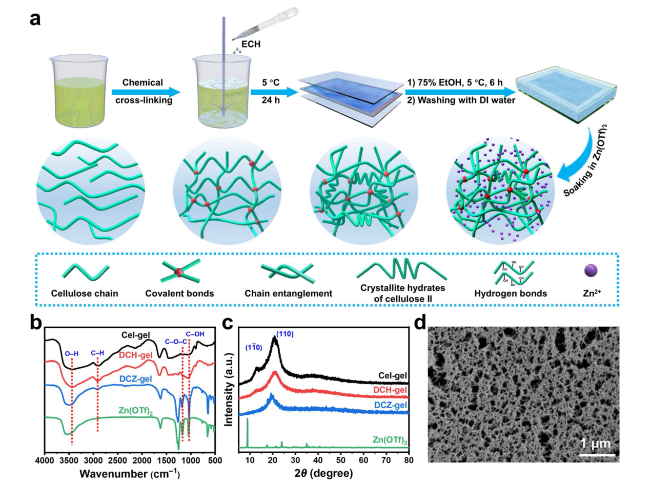
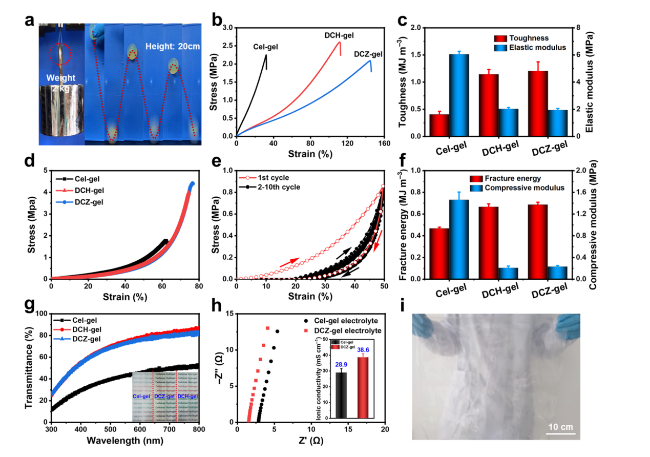
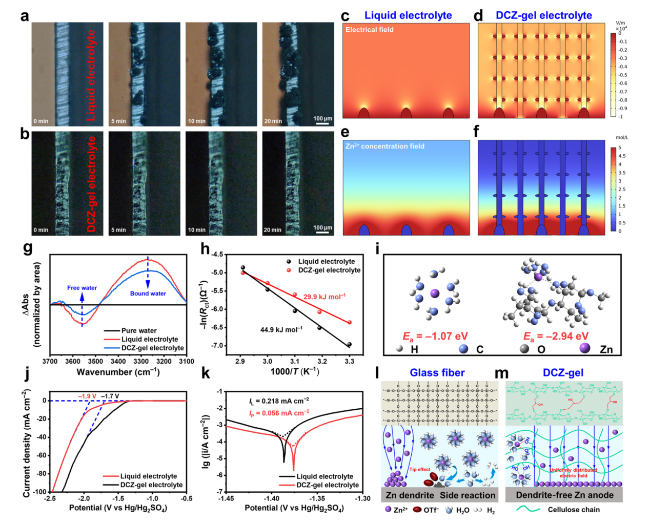
The mechanical properties of hydrogels are key factors for the capacity to suppress Zn dendrites growth and the adaptability to sustain external forces and deformations [
30]. Benefiting from the unique DC structure, the DCZ-gel exhibited high rigidity and elasticity. As displayed in
Fig. 2a, the DCZ-gel could lift steel blocks with a weight of 2 kg and bounce back up to about 80% after falling free from a height of 20 cm. Then, the mechanical properties of the hydrogels were quantitatively investigated by tensile tests (
Fig. 2b). The DCZ-gel (2.08 MPa, 145%) exhibited a similar tensile stress and higher tensile strain than DCH-gel (2.60 MPa, 111%) and Cel-gel (2.25 MPa, 32%), which might be due to the role of chemical cross-linking and Zn
2+ ions coordination while maintaining internal physically cross-linked domains. Therefore, the toughness and elastic modulus of DCZ-gel reached 1.20 MJ m
−3 and 1.93 MPa (
Fig. 2c), respectively. Also, the relationship between the dosage of ECH and mechanical properties of DCZ-gel was studied (Fig. S4a, b). As the volume of the ECH increased, the toughness of the cellulosic hydrogels increased while their elastic modulus decreased. However, the toughness reached a maximum when the volume exceeded 3 mL, which was a typical feature of the DC network [
31]. That is, the microstructure of the cellulose hydrogel was controlled by both the density of chemical and physical cross-linking, balancing them is crucial for improvement of the mechanical properties. A similar trend was observed for the compressive behavior of the cellulosic hydrogels in
Fig. 2d. The DCZ-gel (4.42 MPa, 76%) exhibited higher compressive stress and strain than DCH-gel (4.00 MPa, 74%) and Cel-gel (1.77 MPa, 62%). Moreover, successive loading-unloading tests were performed at a maximum compressive strain of 50%, and the curves in different cycles almost overlapped with each other after the first cycle (
Fig. 2e), suggesting that no prominent plastic deformation or strength degradation occurred. The corresponding fracture energy and compressive modulus also reached 0.68 MJ m
−3 and 0.23 MPa (
Fig. 2f), respectively, overcoming the intrinsic fragile properties of cellulose-based hydrogel. Therefore, the structural integrity of DCZ-gel could be retained during the assembly process of coin cells [
32]. Likewise, increasing the ECH content reduced the compressive modulus of the cellulose hydrogel, and the fracture energy reached a maximum when the volume exceeded 2 mL (Fig. S4c, d). Notably, DCZ-gel exhibited other unique properties. As shown in
Fig. 2g, due to the reduction of recrystallization of cellulose chains, the transmittance of DCZ-gel could reach 74.1% at 550 nm. Such a transparency in the visible light region could ensure the alignment of electrodes, which have a great impact on the electrochemical performance [
33]. Attributing to the more ion transport channels, DCZ-gel had an ionic conductivity of 38.6 mS cm
−1 calculated from the Nyquist plot in
Fig. 2h, which was higher than that of Cel-gel (28.9 mS cm
−1). Moreover, we evaluated the transference number of Zn
2+ ions (${\text{t}}_{{\text{Zn}}^{2+}}$) of DCH-gel based on chronoamperometry (CA) and impedance curves (Fig. S5). A higher ${\text{t}}_{{\text{Zn}}^{2+}}$ of 0.73 was obtained, indicating its outstanding Zn
2+ transport capability. Particularly, a large scale (50 cm × 50 cm) of DCZ-gel could be manufactured (
Fig. 2i), highlighting its scalability and transparency.