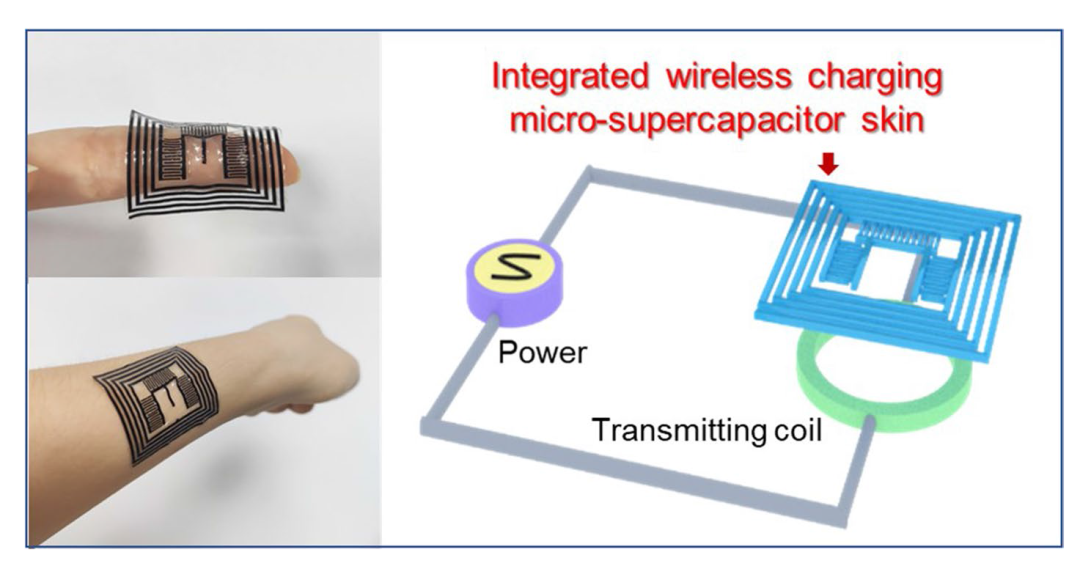
Electrochemical performances of MSC were measured. The IWC-MSC contains three parallel MSCs, and single MSC is chosen to test (
Fig. 3a). In
Fig. 3b, it is obvious to see that the enclosed area of cyclic voltammetry (CV) curves increases with the growth of IWC-MSC thickness. This is because the mass of the electrochemical activated materials PEDOT:PSS/[EMIM][TFSI] increases and the MSC capacitance improves. Benefitting from the high operating voltage of ionic liquid electrolyte, the MSC potential window widens to 2 V, larger than most of aqueous electrolyte-based MSC. Areal and volumetric capacitance are also calculated according to CV curves (
Fig. 3c). With the thickness improvement, the areal capacitance gradually increases to 42.3 mF cm
−2, but the volumetric capacitance decreases on the contrary. To satisfy the practical requirement for ultrathin electronic skin, the thinnest thickness (11.7 μm) of MSC electrode is selected to investigate the electrochemical performance (
Fig. 3d-i).
Figure 3d displays the CV curves of single MSC at different scanning rates ranging from 20 to 100 mV s
−1. All of the CV curves demonstrate near rectangular shape, revealing that the electrical double layers are formed within the thin film MSC electrodes and solid electrolyte interface. Little deviation from rectangular shape of the CV curves is attributed by the overlapping effect of double-layer and pseudocapacitive charge storage mechanisms [
25]. Galvanostatic charge-discharge (GCD) curves are given in
Fig. 3e. The curves are near-triangle shape, confirming the dominant electric double-layer energy storage mechanism of MSC. The IR
drop at the beginning of discharging line is quite small and demonstrates a linear relationship with the current density (from 64.3 to 328.7 mA cm
−3) (Fig. S5), verifying low internal resistance of MSC device [
26].
Figure 3f reveals the volumetric capacitance of the single MSC calculated according to the current density. The highest volumetric capacitance is achieved to 11.39 F cm
−3 at the current density of 64.3 mA cm
−3 (
Fig. 3f). At a high current density of 328.7 mA cm
−3, the volumetric capacitance also has a high value of 8.6 F cm
−3, which is higher than most of micro-supercapacitors (Fig. S6) [
26,
27,
28,
29,
30,
31,
32]. Capacitance of PEDOT:PSS comes from the double-layer charges on the interface between PEDOT-rich and PSS-rich grains [
33]. According to the capacitance in
Fig. 3f, the energy and power density of MSC are obtained (
Fig. 3g). It exhibits a high energy density of 6.3 mWh cm
−3 at the power density of 64.3 mW cm
−3, and 4.8 mWh cm
−3 at the power density of 328.7 mW cm
−3, which is superior to most of micro-supercapacitors (Fig. S7) [
32,
34,
35,
36,
37,
38]. As obviously shown in Fig. S8, the MSC with PE electrodes enabled with larger capacitance than the MSC only with PEDOT:PSS electrodes. It is confirmed that the excellent capacitive behavior of PE electrodes comes from its high conductivity, which is largely improved by [EMIM][TFSI] addition. The increase in electrical conductivity is partially due to a selective removal of PSS over PEDOT. It is well known that the PEDOT:PSS shows poor electrical transport properties, which caused by excessive insulating hydrophilic PSS chains encapsulating the conductive hydrophobic PEDOT cores, and inhibits PEDOT conducting networks formation [
39]. Consequently, the treatment with ionic liquids on PEDOT:PSS is to exchange PSS to [TFSI]
− counterions and induce PEDOT chains fibrillar structure thus shortening the π-π interchain distances. As a result, the delocalization of π-electrons over the PEDOT conjugated polymer backbone brings faster charge carrier mobility, improving the conductivity of PEDOT:PSS [
18,
19]. This is verified by X-ray photoelectron spectroscopy (XPS) spectrum in Fig. S9, which shows sulfur (S) 2
p peaks of PEDOT:PSS and PEDOT:PSS/[EMIM][TFSI] of PE electrode. S atoms of thiophene in PEDOT and of sulfonate in PSS have different binding energies: the lower energy peaks (164.6 and 163.4 eV) correspond to the S atoms in PEDOT and the higher energy peaks (169 and 167.8 eV) correspond to the PSS, respectively. It is obvious to see that the PSS energy peaks of PEDOT:PSS shift to lower energy in PEDOT:PSS/[EMIM][TFSI], demonstrating weaker interactions between PEDOT and PSS [
40].