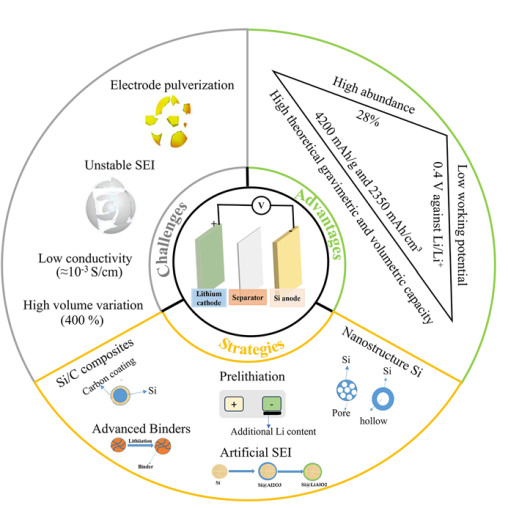
HIGHLIGHTS
1 Introduction
Fig. 1 A schematic diagram illustrating the strategies for addressing the multifaceted challenges inherent to Si-based anodes in LIBs |
Table 1 Electrochemical properties of several materials used as anode in LIBs |
| Anode material | Theoretical gravimetric capacity (mAh g−1) | Working potential (V) | Volume variation (%) |
|---|---|---|---|
| Si | 4200 | 0.4 | 400 |
| Li | 3862 | 0 | 100 |
| Ge | 1625 | 0.5 | 370 |
| Sn | 994 | 0.6 | 26 |
| Graphite | 372 | 0.05 | 12 |
| Li4Ti5O12 | 175 | 1.5 | 1 |
| TiO2 | 167 | 0.8 | < 4 |
1.1 Electrochemical Lithiation Mechanism and Challenges of the Si Anode
Table 2 Theoretical capacity and volume variation of different phases of Li-Si |
| Phase | Theoretical capacity (mAh g−1) | Volume variation (%) |
|---|---|---|
| Si | 0 | 0 |
| LiSi | 954 | 160 |
| Li12Si7 | 1635 | 222 |
| Li2Si | 1900 | 263 |
| Li13Si4 | 3100 | 389 |
| Li15Si4 | 3590 | 390 |
| Li22Si5 | 4200 | 400 |
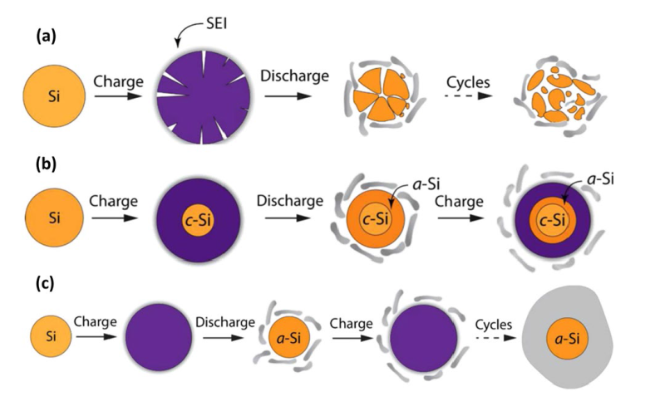
Fig. 2 Degradation Mechanisms of Si Anode—a Demonstrates the fracture and pulverization of Si electrode materials as a key challenge, b addresses lithiation retardation due to compressive stress, which reduces rate performance and lowers the effective capacity, and c shows how large volume changes during operation can induce unstable SEI growth on the electrode surface. Each panel offers a focused examination of these mechanisms, presenting critical insights into the durability and performance of Si-based anode materials. Reproduced with permission from Ref. [26]. Copyright 2017, Springer Nature |
2 Strategies to Improve the Electrochemical Performance of Si-based Anodes
2.1 Carbon-Si Composites
2.1.1 1D Carbon-Si Composites
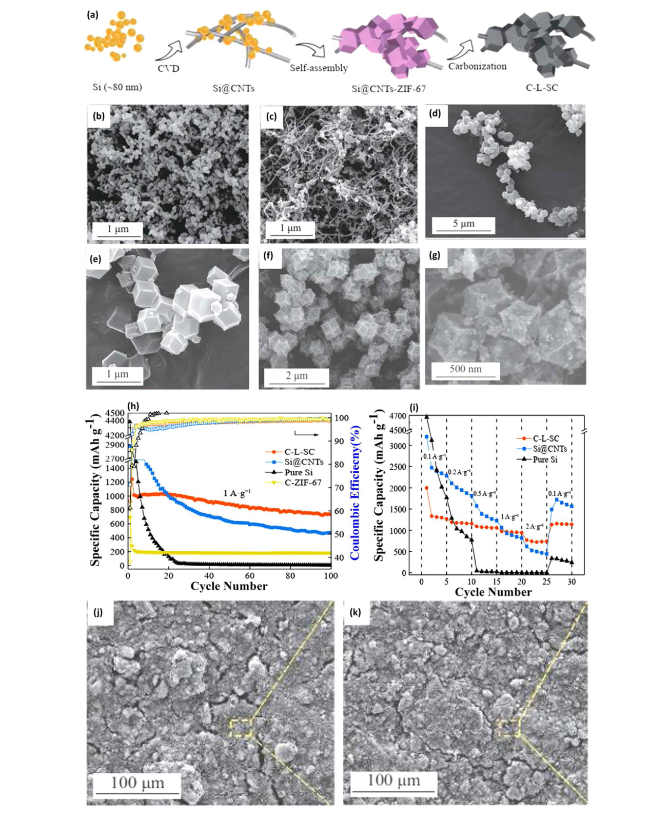
Fig. 3 Chain-like Si CNTs (C-L-SC) Preparation and Evaluation—a Schematic illustration of the preparation process for chain-like Si CNTs (C-L-SC). SEM images showcasing b the morphology of pure Si, c Si encapsulated in carbon nanotubes (Si@CNTs), d, e Si@CNTs after ZIF-67 incorporation (Si@CNTs-ZIF-67), and f, g the final chain-like Si CNTs structure (C-L-SC). h Compares the cycling performances of pure Si, Si@CNTs, C-ZIF-67, and C-L-SC electrodes, demonstrating the enhanced durability and capacity retention of C-L-SC. i Rate capability tests for pure Si, Si@CNTs, and C-L-SC electrodes, highlighting the improved performance of C-L-SC under various current densities. SEM images of C-L-SC electrode j before and k after 100 cycles at 0.5 A g−1, illustrating the structural integrity and stability of C-L-SC over extended cycling. Reproduced with permission from Ref. [42]. Copyright 2021, Springer Nature |
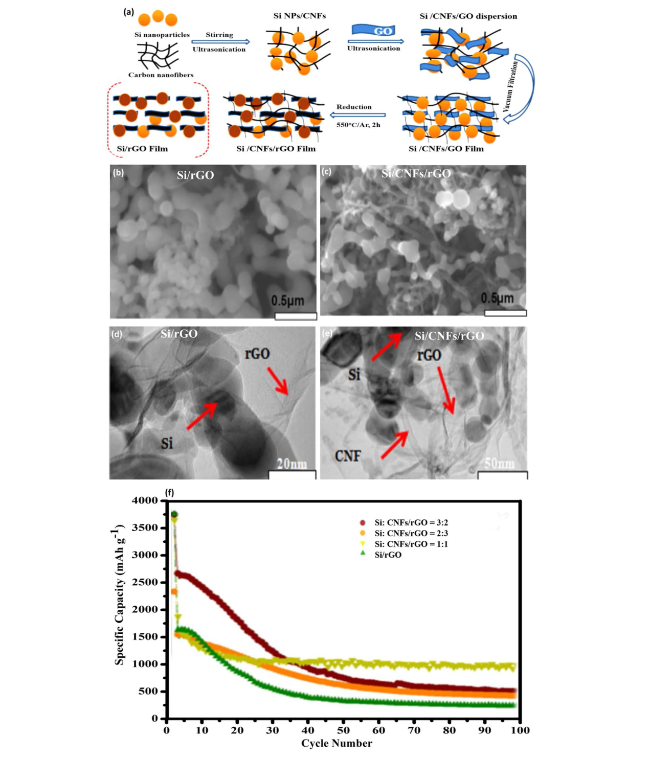
