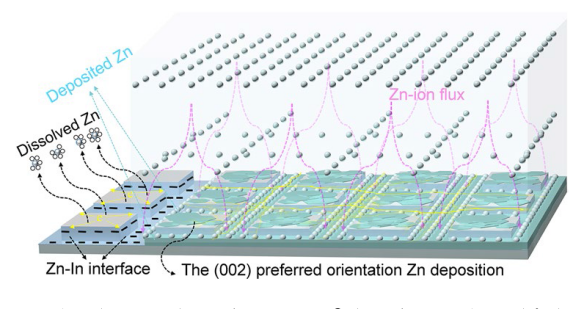
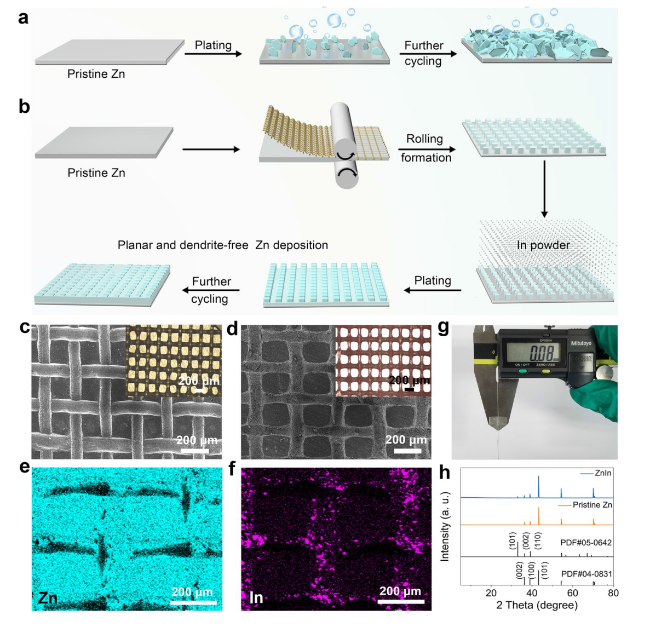
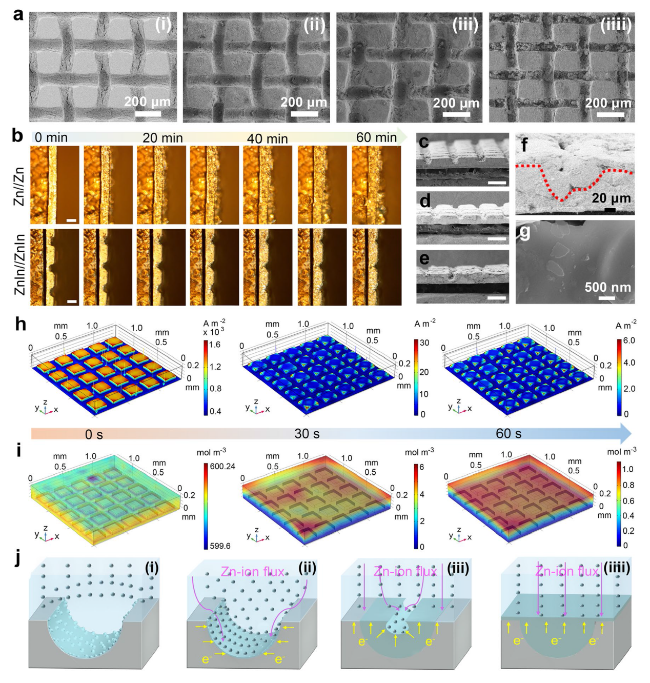
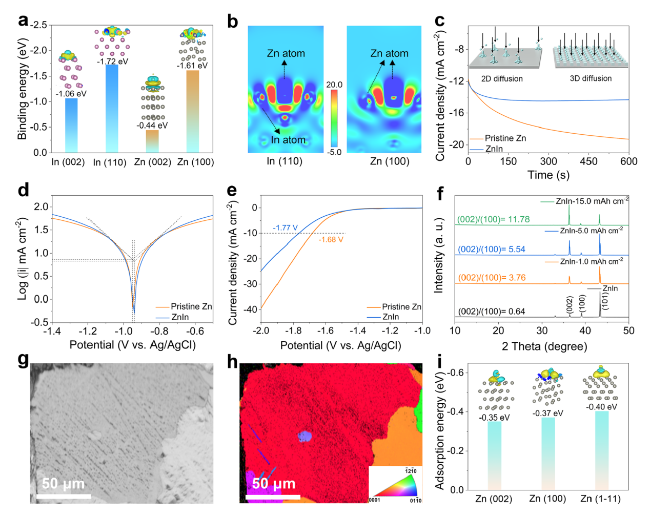
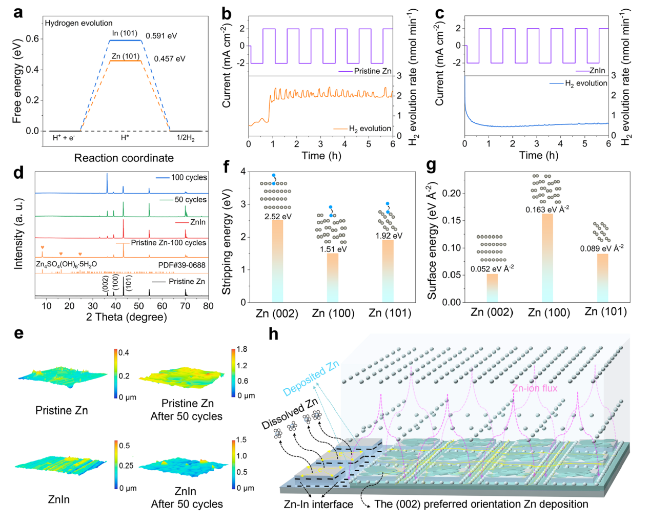
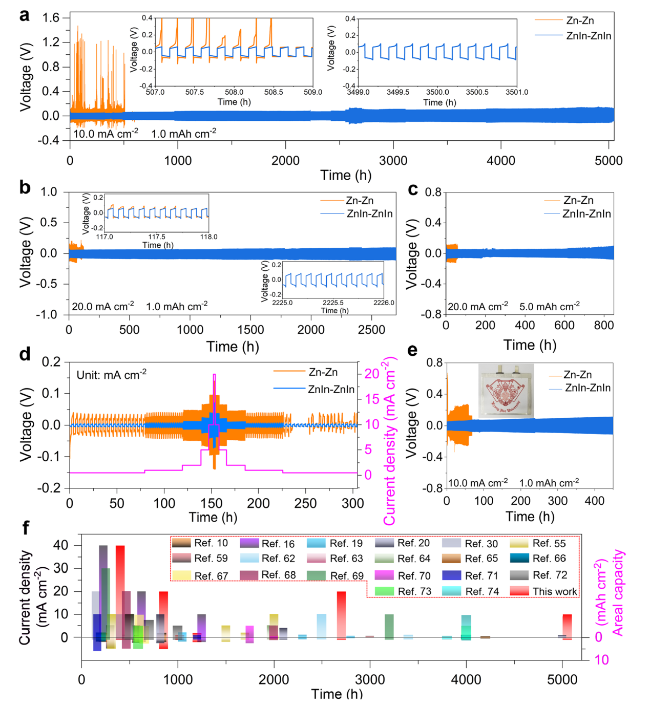
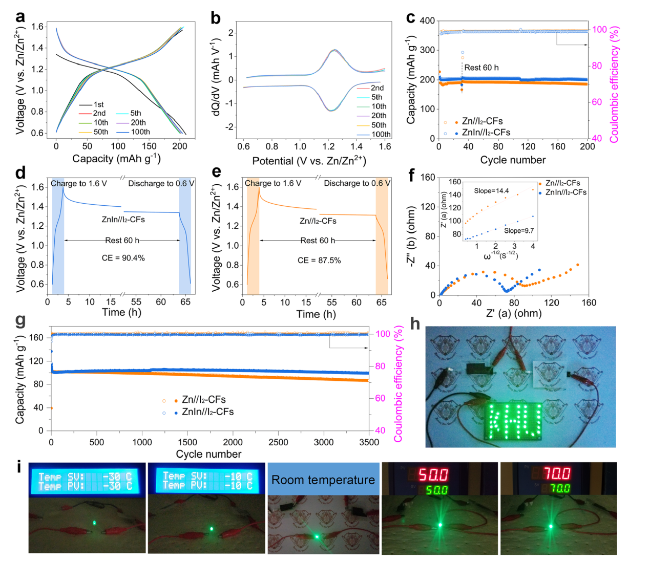
The practical application feasibility of the ZnIn anode was studied by assembling Zn-I
2 cells. Figure S31a-c and Video S4 show the excellent flexibility of the as-prepared CFs cathode host. The SEM and TEM images (Fig. S31d-f) reveal that the CF has a diameter of approximately 600 nm and exhibit a porous architecture. The porous architecture of the CFs facilitates the anchoring of I
2 on them. Figure S32a displays that the thickness of the as-prepared CFs is about 200 μm, and the loose structure is conducive to the I
2 loading and electrolyte infiltration. In addition, the TEM image of the I
2-CFs architecture and corresponding elemental mappings are further shown in Fig. S32b, indicating the uniform space distribution of C, N, and I within the I
2-CFs electrode. As shown in
Figs. 6a and S33a, the ZnIn/I
2-CFs and Zn/I
2-CFs cells deliver the initial discharge capacities of 212.7 and 227.6 mAh g
−1 at the current density of 0.5 C, and the corresponding CE values are 98.6 and 86.5, respectively. Meanwhile, the subsequent charge/discharge curves of the ZnIn/I
2-CFs cell display a high overlap, while the Zn/I
