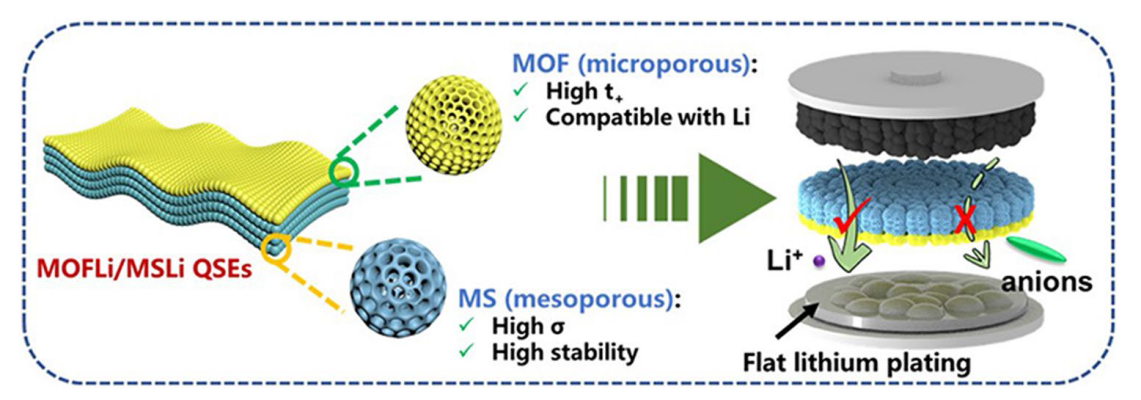
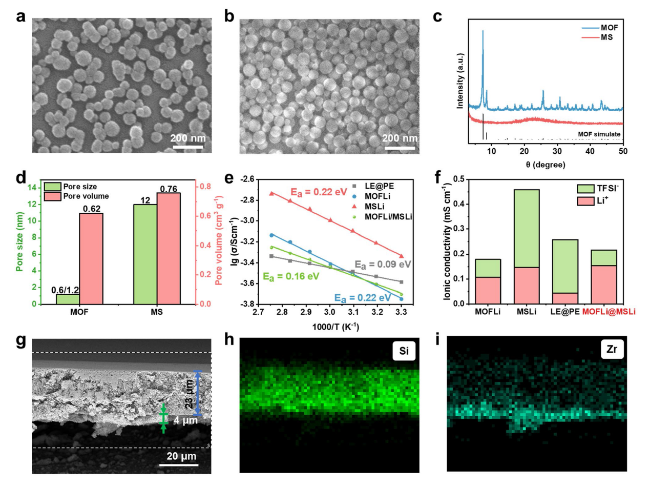
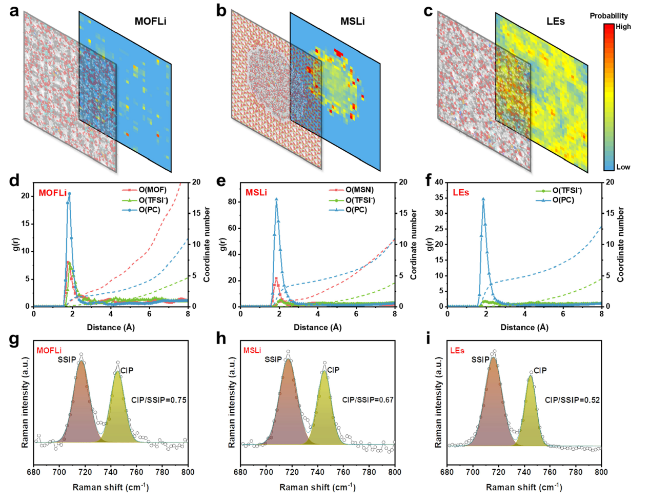
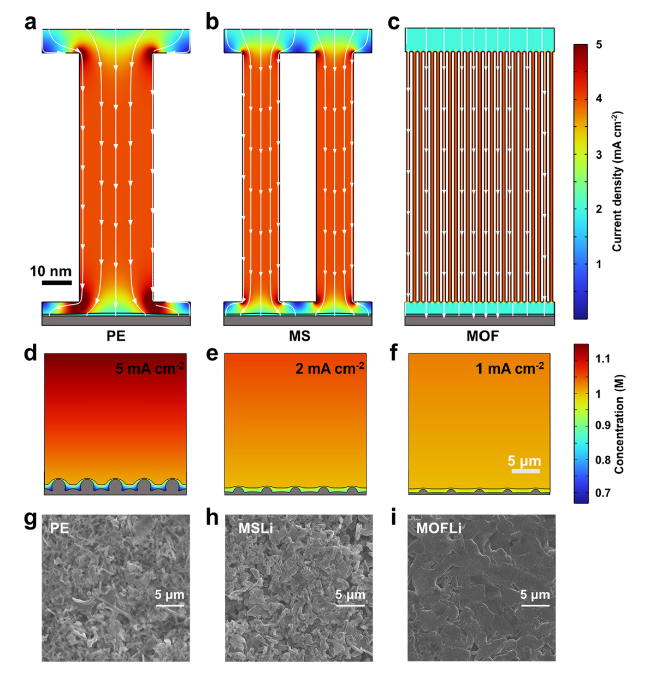
The solvation structure and ion transport dynamics of QSEs were analyzed by molecular dynamic (MD) simulations [
39]. Three models, namely MOFLi, MSLi, and LEs, were set up for MD simulations (
Fig. 2a-c). The change in the solvation structure of Li
+ ions in different electrolytes was revealed by radial distribution function (RDF) (
Fig. 2d-f). In LE, Li
+ was fully solvated by PC molecules, with an average coordination number (CN) of 4.4. When confined in porous hosts, due to the spatial restriction of pore size, a de-solvation process took place on Li
+ ions to generate a smaller solvation sheath, reflected by the reduction of CN of O(PC) (3.8 and 1.9 for MSLi and MOFLi, respectively). Moreover, the Lewis base atoms of the porous hosts (O(Zr-O-C) from MOF and O(Si-O-Si) from MS) seem to participate in the solvation of Li
