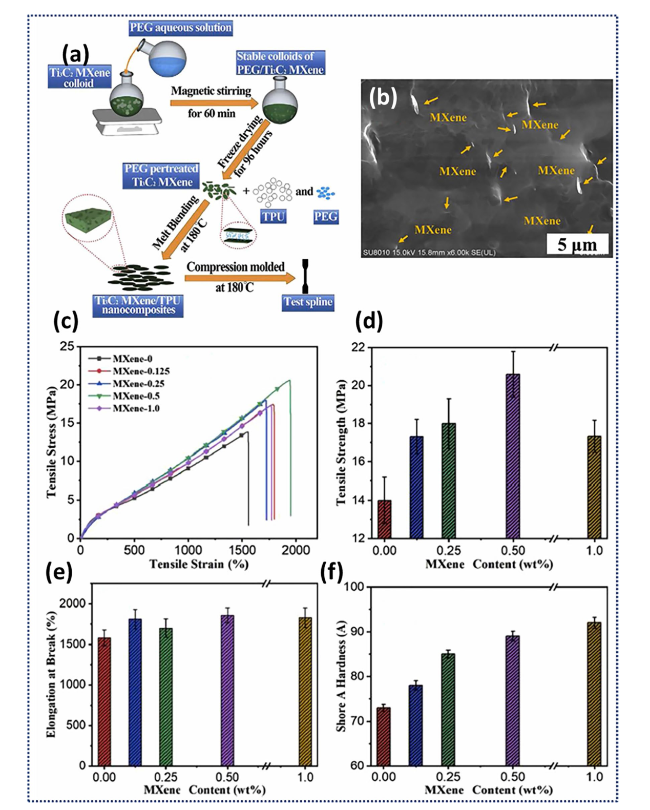
Fig. 3 a Schematic representation of melt blending used to prepare Ti3C2Tx/TPU nanocomposites with the b corresponding SEM image of 1.0 wt% Ti3C2Tx/TPU. Resulting mechanical properties of the Ti3C2Tx/TPU nanocomposites as a function of the MXene content including c tensile stress-strain curves, d tensile strength, e elongation at break, and f hardness. Reproduced with the permission from Ref. [96] Copyright Elsevier 2019 |
Table 2 General information regarding Ti3C2Tx MXene/thermoplastic nanocomposites |
| Thermoplastic Polymer | Method of preparation | References |
|---|---|---|
| PU | Emulsion method | [92] |
| PVA | Casting/evaporation method | [89] |
| UHMWPE | Melt blending | [98] |
| PEDOT:PSS | Vacuum-assisted filtration process | [99] |
| NR | Vacuum-assisted filtration process | [97] |
| PVA | Solution blending method | [88] |
| PVC | Solution blending method | [90] |
2.2 MXene-Based Thermosetting Polymer Composites and Their Properties
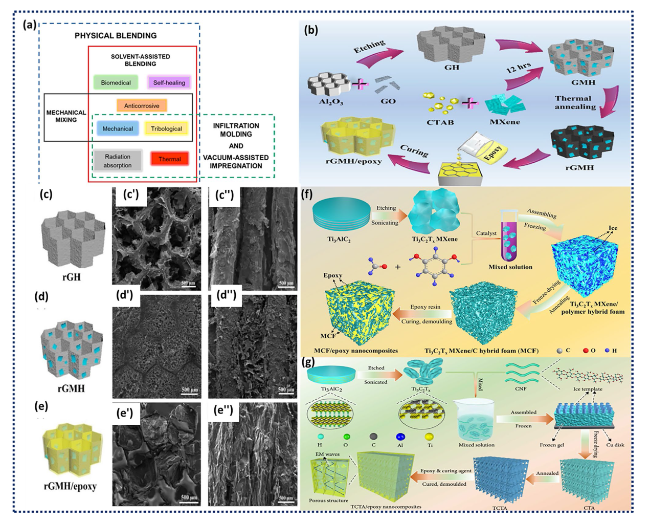
Fig. 4 a Schematics of MXene/epoxy nanocomposites processing techniques. b Schematic illustration of the fabrication process of c rGH, d rGMH, and e rGMH/epoxy nanocomposites. Axial and radial SEM images of c', c" rGH, d', d" rGMH, and e', e" rGMH/epoxy nanocomposites, respectively. Reproduced with the permission from Ref. [111], Copyright Elsevier 2020. f Schematic representation of the fabrication process employed to produce MCF/epoxy nanocomposites. Reproduced with the permission from Ref. [112], Copyright Elsevier 2019. g Graphical representation of the synthesis method for TCTA/epoxy nanocomposites. Reproduced with the permission from Ref. [113] Copyright American Association for the Advancement of Science 2020 |
3 Microstructure and Morphology of MPCs
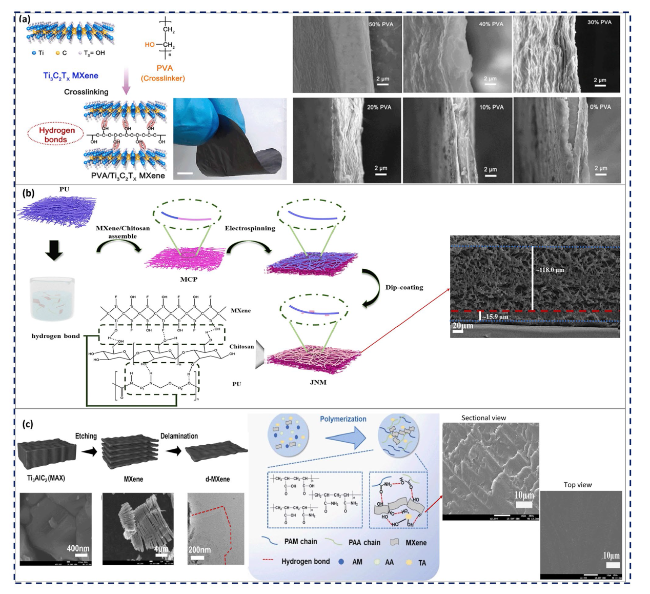
