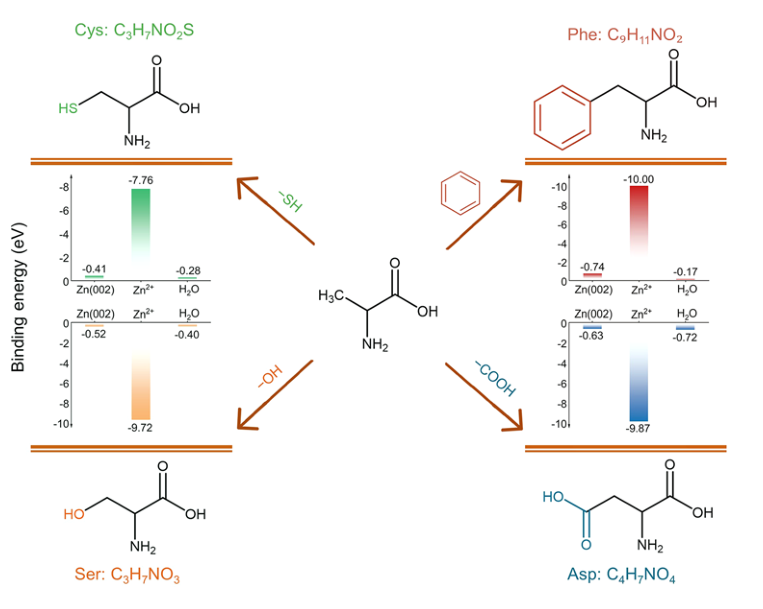
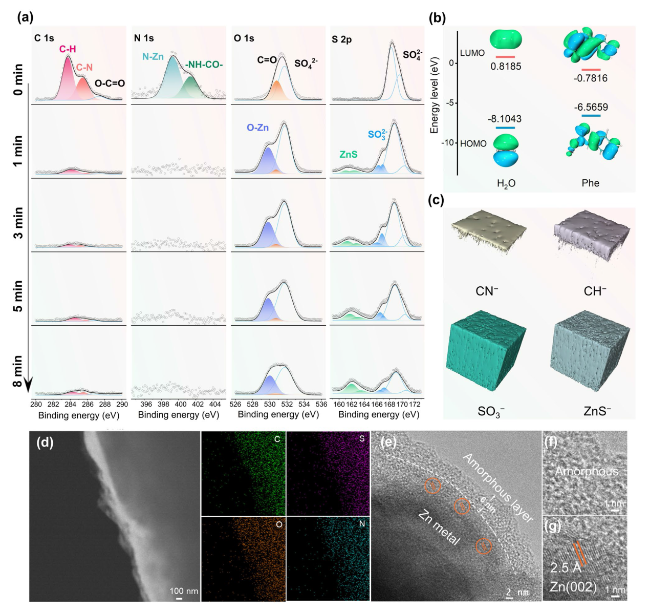
To analyze the electrolyte/electrode interface, the TOF-SIMS andXPS with Ar
+ sputtering depth profiling were employed. As shown in the XPS spectra of N 1
s (
Fig. 2a), the N-Zn (399.3 eV) and -NH-CO- (401.4 eV) bands exclusively exist at the top surface (0 min, before sputtering) of Zn anode cycled in ZSO/Phe electrolyte, demonstrating the chemical adsorption of Phe molecules onto the Zn metal anode surface [
35]. It is obviously observed that the ZSO/Phe electrolyte exhibits better wettability at 86.9° than ZSO electrolyte at 96.8°, which further confirms the strong adsorption ability of Phe molecules on Zn anode surface (Fig. S10). However, the contact angle between ZSO/Phe electrolyte and presoaked Zn anode of Phe-contained aqueous solution increases to 98.6°, exhibiting a hydrophobic tendency. As a contrast, the contact angle between ZSO/Phe electrolyte and presoaked Zn anode of deionized water remain nearly constant of 84.9°. This is ascribed to the adsorption of Phe molecules on Zn anode, where the hydrophobic benzene ring ligands toward the electrolyte, forming a molecular hydrophobic layer at the Zn/electrolyte interface, contributing to suppressing water-mediated parasitic side reactions. Additionally, the signals of C-H (283.8 eV) and C-N (285.5 eV) in the C 1
s spectrum are detected at the top surface [
23], diminishing significantly after 1 min of Ar
+ etching. Meanwhile, the characteristic peak of C = O (530.8 eV) in the O 1
s spectra displays relatively high intensity before sputtering but gradually fades away after 8 min of etching, indicating an organic layer predominantly covering the electrode top surface [
48]. The higher highest occupied molecular orbital (HOMO, − 6.5659 eV) and lower lowest unoccupied molecular orbital (LUMO, − 0.7816) of Phe molecules compared to H
2O molecules (
Fig. 2b) promote preferential electron acquisition by adsorbed Phe. These facilitate Phe reduction on the Zn anode surface ahead of H
2O to form a hydrophobic organic layer with minute quantities of H
2 release [
48]. In contrast, a series of new characteristic peaks in O 1
s and S 2
p spectra corresponding to ZnO (529.8 eV) [
49], ZnS (161.1/162.5 eV) [
50], and ZnSO
3 (166.3/166.9 eV) [
50] emerge after 1 min of Ar
+ etching, and the intensities strengthen with increased etching time. This phenomenon demonstrates the
in situ formation of ZnO, ZnS, and ZnSO
3 inorganic layers at the bottom of SEI. As shown in Fig. S11, the Phe molecule delivers higher binding energy of − 4.47 eV with SO
42− compared to H
2O, which enables the SO
42− ions migrate to Zn anode surface favorably through Phe adsorption layer to be reduced by H
2 with
in situ formation of ZnO-ZnS-ZnSO
3 inorganic layer [
48]. Consequently, the Zn 2