2-CFs battery exhibits slight fluctuations in subsequent cycles, implying excellent stability of the ZnIn/I
2-CFs cell. The higher initial CE and stable capacity output of the ZnIn/I
2-CFs cell suggests that the ZnIn anode can more effectively suppress side reactions than the pristine Zn anode. The corresponding d
Q/d
V curves are shown in
Figs. 6b and S33b. The ZnIn/I
2-CFs cell shows closer cathodic and anodic peaks than the Zn/I
2-CFs cell, which indicates that the Zn-In interface provides better electrochemical reaction kinetics and boosts iodine utilization [
75,
76]. Besides, the ZnIn/I
2-CFs cell displays a stable cycle performance compared to the Zn//I
2-CFs cell at 0.5 C (
Fig. 6c). The ZnIn/I
2 cell shows a better overlap than the Zn/I
2 cell, indicating a more stable electrochemical performance of the ZnIn anode. The capacity of the Zn/I
2-CFs cell tends to fade gradually after performing 80 cycles, which is attributed to the surface corrosion and side reactions of the pristine Zn anode. The self-discharge behaviors of the cells were evaluated by operating them for 30 cycles and discharging after 60 h of rest. The ZnIn/I
2-CFs cell maintains 90.4% of its initial capacity (
Fig. 6d), while the Zn/I
2-CFs cell is 87.5% (
Fig. 6e), demonstrating the anticorrosion of the ZnIn anode [
13]. In addition, the EIS plots show that the ZnIn/I
2-CFs cell has lower SEI resistance and charge transfer resistance than the Zn/I