Fig. 5 a Schematic depiction of the PVA/Ti3C2Tx MPC thin-film synthesis process, the related optical image of the resultant MPC, and the cross-sectional SEM images of the film with different content of PVA varying from 50 to 0%. Reproduced with the permission from Ref. [117], Copyright Elsevier 2021. b Schematic illustration of the Janus nanofibrous MXene/chitosan/polyurethane MPC with interfacial hydrogen bonding and the pertinent SEM images. Reproduced with the permission from Ref. [118], Copyright Elsevier 2023. c Schematic diagram of Ti3C2Tx nanosheet fabrication and SEM images of Ti3AlC2 and multi-layered MXene nanosheets, as well as TEM image of monolayered MXene nanosheet plus the schematic diagram of PAM/PAA/MXene/TA hydrogel fabrication and the sectional and top-view SEM images of the resultant MPC. Reproduced with the permission from Ref. [119], Copyright Elsevier 2022 |
Table 3 Structural properties of MPCs (Dpore: Pore Size, SSA: specific surface area, ESA: electrochemical surface area) |
| MPC composition | Morphology of the microstructure | References |
|---|---|---|
| Ti3C2Tx/polydopamine | Cylindrical mesochannels | [115] |
| (Dpore = 7.6 nm, SSA = 89.5 m2 g−1) | ||
| Ti3C2Tx/SWCNT/PANI | Hierarchical structure | [116] |
| (ESA = 10.80 cm2 per unit area of the platinum electrode) | ||
| Ti3C2Tx/PVA | Hierarchical structure | [117] |
| (Dpore = 0-2 µm) | ||
| Ti3C2Tx/polypyrrole | Cylindrical to spherical mesostructures and spherical macrostructure | [120] |
| (Dpore = 7.8-52 nm, SSA = 129-188 m2 g−1) | ||
| Ti3C2Tx/cellulose acetate/sodium alginate | Unique ordered porous network | [121] |
| (Dpore = 30-50 μm) | ||
| Ti3C2Tx/gelatin/polyacrylamide | Compact honeycomb-like porous structure | [122] |
| Ti3C2Tx/polyacrylamide/polyacrylic acid/tannic acid | Smooth microstructure | [119] |
| Ti3C2Tx/chitosan/polyurethane | Janus core-shell porous network | [118] |
| (Dpore = few micrometers) |
4 Structure-Property Relationship of MPCs
Fig. 6 Graphical representation of the property improvements in MXenes-polymer nanocomposites in comparison to bare polymers |
5 Advanced Polymer-Supported MXenes for Stretchable Sensors
5.1 Synthetic Soft Matrix/MXenes-Based Stretchable Sensors
