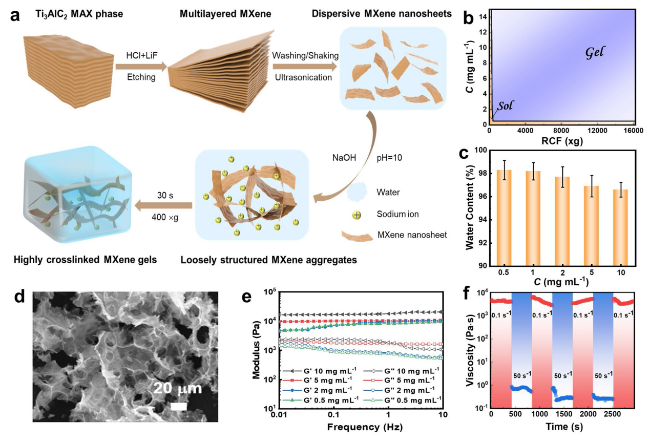
The electrochemical performance of the centrifugation-assisted MXene gel was evaluated to probe its potential applications as a freestanding electrode for supercapacitors. The advantages of using such freestanding gel electrodes include their facile fabrication and no use of inactive binders or conductive additives [
12]. Representative photographs of an electrode produced from the centrifugation-assisted Ti
3C
2T
x gel are displayed in Fig. S22. Figure S23 evidences that the gel could maintain a long-term structural stability with no collapse or swelling occurring after immersed in a water reservoir for about one month, thus being suitable for directly used as a free-standing electrode without drying into an aerogel or printing into a pattern [
58]. The water content before and after immersion was determined to be 96.4 ± 0.9 wt% and 96.9 ± 0.7 wt%, respectively.
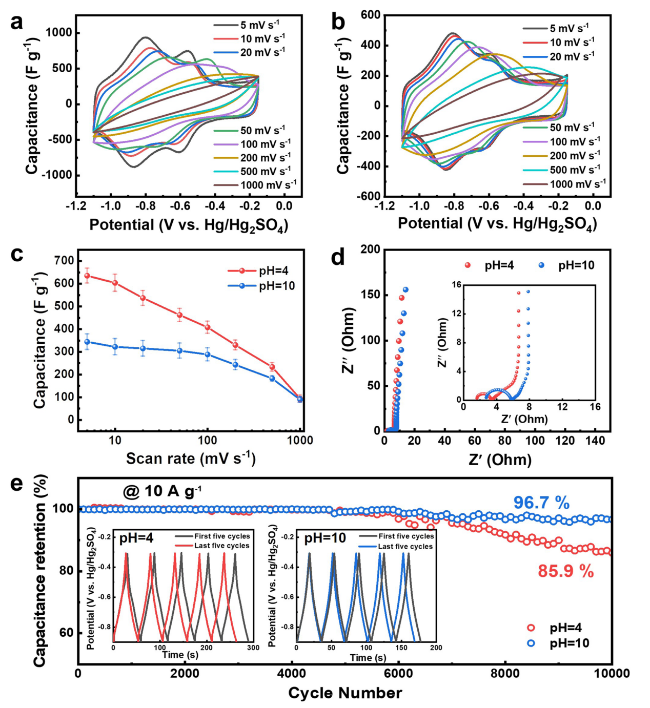
Figure 4a,
b shows that the gel yielded cyclic voltammetry (CV) curves with two pairs of redox peaks detected at ~ -0.9 and -0.6 V (vs
. Hg/Hg
2SO
4) in the potential range between -1.1 and -0.15 V at both pH 4 and 10, featuring a signature pseudo-capacitive behavior of Ti
3C
2T
x MXene [
2]. The redox peaks can be reserved as the scanning rate attained as high as 200 mV s
-1, indicative of a high surface utilization of Ti
3C
2T
x nanosheets and a good rate capability [
12]. Figure S24 displays the galvanostatic charge-discharge (GCD) profiles of the Ti
3C
2T
x gel at current densities between 0.1 and 50 A g
-1, where the charge/discharge curves for both pH 4 and 10 gels were nonlinear with no presence of apparent potential platforms, suggestive of a synergistic contribution of pseudo- and double-layer capacitances. The discharge time of a pH 4 gel was also much longer than that of a pH 10 gel at the same current density, corresponding to its much higher specific capacitance, which was consistent with the experimental results of the CV curves.
Figure 4c reveals a remarkable specific capacitance at between 635.2 ± 34.3 and 407.9 ± 29.1 F g
-1 for the pH 4 MXene gel in a scanning rate range from 5 to 100 mV, which consequently exhibits a massive potential for creating high-performance energy storage materials, as well as a satisfactory rate capability as it was able to produce a capacitance of 233.1 ± 11.7 F g
-1 even when the scanning rate attains as high as 500 mV. While excellent capacitances such as 322.1 ± 37.6 F g
-1 at 10 mV s
-1 and 288.4 ± 26.9 F g
-1 at 100 mV s
-1 can also be obtained when using the pH 10 Ti
3C
2T
x gel with a strongly improved rate performance, given its capacitance did not fall significantly over a broad scanning speed range from 5 to 500 mV. It is worthwhile to note that the specific capacitance of pH 4 MXene gel (e.g., 635.2 ± 34.3 F g
-1 at 5 mV s
-1 and 603.7 ± 35.7 F g
-1 at 10 mV s
-1) was very likely to be the highest among the MXene gel electrodes using the same three-electrode cell configuration with the same Hg/Hg
2SO
4 reference electrode and H
2SO
4 electrolyte reported so far. It was also exceedingly comparable to a list of other previously reported MXene-gel-based electrodes with different reference electrodes and electrolytes as summarized in Table S2.