+ (Fig. S14), delivering coordination number of 2.7 and 1.3, respectively. Such interaction between porous hosts and Li
+ ions results in preferential location of Li
+ ions near the O atoms in the pore wall [
40,
41,
42], contrasting with their homogeneous distribution in LE (
Figs. 2a-c and S15). Additionally, TFSI
− competed to enter the solvation sheath of Li
+ due to the decrease in CN of PC, reflected by the increase in the ratio of g(r) attributed to O(TFSI
−) and O(PC) (Fig. S16). The change in the solvation structure of Li
+ leads to different migration mechanism [
24], which is evidenced by the change in activation energy in Arrhenius plots (0.22, 0.22, and 0.09 eV for MOFLi QSE, MSLi QSE, and LE in PE membrane, respectively) (
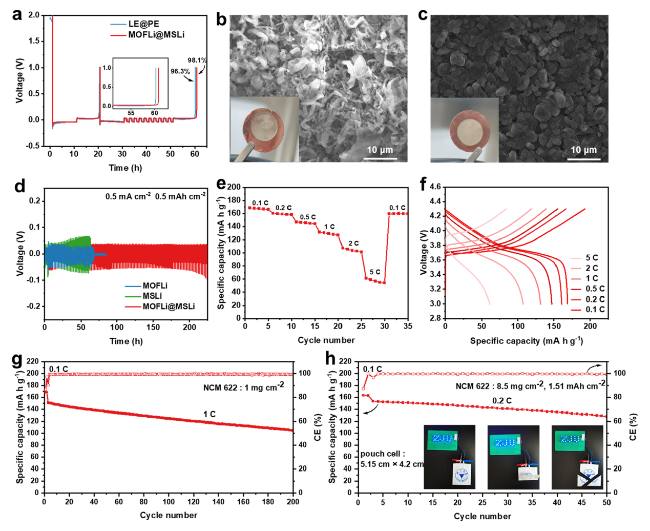
Fig. 1d).