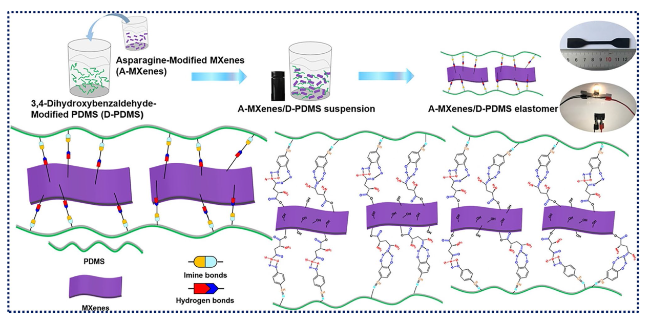
Fig. 7 Primary steps in making MXenes/PDMS-based elastomeric nanocomposites and their physical interactions. Reproduced with the permission from Ref. [145], Copyright American Chemical Society 2020 |
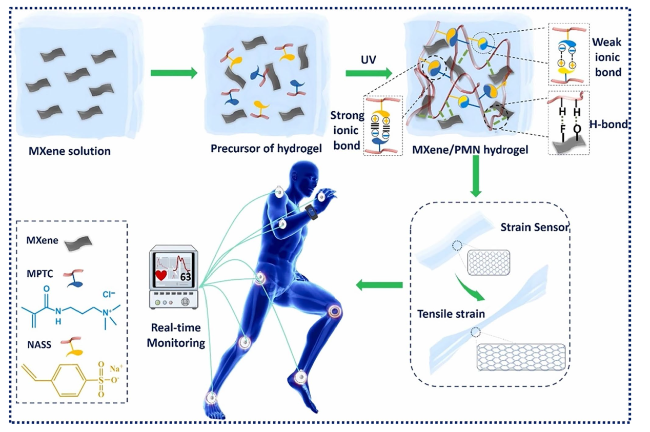
Fig. 8 Diagrammatic representation of the manufacturing process for MXene/PMN hydrogels. Reproduced with the permission from Ref. [154], Copyright Elsevier 2022 |
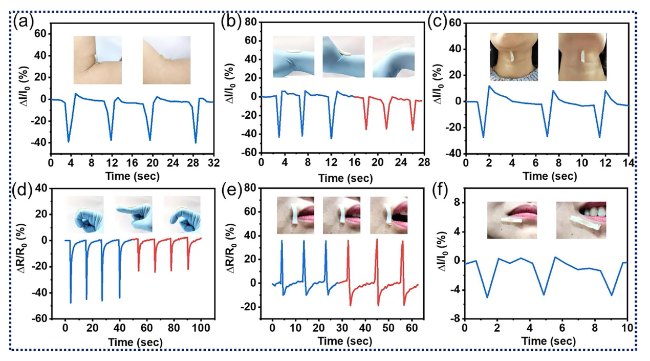
Fig. 9 Human motions detection. a How the bilayer composite hydrogel strain sensor’s electrical signal changes when the arm bends. b When the wrist moves in two different ways, the relative current of the strain gauge changes. c How the piezoresistive reaction of the strain sensor changes when the head moves. d The electrical output from the strain sensor when the finger is bent 45° and 60°. e When the mouth opens, there is a change in the relative resistance of the strain gauge. f Response curves for smiling right now. Reproduced with the permission from Ref. [3] Copyright Elsevier 2021 |
5.2 Biopolymer/MXenes-Based Stretchable Sensors
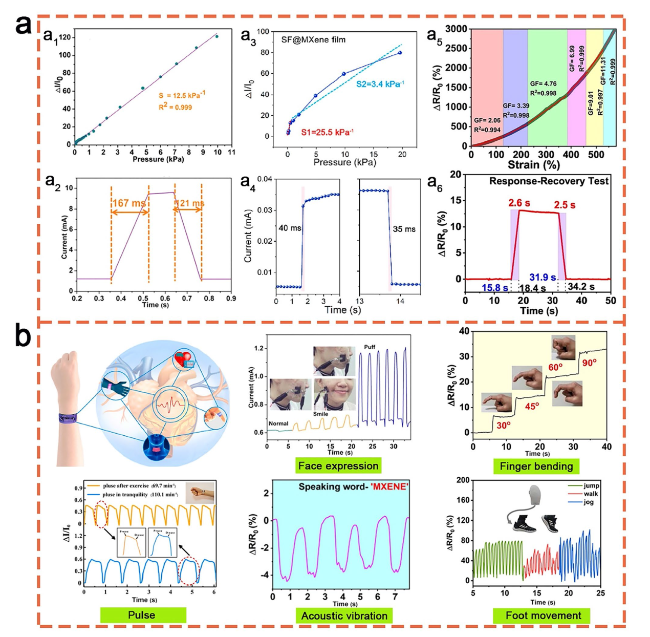
Fig. 10 a Sensitivities (a1, a3, a5) and response times (a2, a4, a6) of MXene/CNF. Reproduced with the permission from Ref [167]. (Copyright American Chemical Society, 2019), MXene/SF [172] (Copyright Elsevier, 2020) and MXene/CS [177] (Copyright Elsevier, 2023) based pressure sensors, as well as b their applications in human health detections, such as face expression [167] (Copyright American Chemical Society, 2019), finger bending [177] (Copyright Elsevier, 2023), pulse measurement [168] (Copyright Elsevier, 2021), acoustic vibration [177] (Copyright Elsevier, 2023), and foot movement [176] (Copyright Elsevier, 2023) |
Table 4 Performance of the biopolymer/MXene-based stretchable sensors |
| Sensing materials | Sensor type | Sensitivity | Sensing range; detection limit | Stability; response time | References |
|---|---|---|---|---|---|
