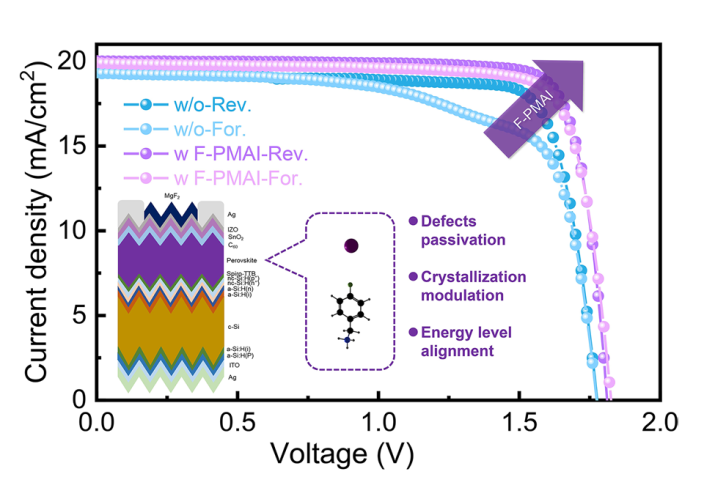
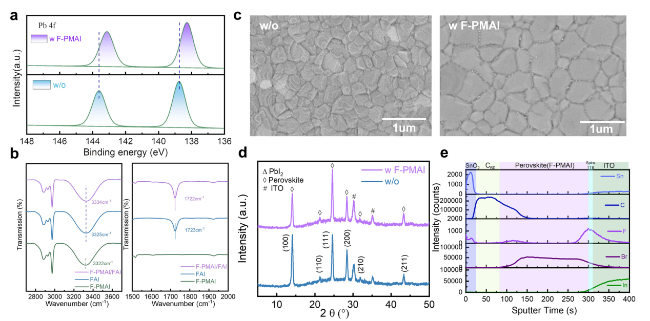
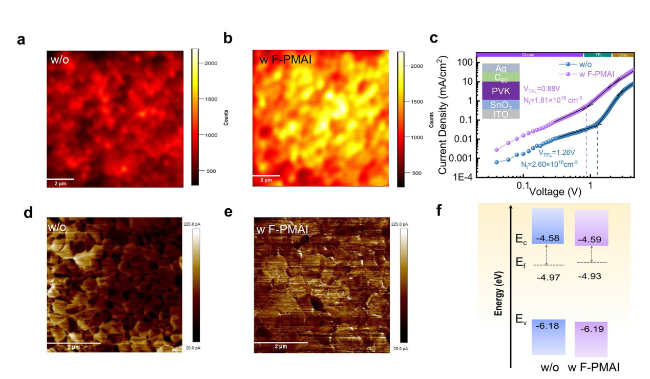
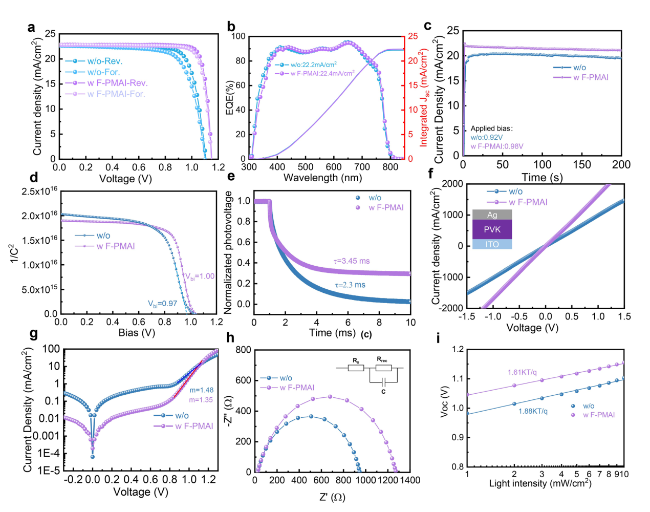
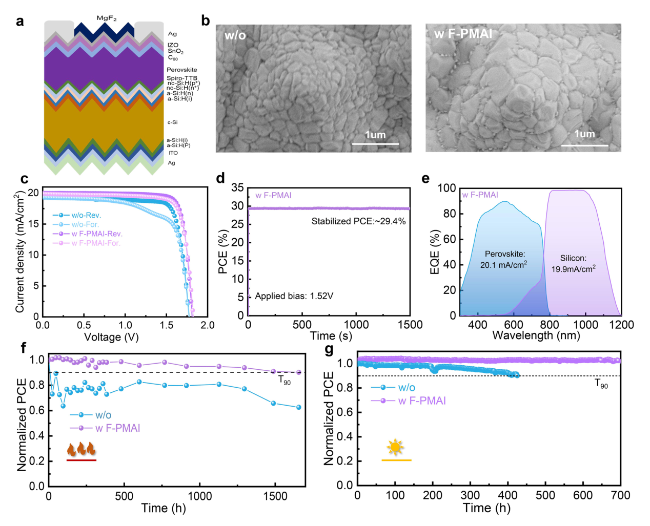
SEM and XRD were performed to reveal how F-PMAI affects perovskite film quality. The PbI
2 and CsCl were co-evaporated to form inorganic precursor films (Fig. S5a, b) and exhibit lead iodide (PbI
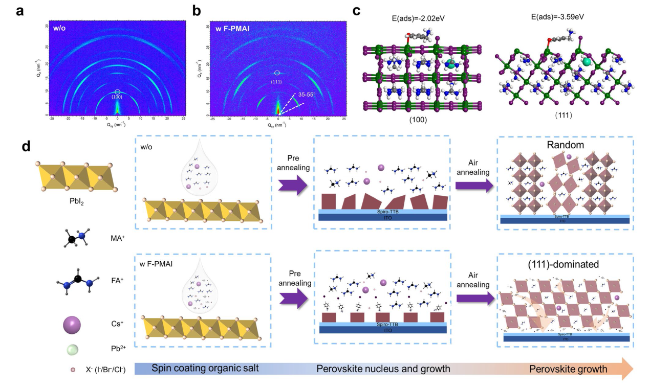
2)-dominated crystalline structure, as confirmed by the XRD peak near 12.5° (Fig. S5c). Before forming the final perovskite films, three stages including organic salts deposition, pre-annealing and air-annealing were gone through. As illustrated in
Fig. 1c, the air-annealing final perovskite film grains with F-PMAI were significantly enlarged than the control film. We also found perovskite films without and with F-PMAI exhibit different morphologies at organic salts deposition and pre-annealing stages as shown in Fig. S6. The control film shows a compact and randomly distorted morphology after drop-coating organic salts. Subsequently, the film further crystallized during the pre-annealing process and surface morphology has hardly changed (Fig. S6a), while for the additive film shows porous morphology after the addition of organic salts, and crystals began to precipitate in Fig. S6b. The difference in morphology demonstrated the much slower phase conversion for additive film, and the time of color change from yellow to black gave another evidence (Fig. S7). Notably, the peak area ratios of (001) PbI
2/(100) perovskite for the additive film are larger than the control film at the stage of organic salt deposition and pre-annealing as shown in Fig. S8, indicating that a slower crystalline process for the additive film. This delayed nucleation kinetics can be attributed to the hydrogen bond interaction between F
− and FA
+, thereby slowing down the reaction of PbI
2 and FAI. Moreover, we found the peak positions of additive perovskite films are similar to those of control films, indicating that the F-PMAI does not enter the lattice and change the lattice parameters of the perovskite (