Fig. 4 Fabrication and Analysis of Si/CNF/rGO Composite Films—a Illustration of the preparation process for Si/CNF/rGO and Si/rGO composite films, outlining the steps taken to synthesize these materials. SEM images of b, c the Si/rGO and Si:CNF/rGO = 1:1 composites, respectively, showing the morphology and distribution of materials within the composites. TEM images of d, e similarly showcase the microstructure of Si/rGO and Si:CNF/rGO = 1:1 composites, providing a closer look at the nanoscale interactions and structure. f Cycling performances of the synthesized Si/rGO and Si/CNF/rGO composites at ratios of 1:1, 2:3, and 3:2, evaluated at a current density of 0.1 A g−1, highlighting their electrochemical stability and capacity retention over cycles. Reproduced with permission from Ref. [46]. Copyright 2021, Springer Nature |
2.1.2 2D Carbon-Si Composites
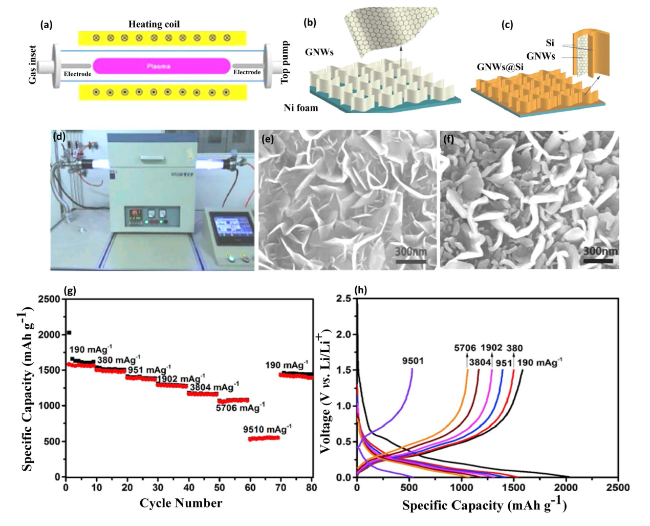
Fig. 5 RF Plasma-Enhanced Fabrication of GNWs@Si Composites and Their Electrochemical Evaluation in LIBs—a Schematic diagram of the radio-frequency plasma-enhanced horizontal tube furnace deposition system. b Schematic images depicting the growth of graphene nanowalls (GNWs) on Ni foam. c Schematic representation of GNWs@Si composite preparation on Ni foam. d Actual image of the radio-frequency plasma-enhanced horizontal tube furnace deposition system. e SEM images showcasing the morphology of GNWs. f SEM images of GNWs@Si composite, illustrating the composite’s detailed structure. g Voltage profiles of LIBs based on GNWs@Si composite at varying current densities from 190 to 9510 mA g−1. h Galvanostatic charge-discharge capacity of the same, depicting the performance across different rates. Reproduced with the permission from Ref. [52]. Copyright 2019, Elsevier |
2.1.3 3D Carbon-Si Composites
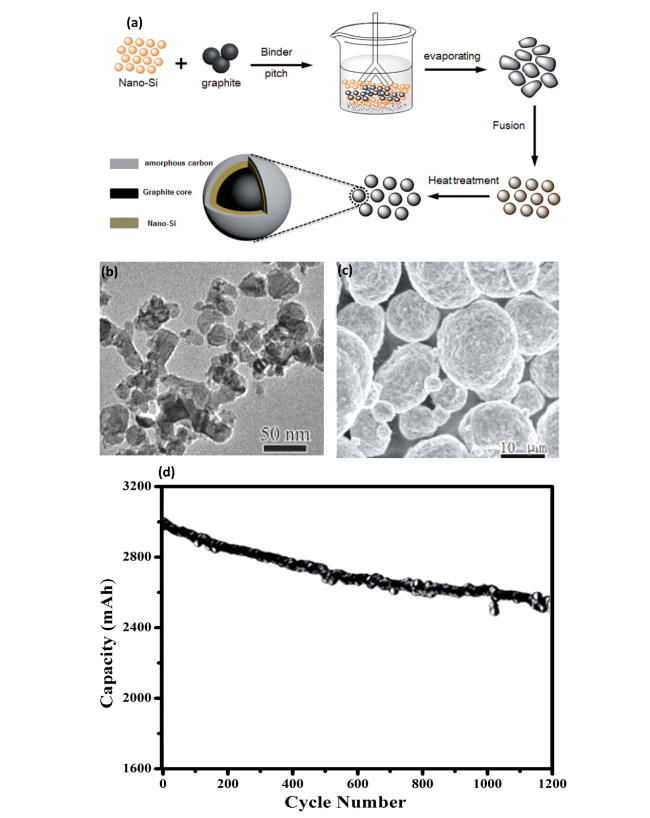
Fig. 6 Preparation and Electrochemical Performance of Si-G/C Composite for High-Energy-Density Batteries—a Schematic preparation procedure of the Si-G/C composite, illustrating the step-by-step synthesis process. b Typical TEM profiles of nano-Si, providing insights into the nanostructure of the silicon used in the composite. c SEM images of the Si-G/C composite, highlighting the microstructural integration of silicon with graphene/carbon. d Cycling performance of the Si-G/C composite in prototype full-cell high-energy-density batteries, demonstrating stability and capacity retention over 1200 cycles at a charge/discharge rate of 0.5 C. Reproduced with the permission from Ref. [59]. Copyright 2018, Royal Society of Chemistry |
Table 3 Cycle stabilities of various Si/C- based anodes for LIBs |
| Si Anodes | Current density (A g−1) | Cycle number | Remaining capacity (mAh g−1) | Refs. |
|---|---|---|---|---|
| Si/C | 0.3 | 100 | 611.3 | [62] |
| Si/p-C(N-SPC) | 0.4 | 100 | 1607 | [63] |
| Si@viod@C | - | 40 | 500 | [64] |
| Si@C@viod@C | 0.1 | 50 | ~ 1350 | [65] |
| Porous Si/C | 0.5 | 30 | 759 | [66] |
| Meso-Si/C | 1 | 1000 | 990 | [67] |
| Porous Si/C nanotubes | 0.2 | 200 | 1300 | [68] |
| Si/graphite/C | 0.5 | 300 | ~ 400 | [69] |
| Si@C@CNTs&CNFs | 0.3 | 50 | 1195 | [70] |
| Si/CNTs | 0.42 | 100 | 1000 | [39] |
