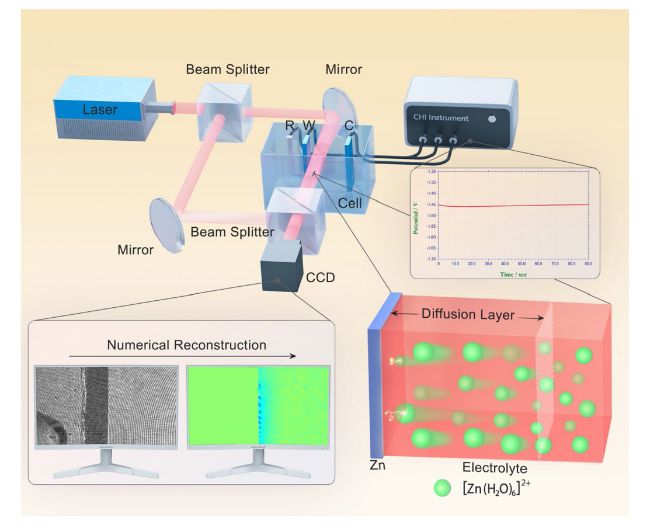
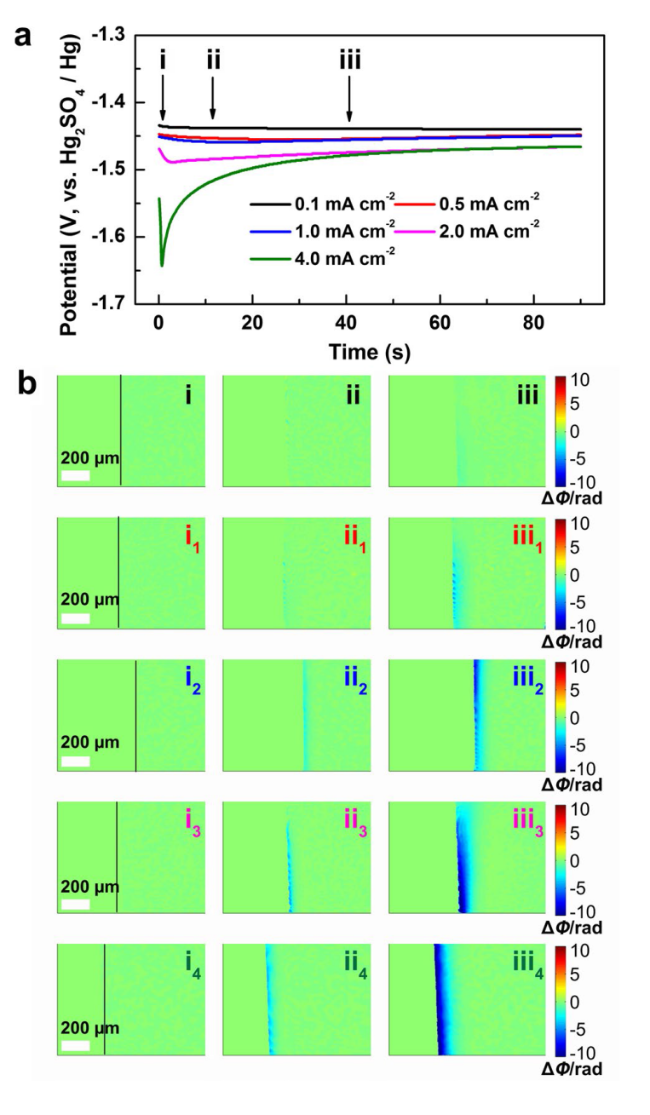
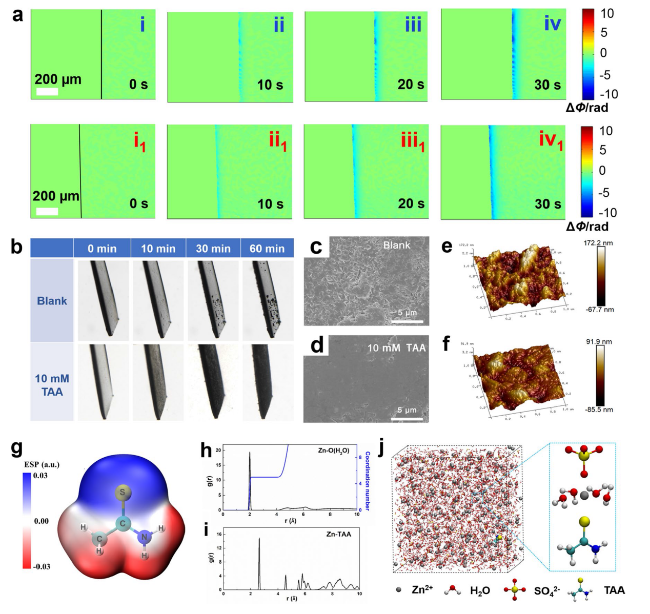
To reveal the effect mechanism of TAA on uniform Zn deposition, DFT calculations were conducted. The van der Waals potential diagram of a TAA molecule shown in
Fig. 2g displays that the nitrogen atom in TAA possesses the most negative electrostatic potential (ESP) value, indicating the concentration of negative charge and strong zincophilic affinity of this site [
54]. It is inferred that the solvation configuration of hydrated zinc ion could be easily changed by the coordination of TAA via the lone pair electrons of nitrogen atom with Zn
2+. Therefore, the molecular dynamics (MD) simulation is used to investigate the slovation structure of Zn
2+ in the electrolytes without/with TAA additive. As shown in Fig. S8a, b, in the blank ZnSO
4 electrolyte, sharp peaks of Zn-O (H
2O) and Zn-SO
4 appear at 0.2 and 0.22 nm, respectively, in the radial distribution functions (RDFs) graph. The coordination number (CN) analysis further confirms that Zn
2+ is surrounded by five H
2O and one SO
4 at the primary solvation shell, corresponding to the MD simulation shown in Fig. S8c. In comparison, with the presence of TAA, a RDF peak of Zn-TAA is located at ~ 0.26 nm, demonstrating the involvement of TAA molecules in the first solvation structure of Zn
2+ (
Fig. 2h-j). The nuclear magnetism (NMR) measurements shown in Fig. S9 present an obvious shift of
1H peak with the presence of TAA additive, experimentally confirming the destruction of hydrogen-bond network in ZnSO
