HIGHLIGHTS
1 Introduction
2 Advanced TMCs Catalysts
2.1 Characteristics of Catalysts for Sulfur Cathodes
2.2 Advantages of TMCs Catalysts for LSBs
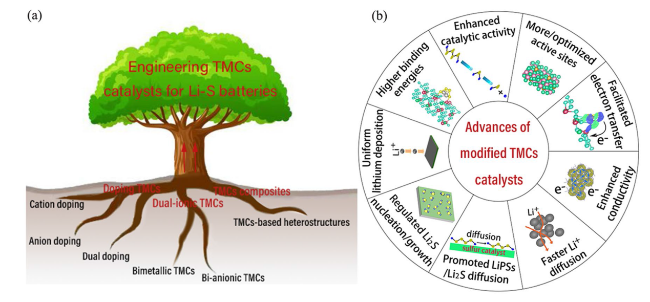
3 Engineering TMCs Catalysts
Fig. 1 a Strategies, b advances of modified transition metal compounds (TMCs) catalysts with multi-cations/anions in LSBs |
3.1 Doping Modification
3.1.1 Cation Single-Doping Modification
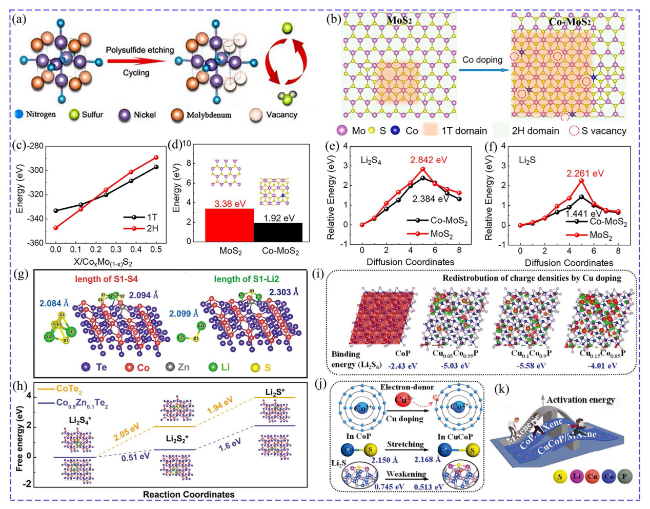
Fig. 2 a Schematic of in situ etching Mo-doped Ni3N by LiPSs [44]. Copyright: 2022, Elsevier. b Schematic of evolution of Co-doped MoS2. c Relationship of the formation energies of 1 T, 2H MoS2 and doped Co content. d Influence of Co doping on the formation energies of sulfur vacancies in MoS2. The catalytic effect of Co-doped MoS2: e Li2S4 decomposition and f Li2S decomposition [45]. Copyright: 2021, American Chemical Society. Effect of Zn doping on the catalytic effect of CoTe2: g S-S bond length of Li2S4 and S-Li bond length of Li2S. h Gibbs free energies [47]. Copyright: 2022, John Wiley and Sons. Effect of Cu-doped CoP: i unbalanced charge densities induced by Cu doping. j Schematic of electron transfer and variation of bond length, k Comparison of LiPSs/Li2S conversion barrier [54]. Copyright: 2019, John Wiley and Sons |
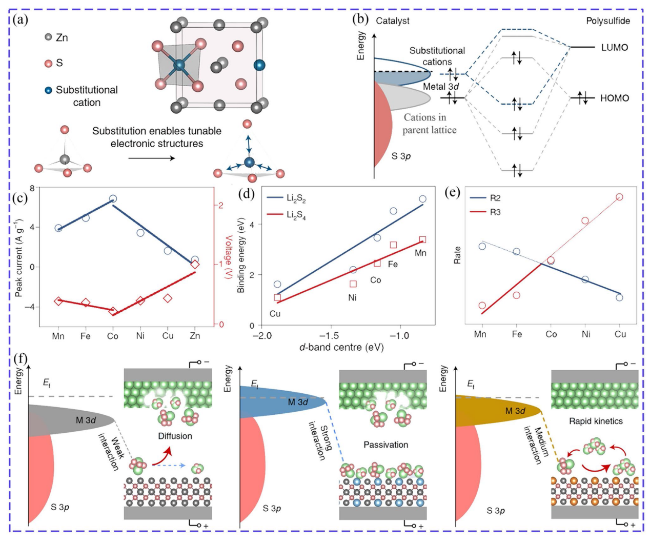
Fig. 3 Relationship between catalytic activity and adsorption ability: a Schematic of doped ZnS by substituting Zn to tune the electronic structures. b Principle of regulating electronic structure by doping. c Voltage difference and peak current of the CV of symmetric cells. d Relationship of binding energies and d-band center. e Volcano diagram of reaction rates for ZnS doped with different ions (R2: LiPSs dissociation, R3: desorption steps). f Relationship of interactions and catalytic effect [51] Copyright: 2022, Springer Nature |
3.1.2 Anion Single-Doping Modification