| Si/CNTs | 42 | 100 | 800 | [71] |
| Si@HC/CNFs | 0.2 | 100 | ~ 1020 | [72] |
| Si/graphene | 0.1 | 200 | 1500 | [33] |
| Si/rGO | 0.1 | 100 | 1433 | [73] |
| Si/C/graphene | 0.2 | 100 | 760 | [74] |
| Si@C-rGO | 0.3 | 400 | 931 | [75] |
| M-pSi@C | 1 | 250 | 1702 | [76] |
| Si/C | 0.1 | 100 | 941 | [77] |
| Si/C | 0.5 | 100 | 605.43 | [78] |
| Si/C | 2 | 400 | 1283 | [79] |
| Si/C/rGO | 1 | 270 | 1004 | [80] |
| C-Si@graphite | 4.2 | 1000 | ~ 900 | [81] |
| C@void/Si-G | 8.4 | 200 | 1082.7 | [82] |
| Si/Cu/Cu3Si@C | 2.1 | 500 | 984 | [83] |
2.2 Nanostructured Si Anode Materials
2.2.1 0D Si-Based Anode Materials
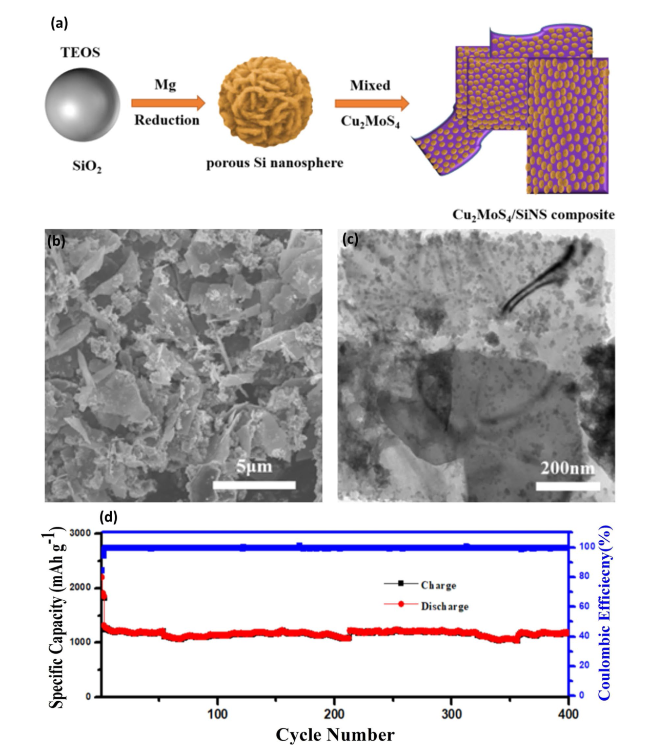
Fig. 7 Synthesis and Electrochemical Evaluation of Cu2MoS4/SiNS Composite for Anode Material—a Illustrates the synthetic strategy process for creating Cu2MoS4/SiNS through the self-assembly of composite porous Si nanospheres with Cu2MoS4, detailing the steps involved in the composite formation. b SEM image showing the morphology of the self-assembled Cu2MoS4/SiNS anode material, highlighting the microstructural characteristics. c TEM images of the Cu2MoS4/SiNS composite, providing a closer look at the nanostructure of the composite material. d Cycling performance of Cu2MoS4/SiNS demonstrated over 400 cycles at a current density of 2.0 A g−1, showcasing the material's durability and electrochemical stability. Reproduced with permission from Ref. [90]. Copyright 2021, American Chemical Society |
2.2.2 1D Si-Based Anode Materials
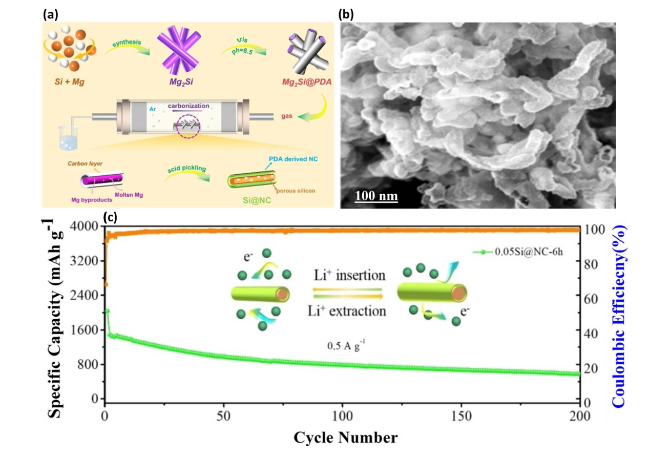
Fig. 8 Synthesis and Performance of Si@NC Nanotube Composite—a Schematic illustration of the synthesis process for the Si@NC nanotube composite, detailing the steps involved in creating this advanced material. b SEM image showcases the Si@NC nanotube, highlighting its unique morphology and structure. c Long-term cycling capability of the Si@NC nanotube composite, evidencing its robustness and potential for use in energy storage applications. Reproduced with permission from Ref. [97]. Copyright 2022, Wiley Online Library |
2.2.3 2D Si-Based Anode Materials
2.2.4 3D Si-Based Anode Materials
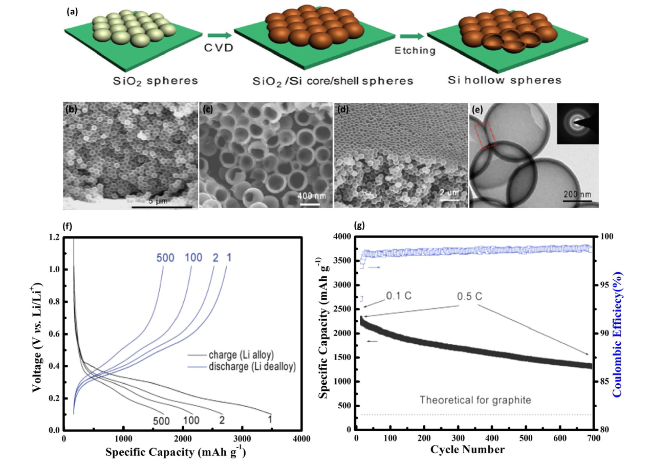
Fig. 9 Fabrication and Analysis of Hollow Si Nanospheres for Energy Storage Applications—a Synthesis process for hollow Si spheres, beginning with the coating of Si nanoparticles on a stainless-steel substrate, followed by CVD deposition of Si, and concluding with the removal of the SiO2 core via HF etching to achieve the hollow structure. b Typical cross-sectional SEM image of the hollow Si nanospheres, illustrating the uniformity and integrity of the hollow structures. c SEM side view of the same sample, offering a different perspective on the morphology. d SEM image of hollow Si nanospheres that have been scraped open with a sharp razor blade, revealing the interior empty space and confirming the hollow nature. e TEM image of interconnected hollow Si spheres, highlighting