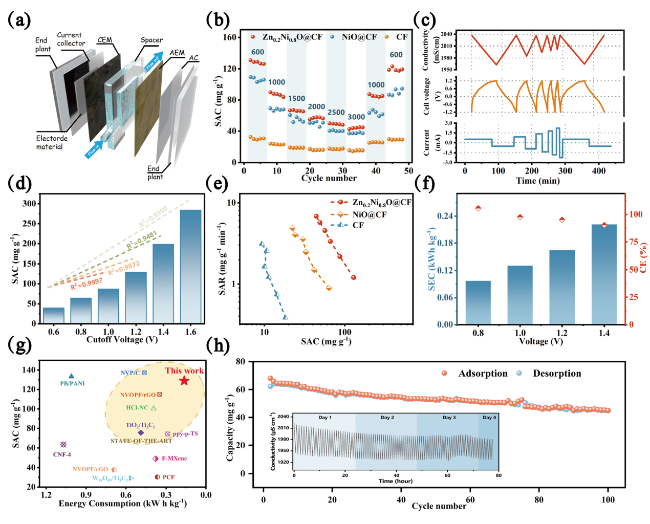
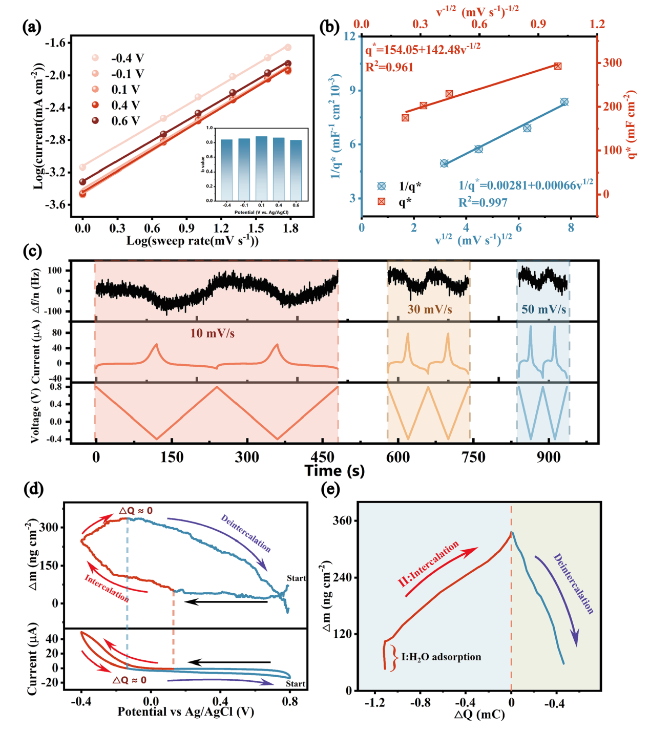
To gain further insight into the mass transport of Na
+ during the adsorption process, we detected the relationship between mass and charge capacity change of electrode materials in the electrochemical process by EQCM-D. As shown in
Fig. 4c, significant changes in the frequency factor of the Zn
0.2Ni
0.8O@CF electrode can be observed during the CV process. Under the condition of various scan rates, the frequency responses basically returned to their initial values and showed good periodic changes, indicating that on the experimental timescale, the mass changes were recoverable and excellent stability of the Zn
0.2Ni
0.8O@CF electrode [
80]. EQCM-D results of the 3rd cycle showed that the frequency (Δf
5/5) decreased at varying scan rates (Fig. S9a), indicating that the mass of the Zn
0.2Ni
0.8O@CF electrode increased qualitatively as a result of the Na
+ adsorption process; additionally, Δf
5/5 returned to approximately 0 Hz, demonstrating the Na
+ desorption process caused the decrease in the electrode mass. It was noteworthy that dissipation factor (D) corresponding change showed a similar trend, decreasing first and then returning to the initial value (Fig. S9b), indicating the absorption of ions on the Zn
0.2Ni
0.8O@CF electrode was reversible, thus confirming the stability of the Zn
0.2Ni
0.8O@CF electrode. The decrease in D when the electrode lost energy quickly implied that adsorbate on the Zn
0.2Ni
0.8O@CF surface was rigid and compact [
81]. In this case, CV curve of the 3th cycle and the simultaneous EQCM-D response were shown in
Fig. 4d, and the corresponding △m − △Q plot was calculated and shown in
Fig. 4e. We found that the electrode mass increases uniformly when the material charge increases; while when the current was reversed (△Q ≈ 0), the △m trend changes and the electrode mass starts to decrease, indicating that the material has excellent pseudocapacitive properties and the de-/intercalation process has a high sensitivity to the current response due to the rapid transfer process. The mass change of electrode closely followed the theoretical mass change in stages II and III, indicating that all of the charges were applied for sodium-ion intercalation and no other side reactions occurred. The Sauerbrey's equation was applied to analyze the EQCM-D results to quantify the mass changes of the Zn
0.2Ni
0.8O@CF [
82]. The Red I region of the △m increased without the change in △Q may be the adsorption process of water molecules. The MPE value in the adsorption region (Red II) was 20.68 g mol
−1, which was marginally lower than 23 g mol
−1, demonstrating that Na
+ adsorption was the main process in this region after partial desolvation of sodium hydrate. However, in the desorption region (Blue), the MPE value was 45.16 g mol
−1, indicating that this process was co-deintercalation of a water molecule and a Na
+ ion. The EQCM-D data revealed the excellent pseudocapacitive properties of the material, the mechanism of the ion storage process (intercalation of one sodium ion and co-deintercalation of one sodium ion with one water molecule) and a high degree of sodium-ion adsorption reversibility within the electrochemical processes of sodium storage of Zn
0.2Ni
0.8O@CF.