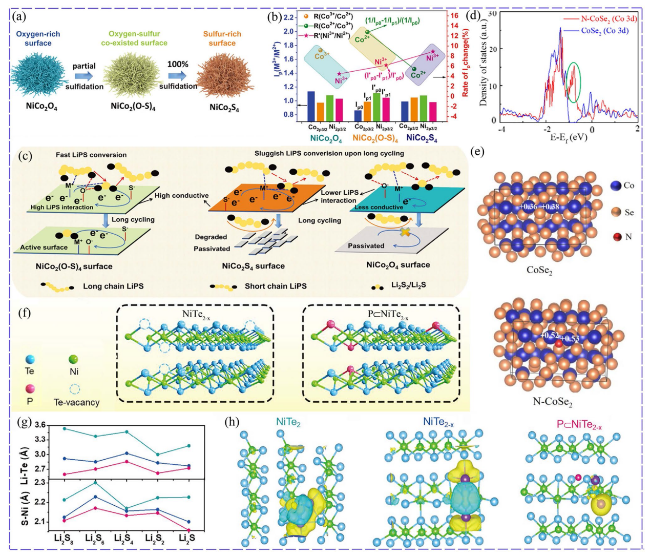
Fig. 4 Superior effect of O-doped NiCo2(O-S)4: a sulfidation process NiCo2(O-S)4 and NiCo2S4. b Ratio of M3+/M2+ 2p3/2 peak intensity (Ip) and the rate of Ip change. c NiCo2(O-S)4 with high conductivity and LiPSs affinity greatly reserved the catalysis active surface [62]. Copyright: 2022, John Wiley and Sons. Improvement of catalytic effect by N-doped CoSe2: d PDOS of CoSe2 and N-CoSe2. e Charge number of Co in CoSe2 and N-CoSe2 [63]. Copyright: 2020, American Chemical Society. P-doped NiTe2−x: f Schematic illustration of P-doped NiTe2−x at Te vacancies. g Difference of Li-Te and S-Ni bond lengths formed between LiPSs and various catalysts. h Charge density difference between Li2S and catalysts, the yellow (blue) distribution corresponds to charge accumulation (depletion) [65]. Copyright: 2022, John Wiley and Sons |
3.1.3 Dual-Doping Modification
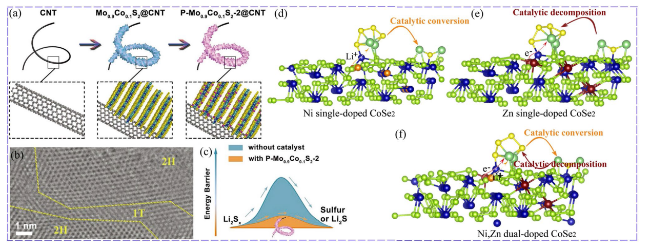
Fig. 5 Co, P co-doped MoS2: a Schematic illustration of Co, P co-doped MoS2 (P-Mo0.9Co0.1S2). b HRTEM images of Co, P co-doped MoS2. c Energy barrier of LiPSs conversion [69]. Copyright: 2019, John Wiley and Sons. Schematic illustration of the functions of different catalysts: d Ni-doped CoSe2, e Zn-doped CoSe2, and f Ni/Zn-doped CoSe2 [55]. Copyright: 2021, John Wiley and Sons |
3.2 Multi-Ionic TMCs
3.2.1 Multi-Metallic TMCs
3.2.1.1 Bimetallic TMCs
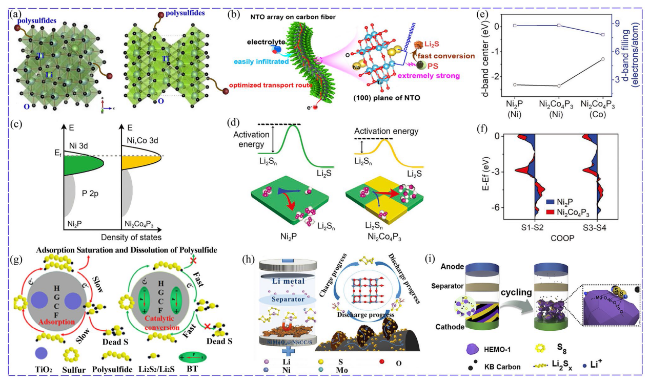
Fig. 6 a Different bonds formed between Li4Ti5O12, TiO2 and LiPSs [83]. Copyright: 2019, Elsevier. b Advantages of Na2Ti6O13 array [84]. Copyright: 2021, American Chemical Society. c DOS of Ni/Co phosphides. d Activation energy of Li2S nucleation on Ni2P and Ni2Co4P3. e d-band center of Ni2P and Ni2Co4P3. f COOP diagram of the S1-S2 and S3-S4 bonds of Li2S4 [90]. Copyright: John Wiley and Sons. g Schematic diagram of TiO2 as adsorption material while BaTiO3 (BT) as electrocatalyst [91]. Copyright: 2020, Elsevier. h Functions of NiMoO4 for LiPSs [98]. Copyright: 2021, John Wiley and Sons. i Schematic diagram of high-entropy metal oxide (HEMO) for adsorbing LiPSs [110]. Copyright: 2019, Elsevier |