their potential for enhanced electrochemical performance through structural connectivity. f GCD profiles at a rate of 0.5 C, noting that the first cycle was performed at 0.1 C to establish baseline performance. g Compares the reversible Li discharge capacity and Coulombic efficiency (CE) of the hollow Si nanospheres against the theoretical capacity of graphite, demonstrating the superior performance and potential of hollow Si nanospheres in energy storage devices. Reproduced with permission from Ref. [108]. Copyright 2011, American Chemical Society |
Table 4 Cycle stabilities of various composites anodes comprised of different Si and carbon materials for LIBs |
| Si Anodes | Current density (A g−1) | Cycle number | Remaining capacity (mAh g−1) | Refs. |
|---|---|---|---|---|
| Si/meso-C | 2 | 50 | 700 | [111] |
| Si/void/C | 0.1 | 80 | 980 | [112] |
| Si/C | 0.372 | 100 | 1500 | [113] |
| Si/MWCNT | 0.4 | 70 | 520 | [114] |
| Si/CNTs | 0.42 | 200 | 925 | [115] |
| Si/graphene/graphite foam | 0.4 | 100 | 370 | [116] |
| Graphene/Si/C | 0.3 | 100 | 902 | [117] |
| Si NWs | 0.26 | 100 | 2000 | [118] |
| SiNTs | 0.4 | 90 | ~ 800 | [119] |
| C@SiNTs | 0.84 | 200 | 2085 | [120] |
| CNTs/SiNTs | 1.7 | 250 | 800 | [121] |
| C@Si@CNTs | 0.294 | 60 | 2200 | [122] |
| Si@C/rGO | 2 | 600 | 1100 | [123] |
| Si/C | 0.5 | 300 | 1041 | [15] |
| Si/rGO | 4 | 100 | 1521 | [124] |
| SI/C | 0.188 | 120 | 1179 | [125] |
| Si/C-CNTs | 0.5 | 200 | 1585.9 | [126] |
| Si/C | 4.2 | 500 | 1145 | [127] |
| Si/NSs@C | 0.1 | 200 | 822 | [128] |
| C/Si | 4 | 500 | 1072.2 | [129] |
2.3 SiOx/C Type Composites and Incorporating Si with Non-Carbonaceous Materials
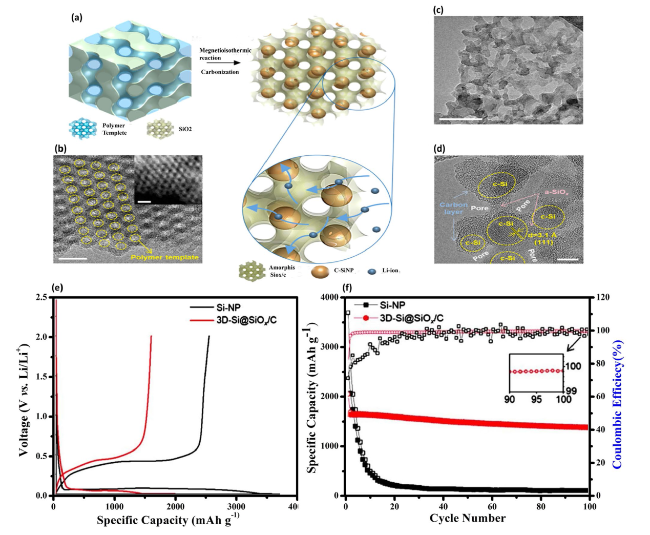
Fig. 10 Development of a Gyroid 3D Network of Si@SiOx/C for Anode Applications—a Schematic illustration of the synthetic route to fabricate the 3D-Si@SiOx/C structure via one-pot magnetoisothermic reduction and carbonization of KIT-6, incorporating the polymer template for structure formation. b TEM image of the highly ordered double-gyroid KIT-6 including the polymer template, with a high-angle annular dark-field scanning TEM (HAADF-STEM) image shown in the inset, demonstrating the intricate gyroid structure and its uniformity. c, d TEM images of the 3D-Si@SiOx/C network, with c providing a broader view of the structure and d a high-resolution TEM (HRTEM) image highlighting the detailed crystalline structure. Scale bars are 20 nm for (b, including inset) and (c), and 5 nm for (d). e Galvanostatic charge-discharge (GCD) profiles of the 3D-Si@SiOx/C and Si nanoparticle (NP) anodes, showing the electrochemical performance comparison. f Cycling performances and Coulombic efficiencies (CEs) of the 3D-Si@SiOx/C and Si NP anodes at a current density of 200 mA g−1, indicating the enhanced durability and efficiency of the 3D-Si@SiOx/C structure. Reproduced with permission from Ref. [130]. Copyright 2019, American Chemical Society |
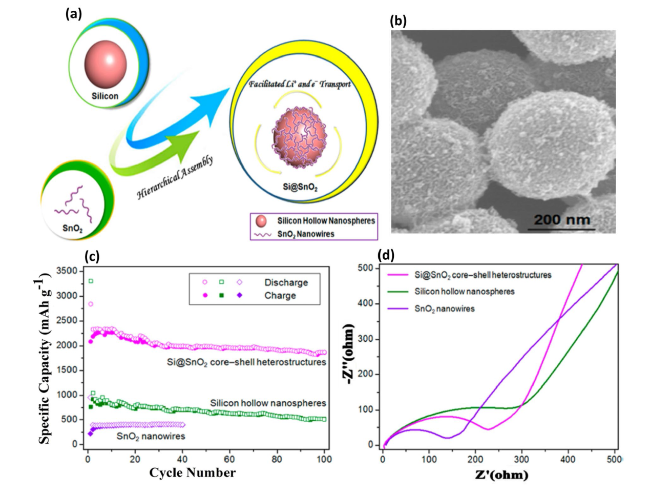