| MXene/BC | Pressure sensor | 12.5 kPa−1 (0-10 kPa) | 0-10 kPa; 1.0 Pa | 100 cycles (99% strain), 100,000 cycles (50% strain); 167 ms | [167] |
| MXene/CNF | 419.7 kPa−1 (0-8.04 kPa), 649.3 kPa−1 (8.04-20.55 kPa) | 0-20.55 kPa; 4.0 Pa | 10,000 cycles; 123 ms | [168] | |
| MXene/CNF | 95.2 kPa−1 (2-50 Pa), 27.5 kPa−1 (0.05-3 kPa), 0.94 kPa−1 (3-10 kPa) | 2-10 kPa; 0.4 Pa | 25,000 cycles; 95 ms | [169] | |
| MXene/CNF | 10.7 kPa−1 (0-240 kPa), 34.6 kPa−1 (240-640 kPa), 16.6 kPa−1 (640-950 kPa) | 0-950 kPa; - | 3000 cycles; 298 ms | [170] | |
| MXene/CNT/CNF | 817 (0-0.2 kPa), 234.9 (0.2-1.5 kPa) | 0-1.5 kPa; - | 2000 cycles (30% strain);74 ms | [171] | |
| MXene/SF | 25.5 kPa−1 (0.1-0.5 kPa), 3.4 kPa−1 (1.0-20 kPa) | 0-20 kPa; 9.8 Pa | 3500 cycles (73 Pa); 40 ms | [172] | |
| MXene/SF/GO | 14.23 kPa−1 (MXene/SF (9:1), 1.4 kPa), 12.53 kPa−1 (MXene/SF (5:1), 1.4 kPa), | 0-1.5 kPa; 72 Pa | - | [173] | |
| MXene/SF/TA | GF: 0.59 | 20-100% strain; - | 600 cycles (self-healing); - | [175] | |
| MXene/SF/HA | Strain sensor | GF: 4.5-11.3 (5-80% strain) | - | 10,000 cycles (50% strain); - | [176] |
| Mxene/CS/PA | Strain sensor | GF: 3.15 (− 20 °C), 3.93 (25 °C), 2.55 (80 °C) | 1-600% strain, 0.08-3.2 kPa stress; 0.5 g | -; 500 ms | [158] |
5.3 Rubber/MXene-Based Stretchable Sensors
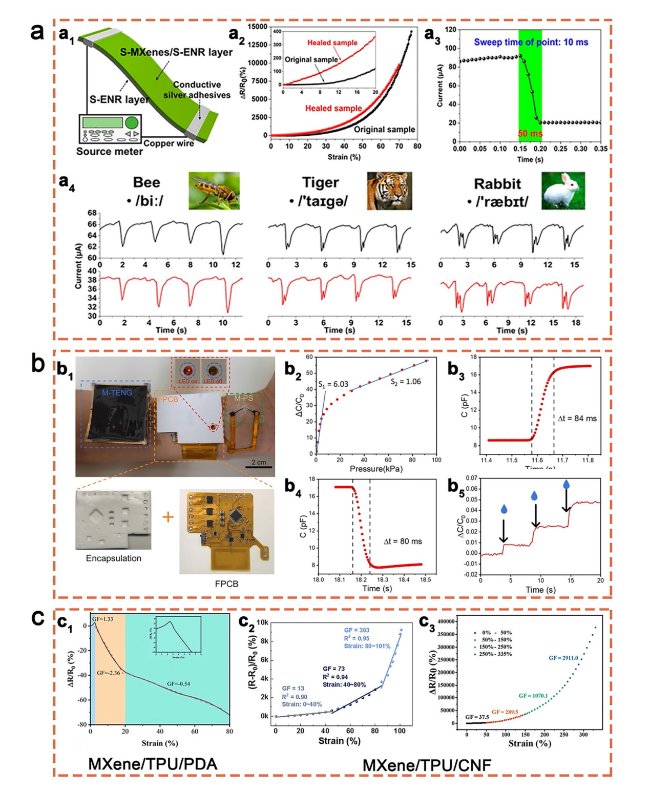
Fig. 11 a Structure (a1), properties (a2, a3), and speech detection application (a4) of the Ti3C2Tx/ENR stretchable sensor. Reproduced with permission from Ref [178]. (Copyright American Chemical Society, 2020). b Optical image (b1), and sensing performance (b2-b5) of the self-powered Ti3C2Tx/SEBS sensing system. Reproduced with permission from Ref [184]. (Copyright Elsevier, 2022). c Comparison of gauge factor between MXene/TPU/PDA and MXene/TPU/CNF stretchable sensors. Reproduced with permission from Refs [181,182,183]. (Copyright Elsevier, 2022), (Copyright American Chemical Society, 2022), (Copyright American Chemical Society, 2021) |
5.4 MXene-Coated Functional Textile-Based Sensors
