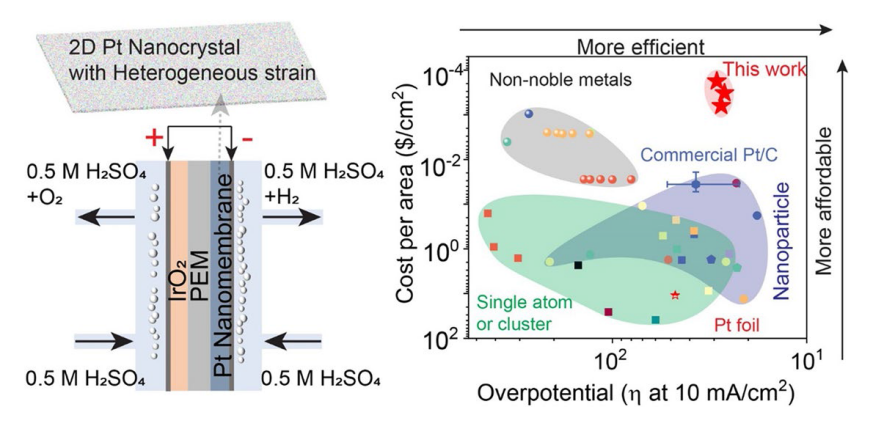
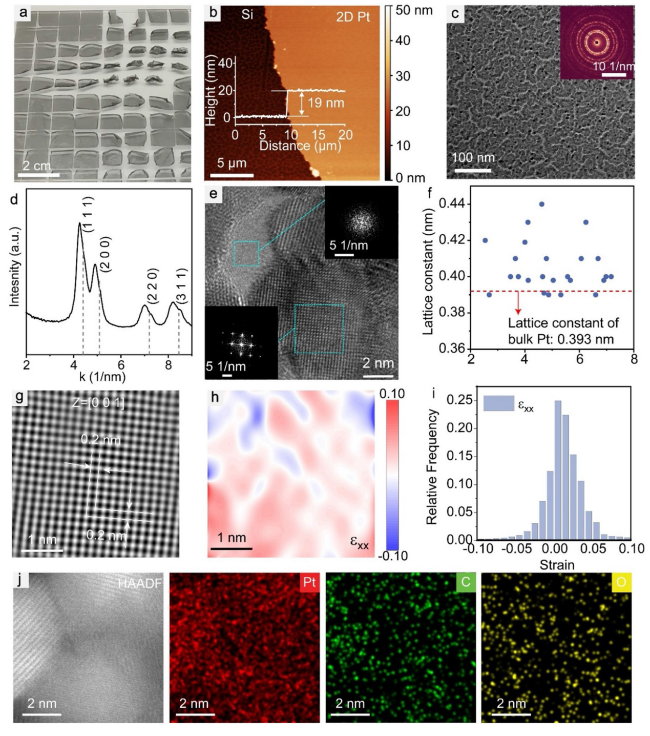
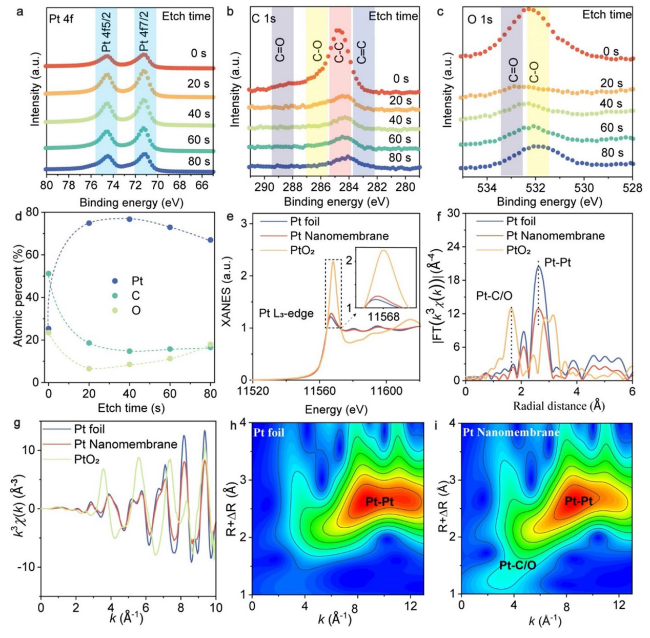
To further characterize the chemistry of our Pt nanomembranes, XPS was conducted (
Fig. 2a-d). The observed XPS spectra for Pt, C, and O after various etching times showed two prominent peaks at 71.1 and 74.5 eV that correspond to Pt
0 in its metallic state (
Fig. 2a). The XPS spectrum of C 1
s (
Fig. 2b) showed four distinct peaks at approximately 282.9, 284.8, 286.2, and 288.5 eV, which were attributed to C=C, C-C, C-O, and C=O, respectively. As previously reported in our studies [
29,
31,
46], these chemical bonds were expected to be residuals from the decomposed PVA during the exfoliation process of 2D metals. The XPS spectrum of O 1
s (
Fig. 2c) exhibited two main peaks at approximately 532.2 and 533 eV, corresponding to C-O and C=O, respectively, which provides further evidence of decomposed PVA. We have performed fitting of the XPS spectra, and the results are presented in Figs. S7 and S8. Based on our fitting analysis (Figs. S7a, c and S8a, c), we did not observe any indications of Pt oxides. However, we did observe that the distributions of binding energy for Pt
0 exhibit a skewness toward high energy values, leading to the presence of fat tails as depicted in Fig. S7a, d. This observation could potentially be attributed to lattice distortion within the Pt nanocrystals. Through data fitting (Fig. S7), we show the relative concentrations of Pt, C, and O as a function of etching time (
Fig. 2d), which demonstrates the chemical gradient in our Pt nanomembrane. The variation in composition with etching time is primarily due to the formation of a gradient nanostructure resulting from the reactions of metals and PVA. This phenomenon of a gradient nanostructure has also been observed in gold nanomembranes fabricated using PSBEE [
29]. Moreover, X-ray absorption spectroscopy (XAS) was performed on the Pt nanomembrane as well as two reference materials (e.g., Pt foil and PtO
2) (
Fig. 2e). Owing to the presence of metallic Pt, the Pt
L3 edge threshold energy and maximum energy for the X-ray absorption by our Pt nanomembrane look quite similar to those of Pt foil. However, upon closer inspection, it is evident that the white line intensity of our Pt nanomembrane is slightly higher than that of Pt foil (Pt
0), but significantly smaller than that of PtO
2 (Pt
IV). This behavior implies that, despite the dominant metallic Pt bonding, there is a tendency to form a cationic environment around Pt [
15,
23]. The Fourier transforms of the extended X-ray absorption fine structure (EXAFS) region yielded two prominent peaks at approximately 2.7 and 1.7 Å, corresponding to Pt-Pt and Pt-C/O coordination, respectively (
Fig. 2f) [
14]. The EXAFS analysis (Fig. S9a-d and Table S1) revealed an average Pt coordination number of approximately 9.8 for our Pt nanomembrane, which is smaller than the conventional Pt coordination number (= 12), indicative of a defected atomic structure in the Pt nanomembrane.
