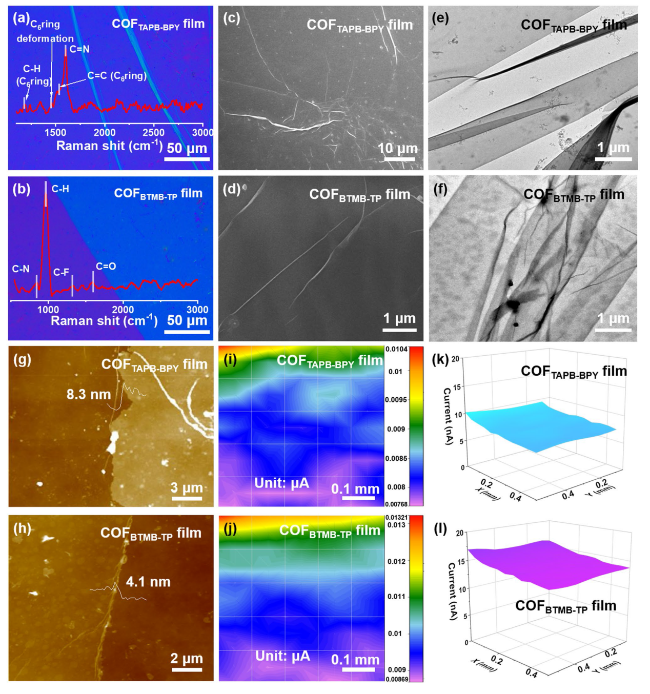
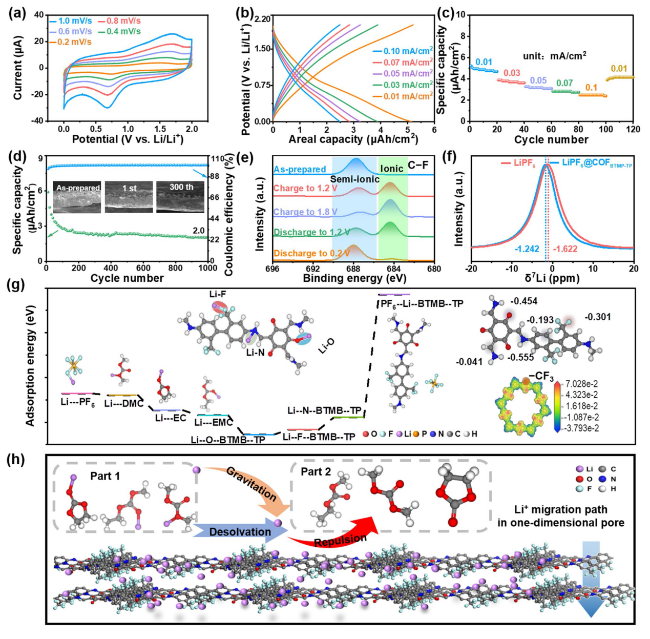
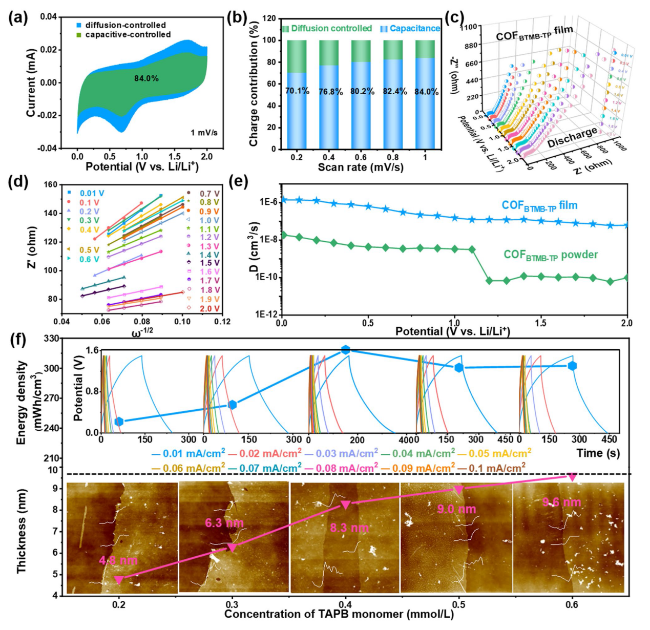
In order to figure out the internal kinetics of COF
BTMB-TP nanofilm in energy storage process, the surface capacitance and diffusion-controlled contribution for COF
BTMB-TP electrode were analyzed at a series of scan rates. In
Fig. 3a, the green-highlighted surface capacitance contribution constitutes a significant portion of CV integral area, amounting to 84% at a scan rate of 1 mV s
−1 and this contribution increases with the scan rates escalate. The proportion of surface capacitance contribution is high, reaching 70.1%, 76.8%, 80.2%, 82.4% and 84.0% at scan rate of 0.2, 0.4, 0.6, 0.8 and 1.0 mV s
−1, respectively (
Fig. 3b). This phenomenon explains the excellent charge storage kinetics behavior of COF
BTMB-TP electrode, which perfectly match the capacitive kinetic process of the LIC cathode. This kinetic process can be also effectively evaluated by ion diffusion coefficient from EIS measurement. In
Fig. 3c, dynamic EIS spectra of COF
BTMB-TP nanofilm at different discharge potentials exhibits the semicircular arc and straight line at high and low frequent region, respectively; these features correspond to the charge transfer resistance and ion diffusion
Warburg resistance. At the ion diffusion region, these plots of Z’ versus
ω−1/2 (
Fig. 3d) were summarized to calculate the slope values at different potentials (Fig. S5e), which were subsequently employed to calculate the Li
+ diffusion coefficient based on Eq. (6). Similarly, as a comparison, the calculated Li
+ diffusion coefficient of bulk COF
BTMB-TP can reach 5.6 × 10
−11 cm
2 s
−1 (Fig. S5f), surpassing many other electrode materials such as LiNi
0.5Mn
0.5O
2 (3.7 × 10
−13 cm
2 s
−1) [
40] and LFP/CZIF-8 (1.17 × 10
−13 cm
2 s
−1) [