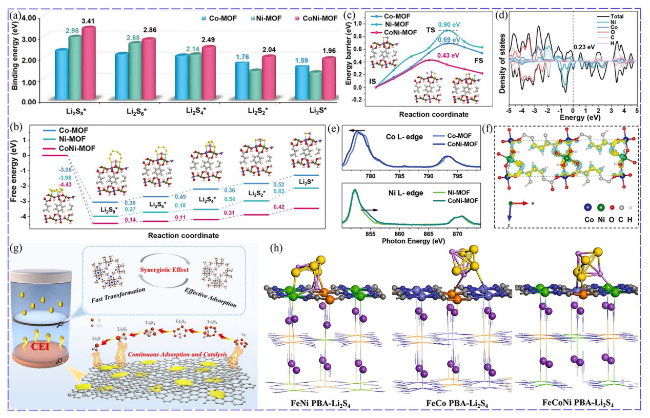
Fig. 7 Synergistic catalysis of NiCo-MOF: a Binding energies. b Free energy for the discharging process. c Li2S decomposition energy barrier. d Calculated DOS. e Co L-edge and Ni L-edge XANES spectra of the MOFs. f Charge density difference of CoNi-MOF [104]. Copyright: 2021, John Wiley and Sons. g Synergistic effect of Zn and Co sites in bimetallic Zn/Co-ZIF MOF [12]. Copyright: 2022, Elsevier. h The strongest interaction obtained for FeCoNi-PBA with the increase in adsorption sites [113]. Copyright: 2021, Elsevier |
3.2.1.2 Medium/High-Entropy TMCs
3.2.2 Bi-anionic TMCs
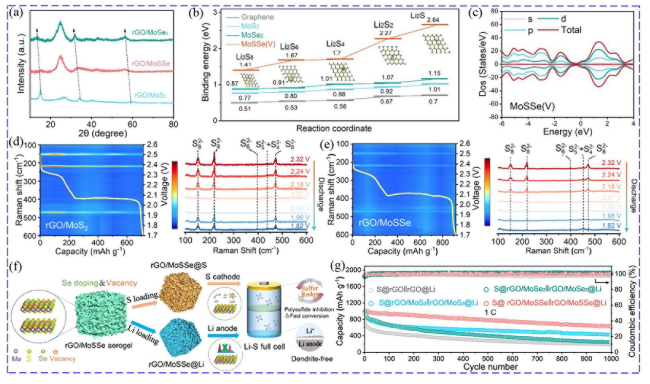
Fig. 8 a XRD of rGO/MoS2, rGO/MoSSe and rGO/MoSe2. b Comparison of binding energies of LiPSs and MoS2, MoSe2, MoSSe (V). c DOS states. d, e In situ Raman characterization of cells with rGO/MoS2 and rGO/MoSSe. f Schematic of rGO/MoSSe as a host for both sulfur and lithium. g Cycling performance at 1C [124]. Copyright: 2022, American Chemical Society |
3.3 TMCs-Based Heterostructure Composites
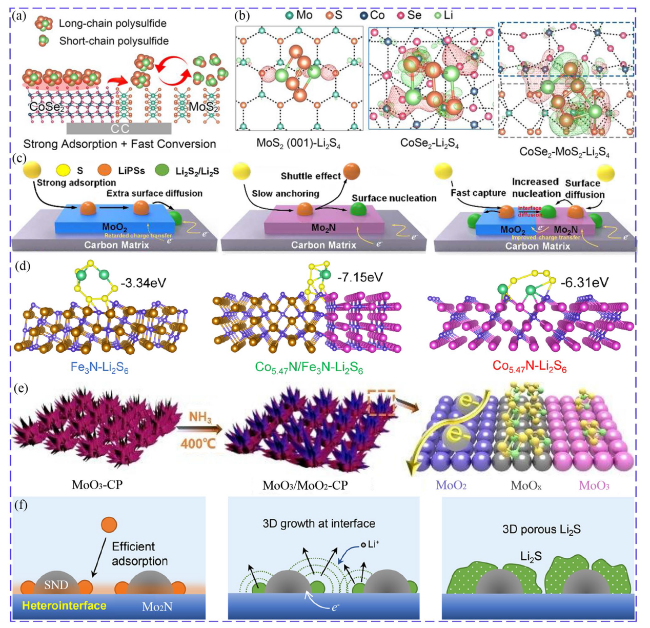
Fig. 9 a Synergistic adsorption and catalysis of CoSe2/MoS2 heterostructure for LiPSs. b electron density differences of Li2S4 on MoS2, CoSe2 and CoSe2/MoS2 (the red and green regions represent negative and positive change [137]. Copyright: 2021, John Wiley and Sons. c Comparison of LiPSs conversion and Li2S precipitation on MoO2, Mo2N and MoO2-Mo2N [140]. Copyright: 2020, Elsevier. d Binding energies between Fe3N, Co5.47N/Fe3N, Co5.47N and Li2S6 [146]. Copyright: 2022, Elsevier. e Schematic diagram of MoO3/MoO2 [147]. Copyright: 2020, Royal Society of Chemistry. f Li2S growth at the SnO2-Mo2N interfaces in 3D model [149]. Copyright: 2021, American Chemical Society |