Fig. 12 Proof-of-concept for HMI systems made with MXene-coated cotton. a, b Digital picture and circuit layout of the MXene-coated cotton pressure-sensing fabric interface. c Software for using MXene-coated cotton pressure-sensing cloth, called MXene Tools. d, e MXene Tools pressure sensing (single area and multi-region) and pressure-sensitive data modelling diagrams in 3D. Reproduced with the permission from Ref. [4], Copyright American Chemical Society 2020 |
5.4.1 MXene-Based Pressure Sensing Mechanisms
5.4.1.1 Piezoresistive
5.4.1.2 Capacitive
5.4.1.3 Piezoelectric
5.4.1.4 Triboelectric
5.4.1.5 Potentiometric
5.5 MPCs for Breath-Based Biomarker Diagnosis
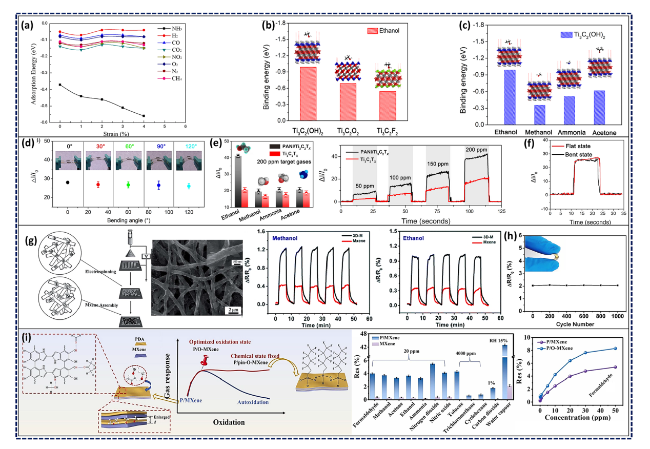
Fig. 13 a Simulated adsorption energy of eight different gas molecules on Ti2CO2 monolayers as a function of applied biaxial strains, and side- as well as top-view of schematic of the pertinent adsorption. Reproduced with permission from Ref. [212], Copyright American Chemical Society 2015. b DFT adsorption energy values of ethanol molecule on Ti3C2(OH)2, Ti3C2O2, and Ti3C2F2 and c Comparison of adsorption energy values for ethanol, methanol, ammonia, and acetone gas molecules on Ti3C2(OH)2; d Optical images of flexible PANI/Ti3C2Tx sensors with the sensitivity values against varied bending angles (exposed to 150 ppm ethanol gas molecules); e The juxtaposition of the pristine Ti3C2Tx and PANI/Ti3C2Tx MPC in terms of selectivity for various gas targets (200 ppm) and dynamic response/recovery trends ranging within 50-200 ppm of ethanol, both performed at RT, and f bending stability of flexible sensors to 150 ppm ethanol gas molecules. Reproduced with the permission from Ref. [219], Copyright Wiley 2019. g Schematic illustration of the fabrication process, SEM image of the 3D PVA/PI/Ti3C2Tx, as well as the resistance changes of the 3D MPC sensor and MXene sensor during 5 successive cycles of exposure to 5 ppm methanol and ethanol; h Sensing performance of the 3D MPC sensor at 20 ppm acetone undergoing different bending cycles. Reproduced with