p spectrum exhibits significantly weaker intensity at the top surface (0 min, before sputtering) compared to the sample under Ar
+ etching (Fig. S12), attributable to both the chemically adsorbed film and the
in situ organic SEI layer [
51]. Furthermore, the existence of SO
42− (168.5/169.9 eV) on the Zn anode surface primarily originates from the precipitation of ZnSO
4 salt [
48]. The TOF-SIMS was conducted to investigate the SEI chemistry induced by the Phe additive. Notably, the normalized intensity of organic CN
− and CH
− species decreases rapidly with increased sputtering depth, while that of ZnS, ZnO, and ZnSO
3 inorganic compounds increases significantly (Fig. S13). These observations indicate the uniform coverage of an organic layer composed of CN
− and CH
− species on the dense ZnS, ZnO, and ZnSO
3 inorganic layer, forming a gradient structure, as confirmed through three-dimensional (3D) visualization (
Figs. 2c, S14, and S15). The organic layer enables the electrode/electrolyte interface with considerable deformability to accommodate the volume changes in repeated charging/discharging processes and excellent hydrophobic properties to protect the Zn anode from corrosion caused by active water molecules. Meanwhile, the ZnO/ZnS/ZnSO
3-rich bottom layer maintains high mechanical rigidity and rapid Zn
2+ transport capability, enabling uniform Zn
2+ ions flux for dendrite-free Zn deposition. On the contrary, the Zn anode cycled in ZSO electrolyte presents thimbleful C element and inorganic ZnO/ZnS/ZnSO
3 species (Fig. S16). Obviously, almost no N element exists on the surface. To accurately determine the composition and structure of SEI formed in ZSO/Phe electrolyte, high-resolution TEM (HRTEM) was performed. The sample, prepared by depositing Zn
2+ on copper micro-grids within Phe/ZSO electrolyte, revealed an ultrathin amorphous layer on the outer layer of the electrodeposited Zn particles in the high-angle annular dark-field (HAADF) image (
Fig. 2d). This amorphous layer is attributed to the
in situ formed adsorption layer and SEI induced by the Phe additive. The energy-dispersive X-ray spectroscopy (EDS) mapping confirms the uniform distribution of the
in situ constructed organic and inorganic hybrid SEI layer composed of C, N, O, and S elements on the surface of the Zn anode (Figs. 2d and S17). Furthermore, HRTEM results reveal that the amorphous organic layer has a thickness of about 6 nm, and numerous lattice fringes of Zn (002) are observed inside the SEI layer (
Fig. 2e-g). This suggests that the newly plated Zn is deposited through the protective interphase and preferentially grows with a favorable (002) plane during the initial deposition stage. As a result, a dual effect is achieved, simultaneously suppressing the corrosion of water molecules and inhibiting the formation of Zn dendrites.