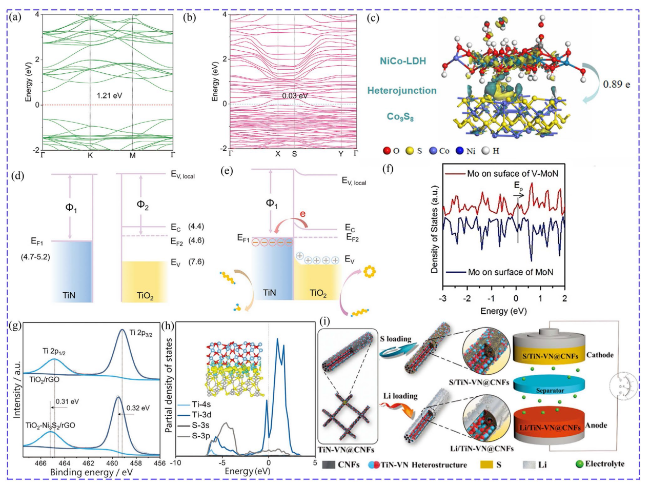
Fig. 10 Band structure of a ZnSe and b CoSe-ZnSe [151]. Copyright: 2021, John Wiley and Sons. c Electron density difference of NiCo-LDH/Co9S8 [153]. Copyright: 2020, Elsevier. Energy band of TiN and TiO2 d before and e after placing in contact [158]. Copyright: 2020, John Wiley and Sons. f DOS of Mo of V-MoN and MoN [159]. Copyright: 2018, John Wiley and Sons. g XPS of Ti 2p peaks of TiO2/rGO and TiO2-Ni3S2/rGO. h PDOS of Ti and S atom of the Ni3S2/TiO2 interface [164]. Copyright: 2020, John Wiley and Sons. i Schematic diagram of TiN-VN@CNFs used as both Li host and S host to construct full battery [165]. Copyright: 2019, John Wiley and Sons |
4 Conclusion and Prospects
Table 1 A brief summary of the improvement of cathode performance by different modified TMCs catalysts |
| Catalysts | Current density (C) | Initial capacities (mAh g−1) | Cycling number | Decay rate (%) | Promoted mechanism | References |
|---|---|---|---|---|---|---|
| Ni-WS2 WS2 | 0.2 | 1160.8 963.5 | 100 | 54.5 55.1 | More chemical anchoring sites, enhanced catalytic activity with surface defect | [36] |
| Ni-MoS2 MoS2 | 0.2 | 1343.6 1287.8 | 100 | 59.5 52.7 | Better adsorption ability, increased catalytic activity more active sites | [39] |
| Ni0.2Mo0.8N Mo2N Ni3N | 1 | 1280.8 1130 1072 | 1400 1400 800 | 36.4 31.7 29.1 | Expanded lattice spacing, In situ etching polysulfide and generating vacancies | [40] |
| Fe(0.1)/Co3O4 Fe(0.2)/Co3O4 Co3O4 | 0.2 | 1392.6 - - | 150 | 73.1 53.91 43.13 | Multi-shelled structure, rich oxygen-defect | [54] |
| N/CoSe2 CoSe2 | 0.2 | 1341 1159 | 250 | 68.9 53.1 | New defect, closer d-band center, higher charge number of Co, shorter Co-S bonds and weakened S-S and Li-S bonds | [59] |
