Highlights
1 Introduction
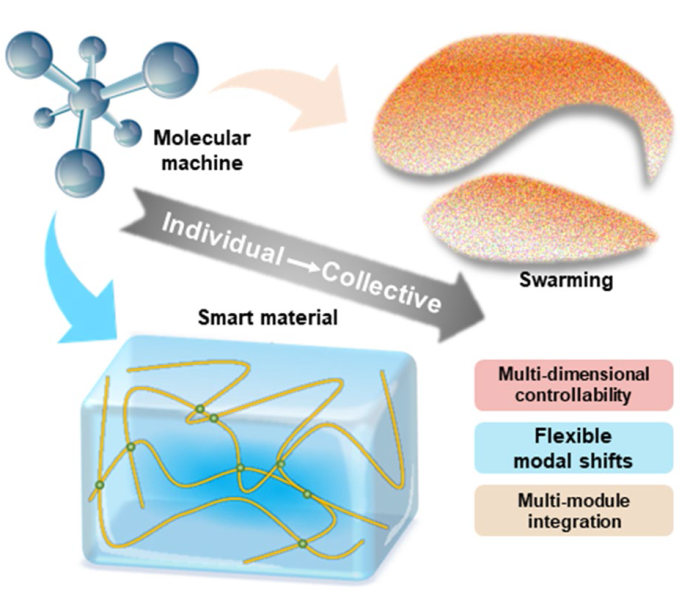
Fig. 1 Synthetic and biological molecular machines are capable of converting energy to produce unidirectional conformational changes. They constitute the building blocks of collective behavior. There are two typical patterns of collective behavior, one that produces macro-behavior and the other that swarms. They are able to amplify the work and exhibit intelligence and versatility. The experience and paradigm of micro/nanorobots may be able to guide the design of collective behaviors of molecular machines to form multi-dimensional and multinodular |
2 Basic Building Blocks of Molecular Machines
Fig. 2 Main types of molecular machines. a Rotating molecular machines utilize external energy to produce rotation and prevent this rotation from being reversed by a ratchet mechanism. b Molecular motor with a different upper and lower part. The upper rotating unit repeats 360° rotation in four different steps under light of appropriate wavelengths and shows that the energy barrier of the thermal step in the rotational motion can be tuned by structural modifications. Reprinted permission from Ref. [34]. Copyright 2000, American Chemical Society. c Azobenzene molecular switch capable of producing cis and trans transitions under light. d The translational molecular machinery is able to migrate between different active sites. e Rearrangement of α-methylene-4-nitrostyrene from one amino group to another. Reprinted permission from Ref. [35]. Copyright 2013, American Chemical Society. f pH-switchable [c2] daisy chain. It mimics the ability of a muscle to produce contraction and stretch. g Kinesin and Dynein produce movement along microtubules. They are in opposite directions. Reprinted permission from Ref. [36]. Copyright 2018, American Chemical Society. h Actin, myosin and troponin make up the microstructure of muscle. Reprinted permission from Ref. [36]. Copyright 2018, American Chemical Society. i Two modes of strand migration of DNA, one toehold mediated and the other mediated by an enzymatic reaction. Reprinted permission from Ref. [37]. Copyright 2022, American Chemical Society |
3 Construction Strategies for Collective Molecular Machines
3.1 Macroscopic Work Produced by Amplification
Fig. 3 Collective behavioral patterns of molecular machines. a Molecular machines grasp objects by self-assembling or integrating into polymer networks that amplify nanometer mechanical motion to produce macroscopic work. This is expected to be used to build smart responsive materials. Two key issues in the design process are aligning and flexible controlling. b Molecular machines operate at micro/nanoscale by swarming. Swarming has parallelism, flexibility and robustness compared to individual behavior. During the design process, the focus needs to consider coupling and mode shifting. Reprinted permission from Ref. [90]. Copyright 2020, Taylor&Francis |
3.2 Swarming of Molecular Machines
4 Feature Regulation of Collective Behavior
4.1 Responsive Smart Material Construction
Fig. 4 Responsive smart material construction. a A polymer with dual optical control based on integrated motors and modulators. UV irradiation induces motor rotation of polymer chain entanglement. VIS light is then used to activate the optical switch to produce its open form. Free rotation effectively releases the elasticity accumulated during motor rotation. b Molecular machines with single chains arranged in bundles to form a layered structure, like myofibrils. Reprinted permission from Ref. [128]. Copyright 2016, Wiley-VCH. c Third-generation daisy-chained dendritic polymers with 21 [c2] daisy-chain-rotor alkane portions. Collective and amplified extension/contraction through each [c2] chrysanthemum-chain-wheel-oxidized alkane branch upon addition of acetate anions or dimethyl sulfoxide (DMSO) molecules as external stimuli. Reprinted permission from Ref. [129]. Copyright 2020, American Chemical Society. d Monomers (blue dashed square) with racemic motor (R,S)-M1 (yellow dashed square). A mixture of the LC monomers and racemic motor (R,S)-M1 is aligned from homeotropic to planar. The mixture is cured into a homogeneous film and is cut along the rubbing direction. The obtained ribbon is able to bend upon UV light irradiation (365 nm) or walk over a surface. Monomers with enantiomerically pure motors (R)-M1 and (S)-M1 (red dashed square). The monomers are mixed with (R)-M1 or (S)-M1 motor and aligned into a twisted nematic structure. The mixture is cured into a homogeneous film and is cut along the rubbing direction. The resulting ribbon with R-motor shows left-handed helical motion when irradiated with UV light, while the ribbon with S-motor shows right-handed helical motion. Reprinted permission from Ref. [130]. Copyright 2020, Wiley-VCH. e Schematic of photo-steerable rolling locomotors controllable on various terrains (top). Photo-structured winding process of spiral ribbon and helicoid helix structures according to the aspect ratio (AR) of the azobenzene-functionalized liquid crystal polymer networks (azo-LCN) strips (middle). Helix structures and their continuous buckling transition according to the AR of the azo-LCN strips(bottom). Reprinted permission from Ref. [131]. Copyright 2023, Wiley-VCH. f Structure of an optically responsive rotary motor and its self-assembly into nanofibers |