Fig. 1d). There are no other diffraction peaks at low angles (2θ < 10°). Therefore, the addition of F-PMAI does not lead to the formation of 2D perovskite phase. The absence of 2D perovskite is beneficial for charge carrier transport and extraction throughout the perovskite absorber. ToF-SIMS were performed to observe the distribution of F-PMAI molecules in perovskite films, based on the structure ITO/Spiro-TTB/perovskite (F-PMAI)/C
60/ALD SnO
2 (
Fig. 1e). Interestingly, a rising peak of F signal at the perovskite and Spiro-TTB interface is observed. This result suggests that the F-PMAI molecules are mostly extruded to the buried interface. This is related to the stronger Lewis acid-base interactions between the F-PMAI and Spiro-TTB. In short, the F-PMAI with large bulk cation cannot incorporate into the lattice, and be repelled to bottom and top interface, severing as a defect passivator via passivating undercoordinated Pb
2+/I
− and A site vacancies defects. Hence, the schematic diagram of the F-PMAI interaction mechanism is shown in Fig. S9. F-PMA
+ passivates uncoordinated Pb
2+/I
− on the perovskite, inhibiting the production of deep-level defects and reducing the non-radiative recombination centers. F
− interacts with organic cations to form hydrogen bonds. In addition, I ions in F-PMAI also possibly fill the iodine vacancies [
34]. Above, we propose that the F-PMAI retards crystallization by hydrogen bond interaction, and the larger steric hindrance of F-PMAI hinders the immediate reaction between the organic salt (e.g., FAI) and the precursor film (e.g., PbI
2), improving the grain size. In terms of additive film, a higher critical Gibbs free energy (ΔG
c) to nucleate contributes to fewer crystal nuclei and growth in more room into larger grains. This agrees with the appearance of a porous morphology at the film initial growth stage (Fig. S6b). The slow crystallization in additive film can provide a longer diffusion distance and self-assembly time between precursor salt ions and organic molecules, conducive to the formation of more complete reactions and larger grains as shown in Fig. S10.