| NiSe2 P-NiSe2 | 1 | 1012.5 931.7 | 500 | 72 61.5 | Higher electron densities, enhanced electron transfers | [62] |
| P-NiTe2-x NiTe2-x NiTe2 | 0.2 | 1309 1270 1207 | 300 | 86.2 - - | Bonds reconstruction, electron densities redistribution | [61] |
| NiCo-LDH-Se-1 NiCo-LDH-Se-2 NiCo-LDH-Se-4 | 2 | - 1332 - | 1000 | 31.6 80.3 74.9 | Improved conductivity, optimized electronic structure, abundant active site | [65] |
| V2O5 LiV3O8 | 0.1 | 1162 1254 | 100 | 69.5 77.3 | Improved adsorption ability | [81] |
| TiO2 BaTiO3 | 0.2 | 913 941 | 120 | 79.5 91.2 | Self-polarization of BaTiO3, strong interaction with LiPSs | [87] |
| Ni3ZnC0.7 Ni3C | 1 | 1275.8 934.6 | 200 | 67.4 - | Extra lithiophilic sites of Zn | [90] |
| HEO CNF | 1 | 879.6 544.2 | 500 | 63.5 - | Synergistic effect of multiple cations, abundant active site | [99] |
| NiCo-MOF Ni-MOF Co-MOF | 0.1 | 974 761 638 | 80 | 92 - - | Different catalytic function of Ni and Co, charge redistribution | [12] |
| FeNi-PBA FeCo-PBA FeCoNi-PBA | 0.1 | 1303.2 1029.4 1234.7 | 100 | 15.9 36 36.5 | Multi-metal synergistic adsorption | [109] |
| CoSe2/MoS2 CoSe2 MoS2 | 0.1 | 1425.3 1092.4 1288.4 | 50 | 84.6 64.1 67.8 | Stronger adsorption ability of CoSe2, higher catalytic activity of MoS2 | [126] |
| Ni Ni/Ni2P Ni2P | 1 | 721.3 836.1 770 | 500 | 60.2 76.1 72.1 | Enhanced conductivity, charge transfer, and adsorption | [127] |
| MoS2 MoN MoS2-MoN | 0.2 | - - 1100 | 100 | 64.9 81.4 93.9 | Catalyzing LiPSs by MoN, promoting Li+ diffusion by MoS2 | [130] |
| CoSe-ZnSe ZnSe | 0.2 | 1260 - | 100 | 74.8 45.1 | Charge redistribution and lattice distortion of heterointerface | [140] |
| V8C7-VO2 V8C7 | 4 | 765.3 666.9 | 900 500 | 45.1 43.5 | Better anchoring ability, lithiophilic nature | [155] |
| TiO2/TiN TiO2 | 0.3 | 1397 1177 | 150 | 58 59.5 | Adsorptive TiO2, catalytic TiN, charge transfer from TiN to TiO2 | [169] |