4.2 Swarming of Molecular Machines
Fig. 5 Swarming molecular machines. a Schematic diagram of motor binding states. In a dynamic system, the motor can enter four states: diffusion in solution, head-bound to microtubules, tail-bound to surface, head-bound to microtubules, and tail-bound to surface. b Microtubules assemble into bundles when green fluorescent protein (GFP)-kinesin aggregates on microtubules (top). Mechanisms of microtubule alignment (bottom). Reprinted permission from Ref. [159]. Copyright 2020, American Chemical Society. c MTs move in response to forces exerted by kinesin motor proteins (top). Assembly pattern of MTs enclosed in double-incorporated ring microwells during 60 mins (middle). At Δ/R = 1.17 (left), a protruding bundle pattern of MTs can be observed from the tip, whereas a bridging pattern of MT bundles can be observed at Δ/R = 1.92 (right). Assembly pattern of MTs confined in triple-conjugate circular microtiter wells within 60 mins (bottom). At Δ/R = 1.17, a similar protruding bundle-like pattern is formed from the tip. In contrast, these protrusions form bridges between the three tips at Δ/R = 1.92. Reprinted permission from Ref. [160]. Copyright 2021, American Chemical Society. d Models of bimodal and branching microtubules and the different morphologies of microtubules were formed with Tau-derived peptide-Azami-Green (TP-AG), single-linear state microtubules (black arrows), bimodal microtubules (red arrows), branching microtubules (blue arrows), multimodal microtubules (orange arrows), branching and multimodal microtubules (cyan arrowheads), and aster-like. Reprinted permission from Ref. [161]. Copyright 2022, AAAS |
Fig. 6 Microtubule-DNA-optical switch system. a Conjugation of r-DNA to azide-functionalized MTs by a click reaction. b Schematic of red and green MTs gliding on kinesins. c Schematic of the association of red and green MTs by l-DNA1 and their dissociation by d-DNA via extraction of l-DNA1 through a DNA strand exchange reaction. d Orthogonal control of swarming of MTs. The flexible MTs (red) were conjugated with r-DNA1 and r-DNA2, while the rigid MTs (green) were conjugated with r-DNA5 and r-DNA6. Upon inputting l-DNA1, the flexible MTs associated into circular shaped swarms through hybridization of r-DNA1 and r-DNA2 with l-DNA1 and appeared in red. Green swarms with translational motion associated with rigid MTs were formed through hybridization of r-DNA5 and r-DNA6 with l-DNA5. Swarms with translational and circular motions were simultaneously formed in response to the introduction of both input DNA signals. e Design of logic gates constructed with MTs. For the YES gate, the l-DNA1 signal was inputted into the system and swarming was obtained as the output signal (1 to 1). For the AND gate, l-DNA2 and l-DNA3 had both to be present to obtain swarming. For the OR gate, the presence of either l-DNA1 or l-DNA4 was sufficient to obtain swarming. Reprinted permission from Ref. [165]. Copyright 2018, Springer Nature |
5 Grand Challenges
Fig. 7 Challenges in the collective behavior of molecular machines, from controllability and stability to multimodularity. a Integration of multiple control sources may increase the dimensionality of modal shifts. b Swarm needs to be precisely controlled, including modes, paths, and targets. c The collective needs to ensure sufficient mechanical strength to prevent internal fracture during mission execution. d Specific forces need to be in place to maintain the swarm's aggregation. e Smart materials need to be combined with other materials to form layered structures to be application-oriented. f The collective development goal is to form orderly modularity and integration, like cells. Reprinted permission from Ref. [184]. Copyright 2020, Wiley-VCH |
5.1 Multi-Dimensional Precision Control
5.2 Sufficient Stability
5.3 Modularity and Integration
6 Experience from Micro/Nanorobots
Fig. 8 Molecular machines vs. micro/nano robots. In terms of size, one is mainly at the nanoscale, the other includes the micrometer and nanoscale. In terms of materials, one is mainly based on organic small molecules and biomolecules, the other is based on active particles, with biomaterials thrown in. In terms of mode of motion, one through conformational changes and the other through gradient force fields. In terms of control, one is dominated by chemistry and light, the other also includes magnetic fields and ultrasound. In terms of desired applications, one is used for building smart materials and micromanipulation, the other for micromanipulation, drug delivery and imaging. In terms of mode switching, molecular machines are currently more limited and less precise, compared to micro/nano robots that exhibit multi-dimensional and multi-layered structures in 2D, 3D and 4D. In terms of modules, molecular machines include receptors, controllers and actuators by virtue of the programmability of DNA. In contrast, micro/nano robots have integrated a large number of application modules, including recognition, loading, catalysis and imaging. Reprinted permission from Refs. [204,205,206,207,208,209]. Copyright @2019, American Chemical Society; @2020, American Chemical Society; @2012, American Chemical Society; @2018, American Chemical Society; @2018, Royal Society of Chemistry |