Fig. 11 Formation and Evaluation of Si@SnO2 Core-Shell Heterostructures for Lithium-Ion Batteries—a Illustrates the process in which Si hollow nanospheres and SnO2 nanowires undergo sonication in tetrahydrofuran (THF), resulting in their spontaneous assembly through van der Waals interactions to form Si@SnO2 core-shell heterostructures. This combination leverages the properties of both materials to enhance lithium and electron transport. b SEM images provide a detailed view of the Si@SnO2 core-shell structures, showcasing the uniformity and quality of the heterostructures. c Cycle behaviors of Si@SnO2 core-shell heterostructures, Si hollow nanospheres, and SnO2 nanowires at a current density of 500 mA g−1, highlighting the superior cycling stability of the core-shell structures. d Nyquist plots illustrate the electrochemical impedance of the Si@SnO2 core-shell heterostructures, Si hollow nanospheres, and SnO2 nanowires, offering insights into the improved ionic and electronic conductivities of the core-shell configuration. Reproduced with permission from Ref. [139]. Copyright 2016, American Chemical Society |
Table 5 Electrochemical capabilities of Si with other materials as anode for LIBs |
| Si Anodes | Current density (A g−1) | Cycle number | Remaining capacity (mAh g−1) | Refs. |
|---|---|---|---|---|
| Si/Ag | 0.84 | 100 | 1163 | [109] |
| Si/Cu | 2.1 | 60 | 2002 | [143] |
| Si/ZnO | 0.84 | 210 | 1500 | [144] |
| SiNPs/V2O5 | 2.1 | 50 | 932 | [145] |
| SiNPs/LIPON | 2.1 | 100 | 1050 | [146] |
| SiNPs/GR/Li7P3S11 | 4.2 | 500 | 1332 | [147] |
| SiNPs/GR/Li4SiO4 | 0.21 | 40 | 1893 | [148] |
| P0.5% Si-Cu | 0.1 | 60 | 1048 | [149] |
| Si@LPO@void@FC | 1 | 500 | 569 | [150] |
| β-Si3N4/Si | 5 | 100 | 480 | [151] |
3 Other Factors Affecting the Performance of Si-Based Anode Materials
3.1 Artificial Solid Electrolyte Interphase for Si Anode
3.1.1 Electrolyte Modulation
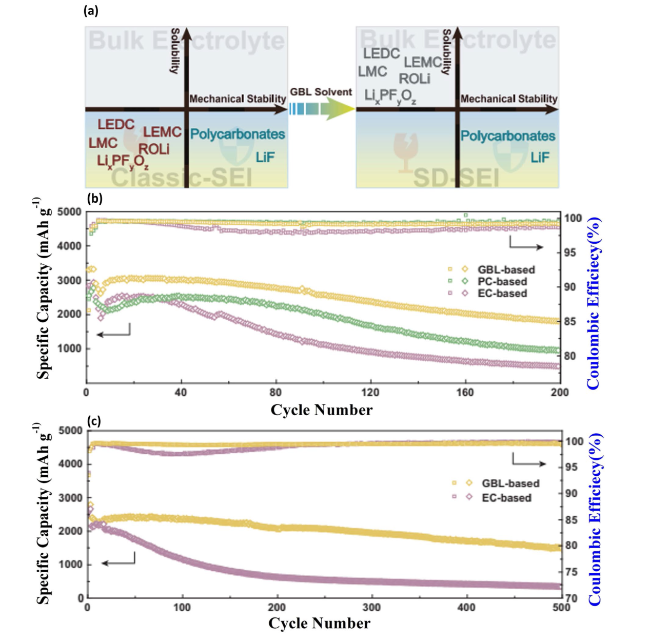
Fig. 12 Solvent-Induced Selective Dissolution for SEI Optimization in Si Anodes—a Schematic representation of the solvent-induced selective dissolution process for solid electrolyte interphase (SEI) layers, highlighting how SEI components are differentiated based on their solubility in various solvents. This technique aims to refine SEI composition for improved anode performance. b Cycling performance and Coulombic efficiencies (CEs) of micron-sized Si anodes tested in different electrolytes at a charge/discharge rate of 0.2 C, where 1 C equals 3000 mA g−1, illustrating the impact of electrolyte choice on anode durability and efficiency. c Similar data for Si@C (silicon-carbon composite) anodes in varied electrolytes at 0.2 C, demonstrating the role of SEI optimization in enhancing the electrochemical performance of composite anodes. Reproduced with permission from Ref. [155]. Copyright 2023, Springer Nature |
3.1.2 Inorganic Material Deposition
3.1.3 Organic Material Coating
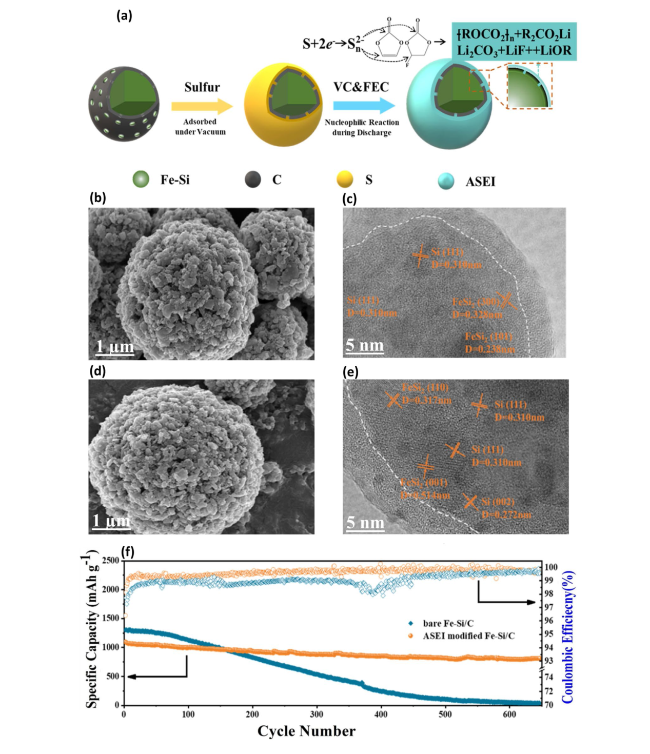
