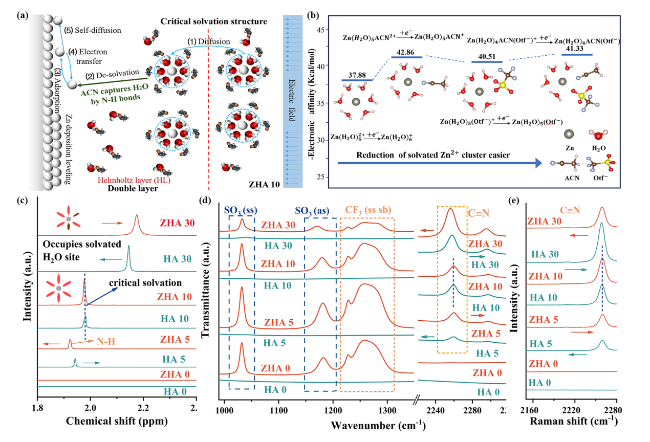
The critical solvation structure proposed in this paper is based on the organic molecule acetonitrile with low polarity and high dielectric constant (
ε = 35.9) that combines with water through N-H bonds to reconstruct a hydrogen bond network [
47,
48,
49,
50]. First, different mass fractions of ACN were dissolved in 1 m Zn(CF
3SO
3)
2 to form ZHA X (X = 0, 5, 8, 10, 20, 30, 50, 70) electrolyte to explore the priority and influence of two different solvation effects. When the electrolyte is ZHA 0, [Zn(H
2O)
6]
2+ is formed in the outer Helmholtz layer (OHL) of the electric double layer by self-diffusion [
51,
52]. Then during the desolvation process, the desorbed active molecules can accept electrons from the surface of the zinc anode, and then a HER occurs to produce H
2 [
53]. The remaining OH
- will generate Zn(OH)
2 by-products on the surface of Zn anode [
54]. The hydrogen evolution effect and the Zn
2+ deposition effect is a pair of competing effects. Hydrogen evolution will reduce the reversibility of Zn
2+ deposition/stripping, resulting in a zinc-poor state and causing corrosion [
16,
55,
56]. The generated H
2 will also be adsorbed on the Zn anode to affect the nucleation process of Zn (Fig. S1). As shown in
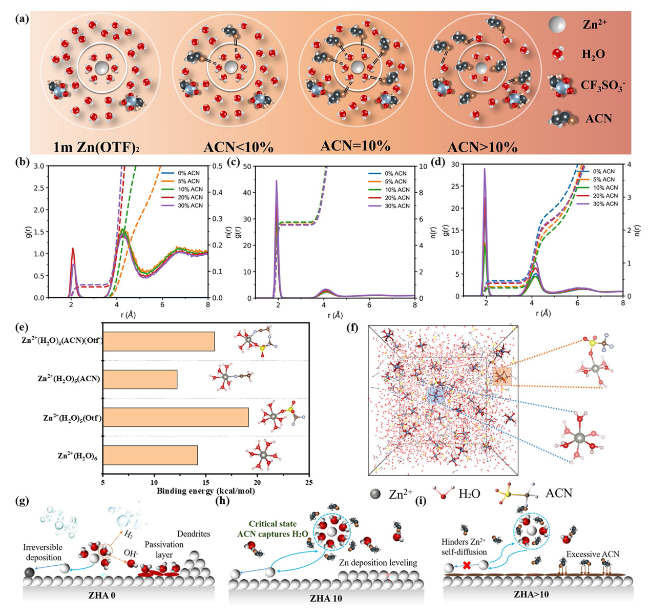
Fig. 1a, when the electrolyte is ZHA 10, The critical solvation structure formed suppresses free water in the electrolyte. During the subsequent desolvation process, the "catcher" can also arrest the detached active molecules, thereby inhibiting the corrosion of HER and Zn anodes. When the electrolyte is ZHA > 10, ACN will enter the solvation structure of Zn
2+ and replace the original position of H
2O (Fig. S2). However, this solvated structure is unstable and easy to self-decompose. Moreover, since the polarity of ACN is weaker than that of H
2O, the contact angle between the electrolyte and the Zn anode becomes smaller and smaller after the addition of ACN (Fig. S3). At this time, excess ACN will be preferentially adsorbed on the Zn anode, thus affecting the deposition of Zn
2+. Excess ACN can significantly affect the viscosity and ion transport efficiency of the electrolyte (Fig. S4). As shown in
Fig. 1b, we compared the electron affinity relationship between Zn(H
2O)
62+ and the different solvated structure. Zn(H
2O)
62+ is lower than Zn(H
2O)
5(ACN)
2+, Zn(H
2O)
5(Otf
−)
+, Zn(H
2O)
4(ACN)(Otf
−)
+. It shows that when ACN participates in the remodeling of the solvation sheath of Zn
2+, the overall stability of the electrolyte is reduced. Further reveal the differences in the stability of different solvated structures.