2-CFs cell in the high-medium frequency region. Meanwhile, the ZnIn/I
2-CFs cell displays a smaller slope of Z′ versus
ω−1/2 in the low-frequency region, which indicates faster Zn-ion transport kinetics in the ZnIn/I
2-CFs cell (
Fig. 6f). Additionally, the ZnIn/I
2-CFs cell also shows excellent long-term stable cycling performance at a current density of 5.0 C (
Fig. 6g). The capacity of the ZnIn/I
2-CFs cell is maintained at 99.5 mAh g
−1 after 3,500 cycles, which was much better than the 86.5 mAh g
−1 of the Zn/I
2-CFs cell, indicating the excellent reversibility of the ZnIn/I
2-CFs cell. The charge-discharge curves of the ZnIn/I
2 cell show a better overlap than those of the Zn/I
2 cell, indicating a more stable electrochemical performance of the ZnIn anode (Fig. S34). Additionally, quasi-solid-state devices using gel electrolyte exhibit high mechanical integrity, good reliability, and a wide working temperature range, which have been widely studied in portable and flexible electronic products [
77,
78]. To verify the reliability of the prepared ZnIn/I
2-CFs battery in practical applications, two quasi-solid-state ZnIn/I
2-CFs pouch cells connected in series can easily power 40 LEDs (
Fig. 6h). Meanwhile, it can also provide stable energy output under repeated bending (Fig. S35) and a wide temperature range from − 30 to 70 °C (
Figs. 6i and S36), offering new insights for the development of flexible electronic devices.