Fig. 13 Fabrication and Characterization of ASEI-Modified FeSi/C Anode for Enhanced Cycling Stability—a Fabrication process for the ASEI-modified FeSi/C anode, detailing the steps involved in creating the advanced sulfur-adsorbed electrode interface. b SEM and c HRTEM images of the pristine FeSi/C composite exhibit the initial microstructure and crystallinity. d SEM and e HRTEM images post sulfur adsorption on the FeSi/C composite showcase modifications in the surface and structural properties aimed at improving electrochemical performance. f Cycling stability and Coulombic efficiencies (CEs) of the ASEI-modified FeSi/C composite at a current density of 500 mA g−1, highlighting its enhanced durability and efficiency in battery applications. Reproduced with permission from Ref. [157]. Copyright 2021, Springer Nature |
3.2 Prelithiation of Anode Material
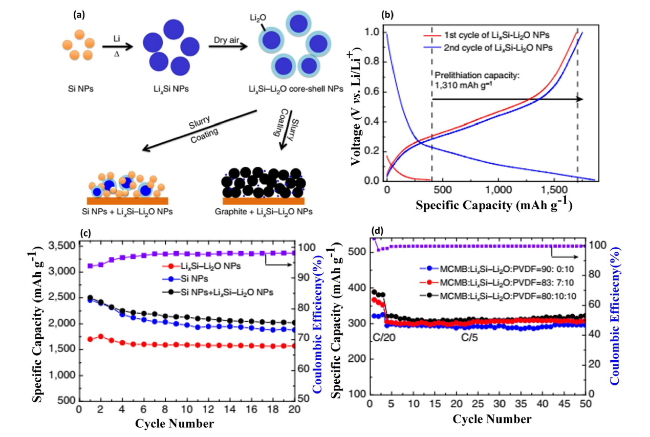
Fig. 14 Interaction of Si NPs with Melted Li and Electrochemical Performance of LixSi-Li2O Nanoparticles—a Schematic diagrams illustrate the reaction of silicon nanoparticles (Si NPs) with melted lithium (Li) to form lithium silicide (LixSi) nanoparticles, depicting the initial step in creating advanced anode materials. b Galvanostatic charge-discharge (GCD) profiles for LixSi-Li2O nanoparticles during the first and second cycles, indicating the electrochemical behavior and capacity retention. c Cycling performance of LixSi-Li2O nanoparticles, Si NPs/LixSi-Li2O composite, and control Si nanoparticles at a rate of C/20, with the purple line representing the Coulombic efficiency (CE) of the Si NPs/LixSi-Li2O composite, demonstrating the enhanced performance of the composite material. d Cycling performance of mesocarbon microbead (MCMB)/LixSi-Li2O composites with varying weight ratios, tested at C/20 for the first three cycles and C/5 for subsequent cycles (1 C = 0.372 A g−1, with capacity based on the total mass of active materials, including MCMB and Si in LixSi-Li2O nanoparticles). The purple line indicates the CE of the MCMB/LixSi-Li2O composite (80:10 by weight), highlighting the composite’s improved cycling stability. Reproduced with permission from Ref. [163]. Copyright 2017, Springer Nature |
3.3 Effect of Binder
3.3.1 Multifunctional Binders
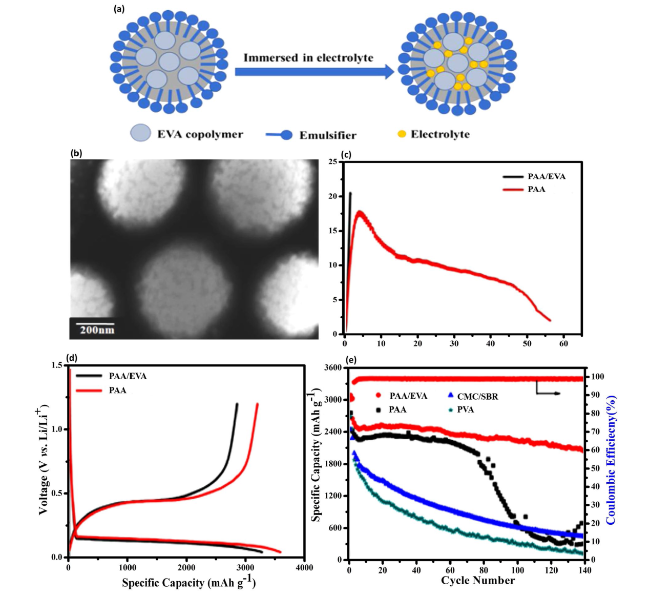
Fig. 15 Evaluation of EVA Colloid in Electrolyte for Enhanced Anode Performance—a Depicts the schematic illustration of the ethylene-vinyl acetate (EVA) colloid structure immersion process in the electrolyte, highlighting the preparation steps for modifying the electrode’s surface. b A TEM image showcases the microstructure of EVA latex, providing insight into the colloidal configuration. c Tensile curves for polyacrylic acid (PAA) and PAA/EVA films are presented, comparing the mechanical properties and flexibility of the films. The electrochemical performance of porous Si anodes using PAA/EVA and PAA as binders is examined through d