the permission from Ref. [220], Copyright Royal Society of Chemistry 2018. i Schematic illustration of P/MXene chemical structure and the pinning strategy to acquire long-term sensing stability, comparison of the sensing responses of P/MXene and MXene at 20 ppm of formaldehyde, response intensities of P/MXene and MXene to various gas species, and sensing responses of P/O-MXene and P/pin-O-MXene to various gas species (P/MXene, P/O-MXene, and P/pin-O-MXene stand for PDA/MXene, partially oxidized P/MXene, and final pinned P/O-MXene, respectively). Reproduced with the permission from Ref. [222], Copyright Wiley 2020 |
5.5.1 Alcohol and Ketone Sensing
5.5.2 Ammonia Sensing
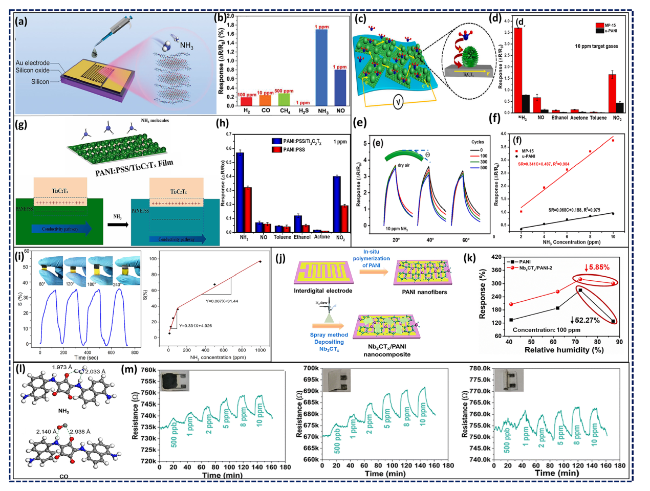
Fig. 14 a Schematic of drop-castedTi3C2Tx/PDDS hybrid films on the electrodes and b the selectivity of the fabricated sensor. Reproduced with the permission from Ref. [228], Copyright Wiley 2022. c Illustration of ammonia sensing mechanism of flexible Ti3C2Tx/urchin-like PANI hollow nanosphere composite sensors; d Comparison between pristine u-PANI and MP-15 (MPC with 15 wt% aniline) selectivity to different targets; e Dynamic response-recovery curves under successive bending cycles with different bending angles (inset depicts the bending orientation), and f Response-concentration fitting plots ranging within 2-10 ppm. Reproduced with the permission from Ref. [229], Copyright Elsevier 2022. g Sensing mechanism of Ti3C2Tx/PANI:PSS MPC, and h the comparison between MPC and pure PANI:PSS in terms of selectivity. Reproduced with the permission from Ref. [230], Copyright Elsevier 2023. i Gas response of the Ti3C2Tx/PANI:PSS composite-based sensor towards 100 ppm NH3 at varied bending angles and the linear dependence between the gas response and ammonia concentration. Reproduced with the permission from Ref. [231], Copyright American Chemical Society 2020. j Fabrication process of the Nb2CTx/PANI sensors; and k humidity influence on the sensing response for 100 ppm ammonia. Reproduced with the permission from