4 electrolyte. The change of the coordination environment of Zn
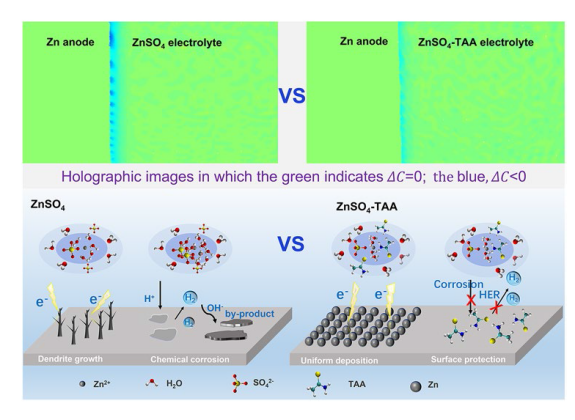
2+ is favorable for regulating the interficial ion flux and inhibiting the activity of free water at the interface. The effect of TAA electrolyte additive on the electrode/electrolyte interface is also investigated by DFT method. As shown in
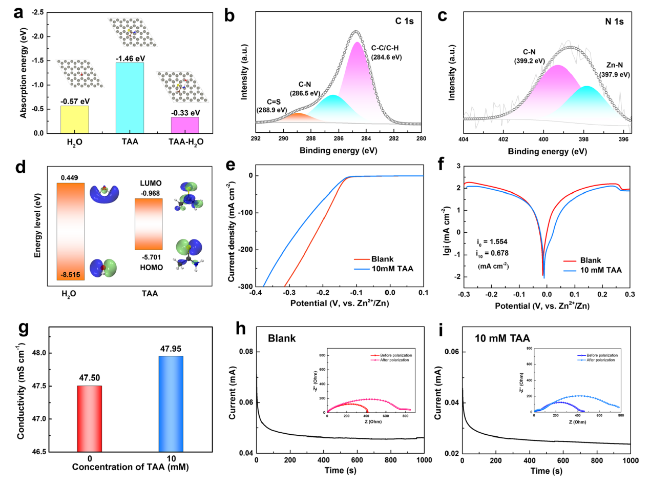
Fig. 3a, TAA molecues shows the adsorption energy of −1.46 eV on the Zn (002) surface, which is lower than that of −0.57 eV for H
2O molecules, indicating that a TAA adsorption layer is preferably formed on Zn surface. Noteably, after TAA adsorption, H
2O displays greatly weakened adsorption ability on Zn surface (adsortion energy is −0.33 eV), implying that TAA adsorption layer possesses a specific waterproof property. Therefore, it is reasonable to deduce that with the addition of TAA, a stable interface adsorption layer can be constructed on the surface of Zn anode and protect it from chemical corrosion caused by the direct contact with electrolyte. XPS survey spectra (Fig. S10) and EDS mapping (Fig. S11) of the Zn anode after 3 cycles show obvious existence of Zn, C, O, S and N elements on the surface. The characteristic peaks of C=S, C-C/C-H and C-N bonds detected in the C 1
s spectrum (
Fig. 3b) prove the adsorption of TAA on the surface of the electrode. The Zn-N peak appearing at 397.7 eV in the N 1
s spectrum (
Fig. 3c) further confirms that N atom is chemically bonded to Zn atom site.