the initial charge-discharge profiles of electrodes at a current density of 50 mA g−1, demonstrating the impact of binder selection on electrode capacity and efficiency. e Reversible capacity and Coulombic efficiency (CE) of electrodes at a current density of 50 mA g−1 for the first two cycles and at 500 mA g−1 for subsequent cycles, indicating the durability and performance enhancement provided by the PAA/EVA binder combination. Reproduced with the permission from Ref. [174]. Copyright 2019, American Chemical Society |
Table 6 Generalized carbon-Si composites for LIBs: evaluating scalability and commercial feasibility |
| Composite type | Strategy/composition | Description | Scalability potential | Commercial feasibility |
|---|---|---|---|---|
| 1D Carbon-Si Composites | CNFs and CNTs Integration with Si | Utilization of CNFs and CNTs to construct conductive networks and host matrices for Si-based anode materials, handling volume changes during charging/discharging. Examples include C-L-SC, Si@CNTs, and Si/CNFs | High scalability due to established manufacturing techniques for CNFs/CNTs | Good, considering growing demand in energy storage technologies |
| 2D Carbon-Si Composites | Graphene and MXene Integration with Si | Incorporation of graphene and MXenes with Si, resulting in structures like Si@G, Si@N-G, and MXene/Si@SiOx@C. Enhanced by the surface area and conductivity of graphene and the unique properties of MXenes | Moderate, challenges in large-scale production of quality graphene and MXenes | Promising, but dependent on cost reduction and production advancements |
| 3D Carbon-Si Composites | Porous Carbon and Graphite Integration with Si | Development of composites like gigaporous carbon microspheres and Si/graphite/carbon (Si-G/C) composites, leveraging the structural benefits of 3D carbon materials | High, given the existing large-scale production for porous carbon and graphite | Very feasible, especially in markets demanding high-performance batteries |
Table 7 Generalized nanostructured silicon anode materials: scalability and commercial prospects |
| Nanostructure type | Strategy/composition | Description | Scalability potential | Commercial feasibility |
|---|---|---|---|---|
| 0D Si-based anode materials | Si nanoparticles and composites | Utilization of porous Si nanoparticles and composites, including Si@TiO2 and Si/Ti2O3/rGO. Techniques include electroless etching, self-assembly, CVD | Moderate, challenges in consistent quality production at scale | Emerging, hinges on integration with current battery manufacturing processes |
| 1D Si-based anode materials | Si nanowires (SiNWs) and nanotubes (SiNTs) | Development of 1D structures like SiNWs and SiNTs using CVD and other methods. Examples include Si@NC, carbon-coated SiNWs | High for SiNWs with established methods; moderate for SiNTs due to complexity | Promising, particularly for high-end applications requiring advanced battery properties |