Ref. [232], Copyright Elsevier 2021. l DFT-simulated adsorption modes and adsorption distance between PDAC and two different target molecules, namely NH3 and CO, and m real-time resistance of the developed Ti3C2Tx@PDAC paper-based sensor versus time with respect to the ammonia concentration in flat, folded, and rolled states. Reproduced with the permission from Ref. [235], Copyright Royal Society of Chemistry 2023 |
5.5.3 Sensing of Non-polar Gases and Other VOCs
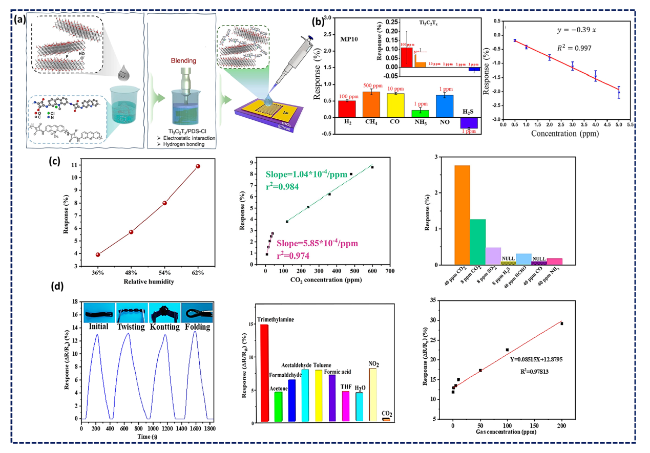
Fig. 15 a Schematic illustration of Ti3C2Tx/PDS-Cl nanocomposite synthesis and sensor fabrication: addition of MXene ink to the water dissolved PDS-Cl polymer, tip sonication (for 2 min at 20 W) for accelerating the physical blending, and fabrication of H2S sensor by drop-casting the MPCs solution on prepatterned Au electrodes; b Selectivity of Ti3C2Tx/PDS-Cl (with 10 wt.- % of MXene: MP-10) toward 6 different analytes with a negative response for H2S (inset shows the selectivity of pristine Ti3C2Tx), dynamic sensing response of MP-10 for various concentrations of H2S, and the related calibration curve. Reproduced with the permission from Ref. [237], Copyright American Chemical Society 2023. c Response of the ternary N-doped- Ti3C2Tx/PEI/rGO as a function of RH, the pertinent linear fitting of response toward 8-600 ppm CO2, and selectivity under 62% RH. Reproduced with the permission from Ref. [240], Copyright American Chemical Society 2020. d Dynamic responses of the MXene@AuNP DN hydrogel sensor to 1 ppm TMA under multiple deformation states (initial, twisting, knotting, and folding states), selectivity of the gas sensor, and the acquired linear fitted response versus TMA concentration at RT. Reproduced with the permission from Ref. [242], Copyright American Chemical Society 2022 |
6 Development of Advanced MPC Sensors
7 Challenges, Potential Remedies, and Future Strategies
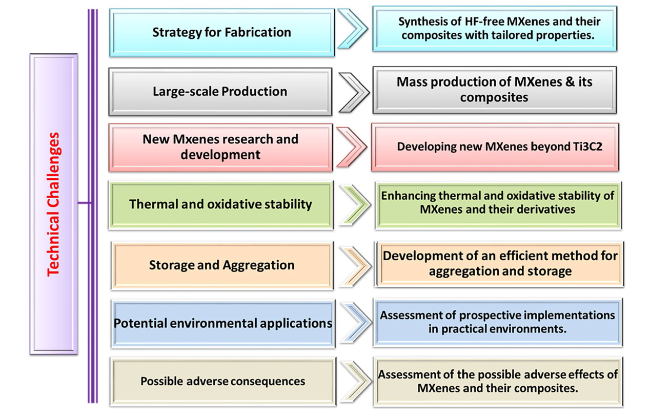
Fig. 16 Significant technical obstacles in the application of MXenes and their composites |