Figure 2g shows the curves of EXAFS
χ(
k) signals versus in
k-space obtained for our Pt nanomembrane, PtO
2 and Pt foil, from which we can see that the Pt nanomembrane is similar to Pt foil. Aside from Pt-Pt bonds, a few Pt-C/O bonds were observed in the nanomembrane (
Fig. 2h,
i). Notably, similar results were observed for the 28-nm-thick Pt nanomembrane (Figs. S8 and S10). Due to the limitation of the EXAFS technique, which does not allow for a direct distinction between Pt-C and Pt-O, it is challenging to measure their coordination numbers. To obtain an indirect estimation, we referred to the XPS data (
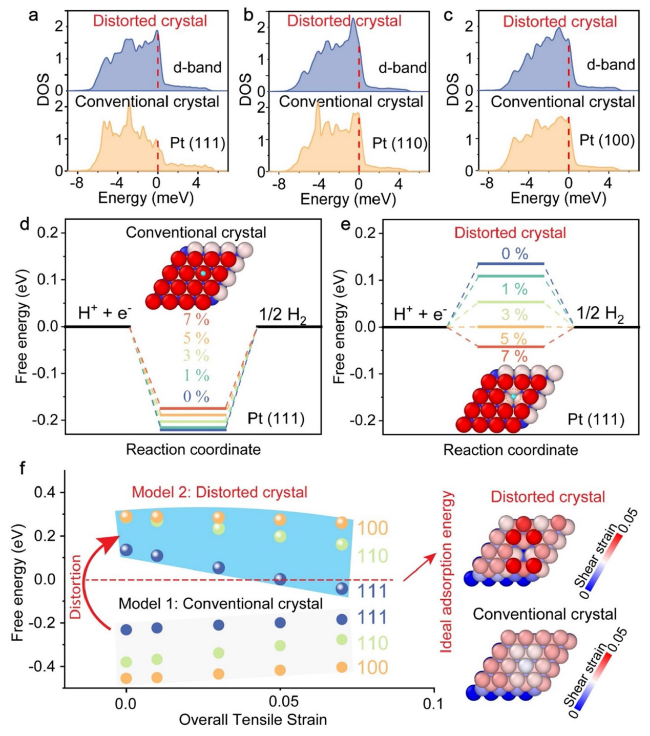
Fig. 2d), which revealed a relatively higher atomic percentage of C compared to O. Based on this observation, it is plausible that the coordination number of Pt-C is larger than that of Pt-O. Following this line of reasoning, we anticipated that, with the thickness decreases, the coordination number of Pt-C/O would increase, as supported by the EXAFS data of 19 and 28 nm Pt nanomembranes (Table S1). However, it is important to note that there is no clear trend, indicating that the Pt-C/O ratio significantly affects the HER performance, as depicted in
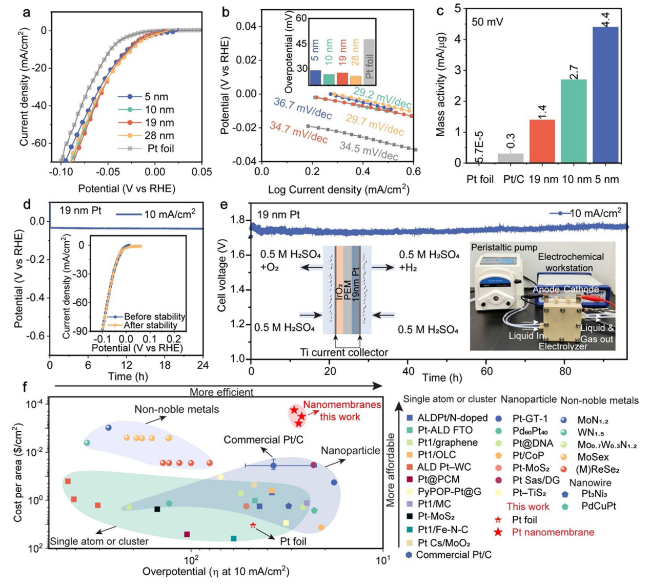
Fig. 3.