| 2D Si-based anode materials | Si thin films | Application of Si thin films on substrates, e.g., amorphous Si on Cu. Techniques include electrodeposition and magnetron sputtering | Moderate, dependent on deposition technologies and material handling | Feasible, with potential in niche markets and specialty applications |
| 3D Si-based anode materials | 3D macroporous Si structures | Creation of 3D macroporous Si using methods like magnesiothermic reduction and galvanic displacement. Examples include Si@C electrodes and hollow Si nanospheres | High, especially with advancements in 3D material synthesis techniques | Good, subject to demonstration of long-term durability and cost-effectiveness |
Table 8 Generalized SiOx/C composites and non-carbonaceous integrations: pathways to scalable and commercially viable LIB anodes |
| Material type | Strategy/composition | Description | Scalability potential | Commercial feasibility |
|---|---|---|---|---|
| SiOx/C composites | Encapsulation of SiOx in Carbon | Utilization of carbon matrices to encapsulate SiOx, enhancing electron/ion transport and forming a stable SEI layer. Examples include 3D-Si@SiOx/C, SiOx/C from rice husk | High, leveraging existing carbon material production infrastructures | Very promising, especially if cost-effectiveness is achieved |
| Si with non-carbonaceous materials | Core-shell structures with metals/metal oxides | Development of core-shell structures combining Si with metals/metal oxides like TiO2, SnO2, Nb2O5. Techniques include CVD, solvothermal methods | Moderate, challenges in uniform core-shell structuring at scale | Emerging, with potential in high-performance battery sectors |
Table 9 Generalized enhancements in Si-based anode materials: a focus on scalability and commercial application |
| Aspect | Strategy/Technique | Description | Scalability potential | Commercial feasibility |
|---|---|---|---|---|
| Artificial solid electrolyte interphase (ASEI) for Si anode | Engineered design of ASEI | Construction of artificial SEI layers using ex situ and in situ techniques to provide a robust protective layer on the anode | Moderate, requires precise control over layer formation | Promising, essential for high-capacity, long-life batteries |
| Prelithiation of anode material | Various prelithiation Techniques | Techniques like lithium foil contact (LFC), stabilized Lithium Metal Powder (SLMP), and chemical prelithiation to compensate for lithium loss | High, especially with advancements in lithiation technologies | Very feasible, can significantly enhance the market competitiveness of Si-based LIBs |
| Effect of binder | Use of advanced binders | Exploration of alternative binders (e.g., alginate, PAA, SHP) tailored for Si-based anodes to accommodate extensive volume changes | High, as alternative binders can be integrated into existing battery production lines | Very promising, especially for advanced batteries requiring high stability and performance |