41]. In view of the nano-level thickness of film, the volumetric Li
+ diffusion coefficient at different potentials (
Fig. 3e) show that COF
BTMB-TP nanofilm exhibits the significantly higher volumetric Li
+ diffusion coefficient of 1.15 × 10
−6 cm
3 s
−1, compared with that of bulk COF
BTMB-TP (1.86 × 10
−8 cm
3 s
−1). Moreover, with the increasing potential, the attenuation of Li
+ diffusion coefficient appears at 1.2 V versus Li/Li
+ for bulk COF
BTMB-TP; there is no change for COF
BTMB-TP nanofilm. This suggests the wider operating voltage range for highly ordered COF
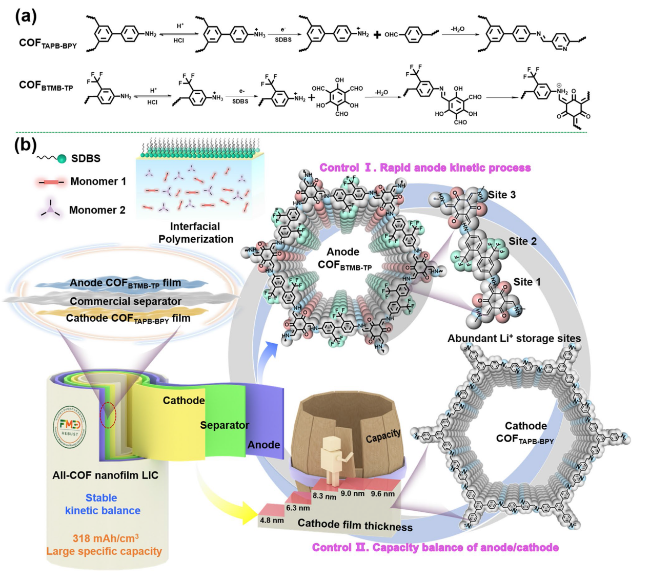
BTMB-TP nanofilm. To bridge the enormous gap of cathode/anode in the output capacity arising from different energy storage mechanisms in LICs, the thickness values of cathodic COF
TAPB-BPY are adjusted about 4.8, 6.3, 8.3, 9.0, and 9.6 nm (
Fig. 3f), by varying the concentration of adding TAPB at 0.2, 0.3, 0.4, 0.5, 0.6 mmol L
−1 and corresponding BPY monomers (molar ratio of BPY to TAPB = 1.5) to ensure a balanced charge of Q
+ = Q
−. It can be found that the thickness of cathodic COF
TAPB-BPY is not increased significantly after adding 0.3 mmol L
−1 TAPB and 0.45 mmol/L BPY monomers. Correspondingly, the energy density of assembled LIC devices incorporated these cathodic COF
TAPB-BPY nanofilms shows the similar trend with the thickness of cathodic nanofilms. Therefore, the COF
TAPB-BPY nanofilm with a thickness of 8.3 nm was selected to serve as cathode in the LIC, matched with the COF
BTMB-TP nanofilm anode to ensure capacity balance. In the inset of
Fig. 3f, the coulomb efficiency of the assembled LIC device decreases to a certain extent at the current density of 0.01 mA cm
−2, which can be explained by the impact of internal resistance from incomplete electrode activation at the low current density.